3.2
Impact Factor
ISSN: 1837-9664
J Cancer 2018; 9(7):1248-1258. doi:10.7150/jca.23967 This issue Cite
Research Paper
Overexpression of Klotho Inhibits HELF Fibroblasts SASP-related Protumoral Effects on Non-small Cell Lung Cancer Cells
1. Jiangsu Provincial Key Laboratory of Geriatrics, Department of Geriatrics, the First Affiliated Hospital with Nanjing Medical University, Nanjing 210029, China
2. Department of General Medicine, Zhongda Hospital, Southeast University, Nanjing 210009, China
3. Department of Mechanical Engineering, Johns Hopkins University, Baltimore 21218, USA
4. Department of Respiration, Nanjing First Hospital, Nanjing Medical University, Nanjing 210006, China
Received 2017-11-21; Accepted 2018-2-28; Published 2018-3-14
Abstract
Lung cancer (LC) is the most common cause of death from cancer worldwide, and it is also a closely aging-related disease. Klotho, a new anti-aging gene, has been proven to play a critical role in regulating aging and the development of age-related diseases including LC. However, whether Klotho is a key link between aging and LC is still unknown. Here we report that Klotho can indirectly inhibit LC growth and development through regulating senescence-associated secretory phenotype (SASP). We found that senescent lung fibroblasts (SLF) can promote production of IL-6 and IL-8, which can be effectively inhibited by overexpressing Klotho. Using conditioned medium (CM) derived from SLF to culture LC cells, the LC cells show obvious increase of viability and migration rates, significant increase expression of p-STAT3 and α-SMA, and decrease expression of P53 and E-cadherin. However, using CM derived from SLF overexpressed Klotho to culture LC cells, all above results are nearly completely reversed. Thus, these results suggest that Klotho can regulate SLF extracellular release of IL-6 and IL-8, which can influence STAT3 activation, P53 expression and epithelial-mesenchymal transition (EMT) of LC cells, finally inhibiting LC cells growth and migration indirectly.
Keywords: Klotho, cellular senescence, lung cancer, IL-6
Introduction
Non-small cell lung cancer (NSCLC) is a leading cause of death among cancer patients, with 5-year survival rates less than 10%. Incidence and mortality rates increase exponentially in adults older than 65 years compared to their younger counterparts. Median age at diagnosis and death is 70 and 72 years, respectively [1]. Thus, lung cancer (LC) is an aging-related disease. It is of great importance to explore potential associations between LC and aging, which may have implications for lung cancer prevention and treatment.
Recently, Klotho, a new anti-aging gene, has gained great attention. The Klotho gene plays a critical role in regulating aging and the development of age-related diseases: Loss of Klotho can result in multiple aging-like phenotypes, while overexpression of Klotho gene extends lifespan by 20-30% [2, 3]. Klotho can work as a cofactor/coreceptor regulating fibroblast growth factor (FGF) 23 signaling, and can also influence intracellular signaling pathways including insulin/IGF-1, p53/p21, cAMP, protein kinase C (PKC) and Wnt signaling pathways. Thus, the discovery of Klotho has a great impact on aging research. However, the role of Klotho in tumorigenesis, cancer progression, and prognosis is attracting more and more attention. Recent studies have shown that Klotho participates in the progression of several types of human cancers, including breast, lung, liver, gastric, and other cancers [4]. Klotho functions as a tumor suppressor mainly by regulating insulin/IGF-1, p53/p21, and Wnt signaling pathways. Thus, Klotho involves in multiple biological processes, especially in anti-aging and inhibition of cancer, and it may be a potential key link between cancer and aging.
There is substantial evidence that aging-related changes can influence stromal-epithelial interactions leading to a microenvironment permissive for tumor growth [5]. Cancer is typically a disease of old age, and there is increasing evidence that senescence within the stroma, particularly in the fibroblast compartment, can drive tumor development. A number of studies have shown that senescent fibroblasts stimulate premalignant and malignant epithelial cells to grow in cell culture and to form tumors in animal models [6-9]. These involve the secretion of factors from senescent fibroblasts, named as senescence-associated secretory phenotypes (SASP) by Coppé and colleagues [7]. The secretory factors include matrix metalloproteinase (MMP) , IL-6, IL-8, TGF-β, etc., which in turn remodel the microenvironment, alter epithelial differentiation, promote endothelial cell motility, and stimulate tumor cell growth [10].
Our previous studies demonstrated that Klotho can directly inhibit LC cells growth and invasion by regulating insulin/IGF-1, Bax/Bcl-2, Wnt/β-catenin and PI3K/AKT signaling pathways [11-14]. However, whether Klotho can indirectly affect LC by remodeling tumor microenvironment is still unknown. In this study, we sought to characterize the molecular alterations that occur during the process of lung fibroblasts senescence to identify factors in the aged tissue microenvironment capable of promoting the proliferation and potentially the neoplastic progression of lung epithelium. We evaluated two mechanisms leading to cell senescence (i.e., oxidative stress and DNA damage) and identified a common and consistent program of gene expression that includes a subset of paracrine factors capable of affecting adjacent lung epithelial growth. Using in vitro fibroblasts co-culture experiments, we also learned that these changes help the growth and invasion of LC cells. And Klotho can alter microenvironment of senescent lung fibroblasts (SLF), finally influencing LC growth and invasion. These results support the concept that aging-related changes in the lung microenvironment may contribute to the genesis and progression of LC, and Klotho can prevent normal lung from aging-related changes and tumorigenesis.
Materials and methods
Cell culture
Human embryonic lung fibroblast (HELF) cell line and human lung adenocarcinoma cell line (A549) were purchased from the ATCC (American Type Culture Collection) , cultured in Dulbecco's modified Eagle's medium (Invitrogen), supplemented with 10 mM HEPES (Invitrogen), 5 mM L-glutamine (Invitrogen) and 10% fetal bovine serum (Invitrogen) in a humidified atmosphere of sterile air, 5.0% CO2 at 37℃.
Induction of lung fibroblasts senescence
Bleomycin treatment. Bleomycin (Nippon Kayaku Co., Ltd.) was used to induce senescence of fibroblasts. Cells were incubated with 100 μg/mL bleomycin for 24 h in complete medium. After treatment, the cells were rinsed thrice with phosphate-buffered saline (PBS), left to recover 3 days in complete medium, and collected for different assays. Following recovery, the cells were designated HELF-BS (BS).
Hydrogen peroxide treatment. Hydrogen peroxide (H2O2; BDH Prolabo) was used to induce senescence of fibroblasts. Cells were incubated with 550 μM H2O2 for 1.5 h in complete medium. After treatment, the cells were rinsed thrice with PBS and cultured in complete medium for 4 days. Cells were then treated with 200 μM H2O2 for 2 h, washed with PBS and cultured for additional 4-6 days. After 10 days from the first treatment, cells were collected and processed for different assays. Following recovery, the cells were designated HELF-HS (HS).
Senescence-associated beta-galactosidase activity (SA-β-gal) assay
SA-β-gal activity was measured with a β-gal staining kit (Cell Biolabs Inc.) according to the manufacturer's instructions. Briefly, treated cells were washed twice with PBS and fixed with fixative solution for 15 minutes at room temperature. The fixed cells were washed with PBS and stained with the staining solution containing X-gal and staining supplement and incubated at 37°C overnight without CO2. Cells containing blue stain were counted as senescent, proportionate to the total number of cells per field of vision. At least 300 cells in at least 4 fields of vision were counted under a phase contrast microscope.
Plasmids and transfection
The MYC-tagged Klotho expression vector (pCMV6-MYC-KL, KL) and its entry vector (pCMV6-Con, NC) were designed and purchased from OriGene (Rockville). Transfection of constructs in HELF cells was performed using Lipofectamine 3000 Reagent (Invitrogen), according to the manufacturer's recommendation. Stable clones were generated by selection in complete culture medium containing 700μg/ml G418.
Collection of conditioned medium (CM)
Confluent cultures of treated HELF cells were rinsed thrice in PBS and incubated for 3 days in DMEM + 0.5% fetal calf serum (FCS). The supernatant was harvested and stored frozen at -80℃. Conditioned medium was thawed and diluted 1:1 with fresh DMEM + 0.5% FCS before use.
Enzyme-linked immunosorbent assay (ELISA)
ELISAs were performed using Human Quantikine ELISA Kit (IL-6 #D6050, IL-8 #D8000C, TGF-β #DB100B; R&D Systems) according to the manufacturer's instructions. Data were normalized and expressed as picograms per milliliter.
Western blot assay
Protein level was measured by western blot. Briefly, equal amounts of protein (10μg per lane) were separated by SDS-PAGE electrophoresis, and transferred to PVDF membrane (Bio-Rad Inc.). The membranes were incubated in blocking buffer (0.2 mM Tris, 137 mM NaCl, 5% no-fat milk, and 0.1% Tween-20) for an hour and then probed at 4℃overnight with relevant antibody. The membranes were rinsed with washing buffer (0.1% Tween 20, 0.2 mM Tris, and 137 mM NaCl) and incubated with HRP-conjugated secondary antibody for an hour at room temperature, followed by chemiluminescent detection. Antibodies against Klotho (Santa Cruz), GAPDH, phosphor-STAT3, total STAT3, P53, α-SMA and E-cadherin (all purchased from Cell Signaling) were used to probe the blot.
Culture of human lung adenocarcinoma cell line with senescent fibroblasts CM
The CM was thawed and diluted 1:1 with fresh DMEM + 0.5% FCS before use. A549 cells were seeded in six-well plates in CM or fresh DMEM + 0.5% FCS. The cultures were incubated for 4 days and the cell viability and motility were separately explored.
Cell viability assasy
Cell viability was assessed by 3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT; Sigma) assay according to the manufacturer's instructions. A549 cells were cultured with aforementioned different groups of CM for 24, 48, 72 and 96h, respectively. The absorbance was evaluated by OD values at 450 nm using a microplate reader (Bio-Rad Co.).
Scratch motility assay
A549 cells were cultured with aforementioned different groups of CM and grew to 80% confluency. Then the monolayer was scratched with a pipette tip to create a cell free strip area, washed with PBS gently to remove floating cells and took photograph at three random locations (0h). The microscope photograph of migration was taken every 24 h for 48h at the same locations.
Statistical analysis
Statistical significance was assessed using Student's t-test or analysis of variance (ANOVA). Data were presented as mean ± standard deviation (SD), and values of p < 0.05 were considered statistically significant. All the experiments were repeated at least in triplicate.
Results
Overexpression of Klotho attenuates oxidative stress- and chemotherapy exposure-induced lung fibroblasts senescence
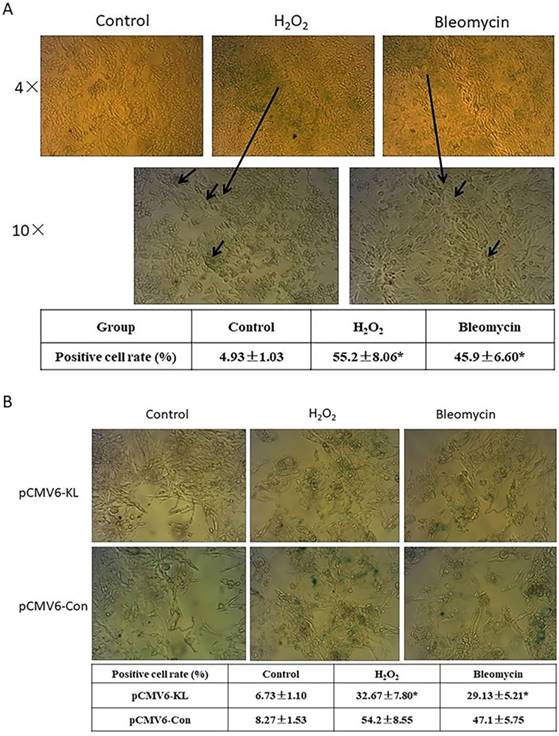
Several factors have been shown to induce a phenotype of cellular senescence. In this study, we evaluated two senescence mechanisms that lung fibroblasts can reasonably encounter in their natural environment: oxidative stress and DNA damage due to chemotherapy exposure. We studied human embryonic lung fibroblast cell line HELF to determine the phenotypic and gene expression features of the senescence program in this cell type. To verify a senescence phenotype associating with each mechanism, we visually inspected cell cultures for morphologic features of senescence and measured expression of β-Gal by staining at pH 6 (SA-β-Gal) (Fig. 1A). Morphologic changes previously associated with senescence, including cell enlargement and flattening, were clearly apparent. The proportion of SA-β-Gal positive cells in H2O2- and bleomycin-treated groups is obvious higher compared with control groups (Fig. 1A). The results indicated that the reagents we used successfully induced HELF senescence.
Klotho, a widely recognized anti-aging protein, which are found in recent years, shows different roles in tissues. We explored potential anti-aging function in oxidative stress and DNA damage-induced SLF by overexpression of Klotho. The results indicated that Klotho can obviously inhibit the proportion of SA-β-Gal positive cells both in H2O2- and bleomycin-induced SLF groups when compared with their corresponding control groups (Fig. 1B). In contrast, Klotho had no significant effects in normal control lung fibroblast cells (Fig. 1B).
Induction of lung fibroblasts senescence and exploring potential roles of Klotho in senescent lung fibroblasts (SLF). (A) Induction of HELF cells senescence. HELF cells were respectively treated with H2O2 and bleomycin, then we visually inspected cell cultures for morphologic features of senescence and measured expression of β-Gal by staining at pH6 (SA-β-Gal). *p<0.05, H2O2- or bleomycin-treated group vs. Control group. (B) Effects of Klotho on H2O2- and bleomycin-induced senescence were evaluated with SA-β-Gal assay. H2O2- or bleomycin-induced senescent HELF cells were transfected with pCMV6-KL (KL) or pCMV6-Con (NC) plasmids, followed with SA-β-Gal assay. *p<0.05, KL group vs. NC group.
Overexpression of Klotho inhibits activation of STAT3 signaling pathway, and decreases senescence-induced production of IL-6 and IL-8 in SLF
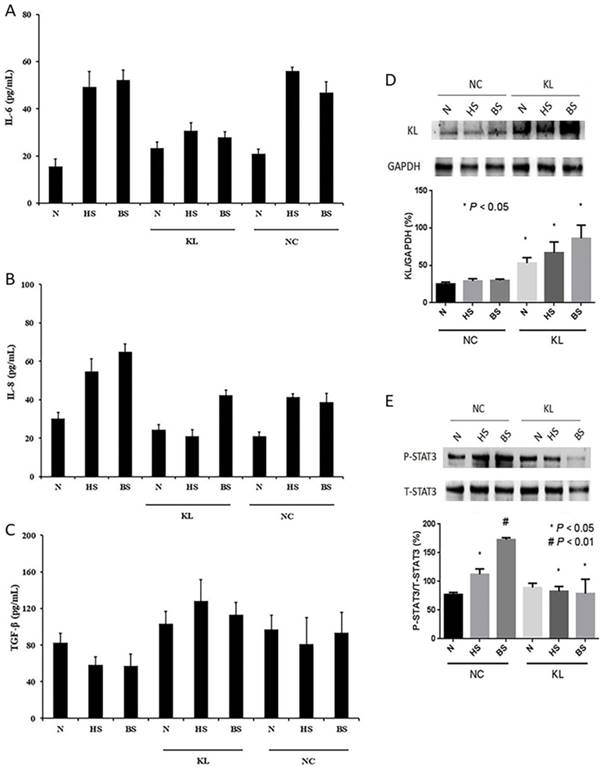
Substantial evidence show that senescence-associated transcript alterations called SASP (especially IL-6, IL-8 and TGF-β) can affect tumor microenvironment, which can influence genesis and development of cancer. To verify that senescence-associated changes in extracellular protein levels, and to explore potential effects of Klotho in senescent HELF cells, we firstly overexpressed Klotho in normal and senescent HELF groups, and then evaluated CM obtained from senescent HELF for the presence of IL-6, IL-8, and TGF-β by ELISA. The results showed that expression levels of IL-6 and IL-8 in both H2O2- and bleomycin-induced groups are significant higher than normal control groups (Fig. 2A and B). However, when overexpression with Klotho, the expression levels of IL-6 and IL-8 in senescence groups were significant decrease compared with their corresponding control groups (Fig. 2A and B). Meanwhile, we did not find any significant difference in TGF-β expression levels between senescence groups and normal groups, no matter with or without overexpression of Klotho (Fig. 2C). The results indicated that Klotho can inhibit senescence-associated changes in extracellular IL-6 and IL-8 protein levels.
Next, we explored potential mechanism of aforementioned roles of Klotho. Several studies indicated STAT3 pathways involved in IL-6 and IL-8 expression, thus, we evaluated whether STAT3 pathway appeared changes in senescent HELF cells and whether Klotho can affect STAT3 activity. We firstly transfected pCMV6-KL (KL) or pCMV6-Con (NC) plasmids in different groups of HELF, and western blot results showed that we successfully overexpressed Klotho in HELF cells (Fig. 2D). We found that phosphorylation levels of STAT3 are much higher in senescent HELF groups than that of normal HELF groups (Fig. 2E). However, when overexpression of Klotho, the phosphorylation levels of STAT3 in both H2O2- and bleomycin-induced senescence HELF groups are significant decrease compared with their corresponding control groups (Fig. 2E). Thus, the results indicated that Klotho can inhibit oxidative stress- and DNA damage-induced activation of STAT3 in senescent HELF cells.
Klotho decreases senescence-induced production of IL-6 and IL-8 in SLF, partly through inhibiting activation of STAT3 signaling pathway. (A) IL-6 levels of HELF cells treated with pCMV6-KL (KL) or pCMV6-Con (NC) plasmids were evaluated by ELISA assay. p<0.01, HS or BS vs. N; p<0.01, HS-KL or BS-KL vs. HS-NC or BS-NC, respectively. (B) IL-8 levels of HELF cells treated with KL or NC were evaluated by ELISA assay, and the results are similar as IL-6. (C) TGF-β levels of HELF cells transfected with KL or NC were evaluated by ELISA assay. TGF-β results did not show any significant difference in all groups. (D) Different groups of HELF cells were transfected with KL or NC, and the Klotho protein levels were evaluated by western blot. *p<0.05, N, HS and BS transfected with KL vs. N, HS and BS transfected with NC, respectively. (E) STAT3 pathway activities were evaluated in different HELF groups transfected with KL or NC plasmids. P<0.05, HS-NC vs. N; p<0.01, BS-NC vs. N; p<0.05, HS-KL vs. HS-NC; p<0.05, BS-KL vs. BS-NC. N, normal; HS, H2O2-induced senescence; BS, bleomycin-induced senescence; KL, pCMV6-KL transfected; NC, pCMV6-Con transfected.
Prognostic values of KL, IL-6 and IL-8 expression in non-small cell lung cancer (NSCLC) patients were assessed by using the Kaplan-Meier (KM) plotter integrative data analysis tool (http://www.kmplot.com). (A) Overall survival (OS) of lung adenocarcinoma (LUAD) and lung squamous cell carcinoma (LUSC) patients with different expression levels of KL. (B) Overall survival of LUAD and LUSC patients with different expression levels of IL-6. (C) Overall survival of LUAD and LUSC patients with different expression levels of IL-8.
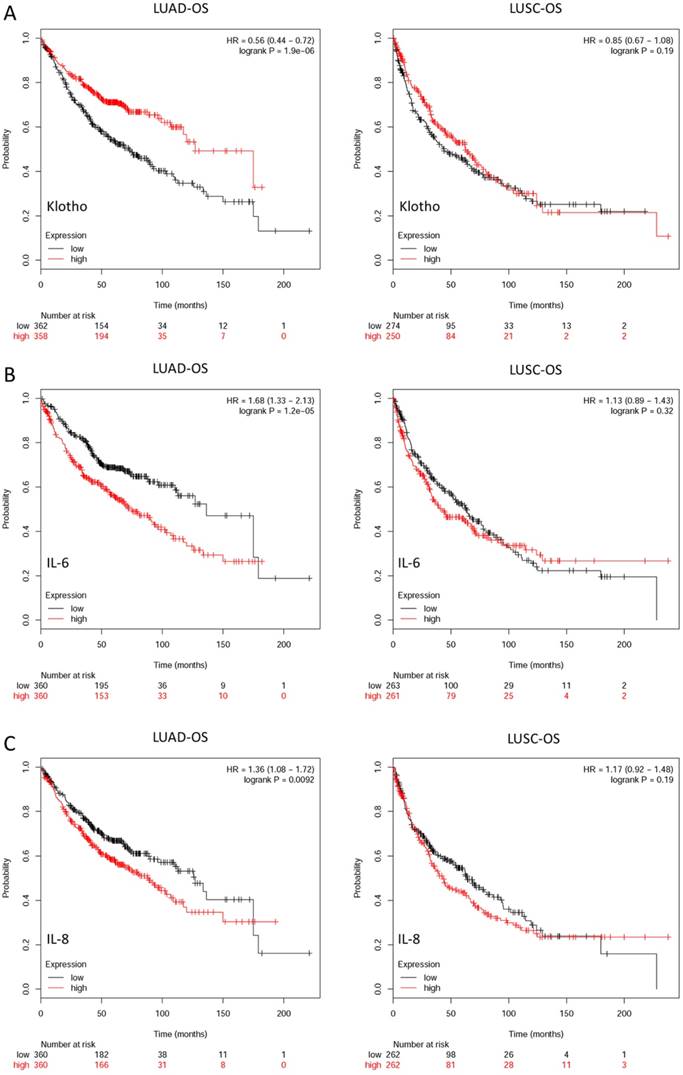
Expression levels of Klotho, IL-6 and IL-8 are closely correlated with overall survival of NSCLC patients
Multiple studies report that anti-aging gene Klotho and SASP-related secretary factor IL-6 and IL-8 are involved in different carcinogenesis. In this study, we found that SLF can induce production of IL-6 and IL-8, and overexpression of Klotho can inhibit this process. To verify potential roles of aforementioned three genes in NSCLC patients, we evaluated and analyzed the relevant databases of the online Kaplan-Meier plotter (www.kmplot.com). This is a website which can assess the survival effect of gene or gene combination in lung, breast, ovarian and gastric cancer. The gene expression data and relapse free and overall survival information are downloaded from GEO, EGA and TCGA database. The effects of KL, IL6 and IL-8 mRNA expression levels on survival of lung adenocarcinoma (LUAD) and lung squamous cell carcinoma (LUSC) patients were assessed from Kaplan Meier plotter, including 1928 cancer samples, of which there were 866 LUAD samples and 675 LUSC samples. The corresponding valid Affymetrix IDs of KL, IL-6 and IL-8 are 205978_at, 205207_at and 202859_x_at. The Kaplan-Meier survival curves and relevant statistical results are shown in Fig. 3. The results indicated that expression levels of all the three genes are closely correlated with OS of LUAD patients, higher expression level of Klotho and lower expression levels of IL-6 and IL-8 patients show better OS status. However, there are no significant differences in LUSC patients.
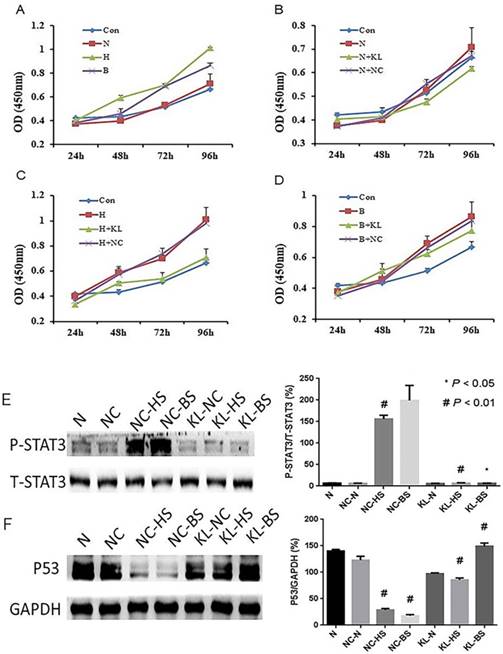
Proliferation and relevant mechanism of A549 cells cultured with conditioned medium (CM) which derived from different HELF groups were evaluated. (A-D) A549 cells were cultured with different HELF groups derived CM, and A549 cells viability were determined by MTT assay. The results indicated senescent HELF derived CM could promote A549 cells proliferation, and these effects can be effectively inhibit when HELF transfected with KL. Con, normal cell medium DMEM; N, normal HELF derived CM; H, H2O2-induced senescent HELF derived CM; B, bleomycin-induced senescent HELF derived CM; N+KL, KL-transfected normal HELF derived CM; N+NC, NC-transfected normal HELF derived CM; H+KL, H2O2-induced senescent with KL-transfected HELF derived CM; H+NC, H2O2-induced senescent with NC-transfected HELF derived CM; B+KL, bleomycin-induced senescent with KL-transfected HELF derived CM; B+NC, bleomycin-induced senescent with NC-transfected HELF derived CM. (E) STAT3 pathway activity in A549 cells cultured with different HELF groups-derived CM was evaluated. #p<0.01, NC-HS or NC-BS vs. NC-N, KL-HS vs. NC-HS; p<0.05, KL-BS vs. NC-BS. (F) Expression of P53 in A549 cells cultured with different HELF groups-derived CM was evaluated. #p<0.01, NC-HS or NC-BS vs. NC-N, KL-HS vs. NC-HS and KL-BS vs. NC-BS.
Klotho indirectly inhibits LC cells proliferation through regulating SLF extracellular production of IL-6 and IL-8
Studies reported that senescence-associated changes in extracellular protein could affect cancer viability, so we follow-up evaluated whether oxidative stress and DNA damage-induced senescence-associated changes in extracellular protein can influence viability of human lung adenocarcinoma cell line A549, and also explored whether Klotho can regulate this biological process. We respectively collected CM from aforementioned normal HELF cells, senescent HELF cells with or without KL- or NC-transfected, and cultured A549 cells with these CM. The results indicated that CM from H2O2- or bleomycin-induced senescent HELF cells can obviously promote proliferation of A549 cells, while CM from KL-transfected senescent HELF cells can effectively inhibit proliferation of A549 cells when compared with that from NC-transfected cells (Fig. 4A-D).
Multiple studies reported that IL-6 can promote caner cancer cells growth mainly by regulating STAT3 activity. In addition, recently, a study shows that IL-6 downregulated the expression and activity of p53 in transformed and untransformed human cell lines [15]. Thus, we wondered whether the aforementioned effects of CM in A549 cells were partly due to changes of STAT3 and P53. And we found that phosphorylation levels of STAT3 of A549 cells are much higher using CM from senescent HELF cells groups than that from normal CM groups (Fig. 4E). While using KL-transfected CM to culture A549 cells, the phosphorylation levels of STAT3 are obviously inhibited when compared with their control groups (Fig. 4E). Meanwhile, we found that using senescent HELF cells CM can effectively inhibit P53 expression of A549 cells, while KL- transfected HELF cells derived CM can significantly promote P53 expression of A549 cells (Fig. 4F).
Migration and relevant mechanism of A549 cells cultured with CM which derived from different HELF groups were evaluated. (A) Scratch motility assay was performed to evaluate different groups of A549 cells cultured with different CM. (B) Quantitative assays were performed by calculating cell numbers. The ratio of migrating cells was calculated into histogram. The results indicated that senescent HELF derived CM can promote A549 cells migration, and these effects can be effectively inhibited when HELF transfected with KL. #p<0.01, KL-HS vs. NC-HS; *p<0.05, KL-BS vs. NC-BS. (C) Expression of EMT relevant markers α-SMA and E-cadherin in A549 cells cultured with aforementioned different groups of CM was evaluated by western blot assay. α-SMA: #p<0.01, NC-HS or NC-BS vs. NC-N, KL-HS vs. NC-HS, KL-BS vs. NC-BS; E-cadherin: #p<0.01, NC-HS vs. NC-N; *p<0.05, NC-BS vs. NC-N, KL-HS NC-HS, KL-BS vs. NC-BS.
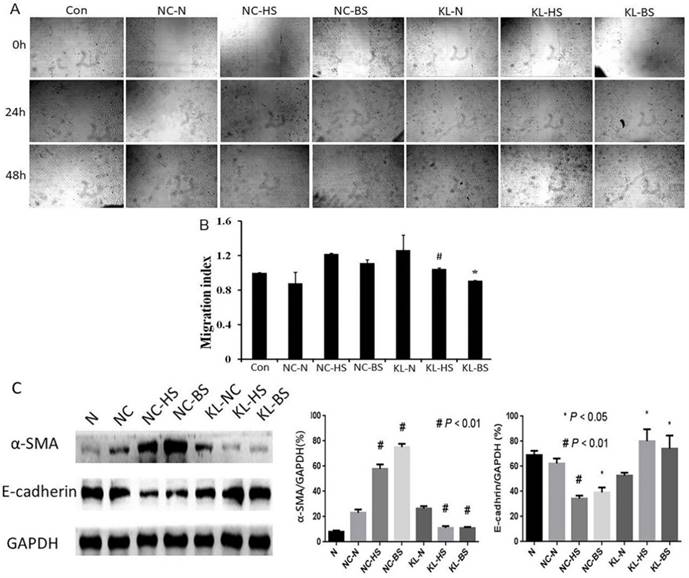
Klotho indirectly inhibits LC cells epithelial-mesenchymal transition (EMT) by regulating SLF extracellular production of IL-6 and IL-8
Similar to the cell viability, we evaluated effects of aforementioned different groups of CM in migration of LC cells, and also explored whether Klotho can regulate this biological process. In view of cancer cells metastasis and migration often involve EMT, we tested relevant indicators. We found that CM from H2O2- or bleomycin-induced senescent HELF cells can promote migration of A549 cells, while CM from KL-transfected senescent HELF cells can effectively inhibit migration of A549 cells when compared with that from NC-transfected cells (Fig. 5A and B).
Next, we tested EMT relevant markers α-SMA and E-cadherin. The results showed that proteins levels of α-SMA are significant higher while E-cadherin are lower in A549 cells cultured with CM from senescent HELF cells groups than that from normal CM groups (Fig. 5C). When using KL-transfected senescent HELF derived CM to culture A549 cells, the expression of α-SMA obviously decrease while E-cadherin increase compared with their corresponding control groups (Fig. 5C).
Discussion
There is a striking correlation between advanced age and increased incidence of cancer, as cancer incidence rises approximately exponentially after fifth decade in humans [16]. Cellular senescence is a potent anticancer mechanism that arrests the proliferation of cells at risk for neoplastic transformation. However, several lines of evidence suggest that SASP may contribute to tumorigenesis. The SASP can create a protumorigenic microenvironment in several ways. First, SASP factors support tumor cell invasion and metastasis by disrupting and remodeling tissue structure [7, 17]. Second, senescent cells can directly or indirectly promote tumor vascularization [18, 19]. Third, the SASP promotes tumor growth by establishing a microenvironment that is immunosuppressive [20]. These various activities of the SASP make it an fascinating target for novel adjuvant anti-cancer therapies.
Klotho is a new anti-aging gene, which can be involved in multiple biological processes, and the precise mechanism has been widely investigated [21]. It is now widely accepted that Klotho inhibits insulin and IGF-1 signaling pathways [3, 22]. Moderate inhibition of the insulin/IGF-1 signaling pathways has been viewed as one of the evolutionarily conserved mechanisms for suppressing aging [23]. Klotho functions as a co-receptor for FGF23, which down-regulates the expression of 1,25-dihydroxyvitamin D3 and phosphate reabsorption [24]. In addition, Klotho can also increase the resistance to oxidative stress [25]. Intriguingly, more and more studies found that Klotho can function as a tumor suppressor in multiple types of cancers [4]. Our previous results showed that Klotho can inhibit LC cells growth, migration, promote its apoptosis, and improve its sensitivity to chemotherapeutic drugs through regulating the insulin/IGF-1, Bax/Bcl-2, Wnt/β-catenin and PI3K/AKT signaling pathways in NSCLC [11-14]. Thus, Klotho plays a critical role in both aging and cancer, it could be a key link between them. However, our previous studies focused on exploring Klotho's direct function in LC cells, we have not known whether Klotho can indirectly affect LC by regulating tumor microenvironment.
Cellular senescence represents an aging-associated process and senescent cells accumulate in tissues with age [26]. Although senescent and tumor-associated reactive stromal fibroblasts differ in growth potential and morphology, they share the ability to stimulate the proliferation and invasion of initiated epithelial cells through direct contact or secreted factors. Several studies reported [27-29] that the ability of senescent human fibroblasts to promote the growth and tumorigenesis of premalignant and malignant lung epithelial cells, a finding that provides a mechanistic link between stromal aging and carcinogenesis.
In this study, we also undertook our experimental investigation of molecular alterations that occur during the process of lung fibroblast senescence to identify factors in the aged tissue microenvironment capable of promoting the proliferation and potentially the neoplastic progression of LC. Meanwhile, we explored the potential role of Klotho in this biological process.
It is well known that aged or senescent cells develop a complex SASP, which is observed both in vitro and in vivo. However, the mechanisms underlying the induction of the SASP are largely unknown. Liu et al. [30] demonstrated that membrane-bound Klotho prevents the retinoic-acid-inducible gene-I-induced expression of IL-6 and -8, suggesting that the anti-aging function of Klotho also includes the suppression of inflammation. In addition, another research [31] showed that with PAI-1 deficiency, levels of both IGFBP-3 and IL-6 were normalized in Klotho-deficient mice. This confirms and extends the recent observations by López-Andrés et al. that modulating the activity of a key member of the senescence-messaging secretome (SMS) can also normalize the levels of other SMS factors [32]. These indicate that Klotho can regulate SASP.
Work by Lee et al. has shown that exposure to the pro-inflammatory cytokine IL-6 commonly expressed in tumors was sufficient to induce Twist1 expression in normal fibroblasts and transdifferentiate them into cancer-associated fibroblasts (CAF) via STAT3 phosphorylation [33]. In this study, we found that SLF can promote secretion of IL-6 and IL-8, and Klotho can effectively inhibit senescence of the cells, and decrease production of IL-6 and IL-8, partly by inhibiting activation of STAT3 pathway. Which may follow-up alleviate transdifferentiation of lung fibroblasts into CAF.
IL-6 and IL-8 have been reported to increase in a variety of tumors, and contributes to aggressive tumor growth and resistance to chemotherapy and radiotherapy [34-36]. These processes mainly involve phosphorylation activation of STAT3 signaling pathway. In our study, we also found that when using SLF derived CM to culture LC cells A549, the proliferation rate of A549 cells is much higher than that derived from normal fibroblasts. Meanwhile, KL-treated SLF derived CM could effectively inhibit A549 cells proliferation. In addition, these changes were closely related with activation of STAT3. Thus, Klotho may indirectly inhibit LC cells growth by regulating extracellular secreted factors of SLF.
Brighenti et al. reported that IL-6 downregulated the expression and activity of p53 in transformed and untransformed human cell lines. This was the consequence of IL-6-dependent stimulation of c-MYC mRNA translation, which was responsible for the upregulation of rRNA transcription. The enhanced rRNA transcription stimulated the MDM2-mediated proteasomal degradation of p53, by reducing the availability of ribosome proteins for MDM2 binding. The p53 downregulation induced the acquisition of cellular phenotypic changes characteristic of EMT, such as a reduced level of E-cadherin expression, increased cell invasiveness and a decreased response to cytotoxic stresses (15). In our study, when using CM derived from SLF with or without Klotho transfection to culture LC cells, the results showed that LC cells proliferation were closely related with their P53 expression levels. Senescent group showed decreasing expression of P53, while KL-treated group showed increasing expression P53. Thus, this may partly because Klotho can inhibit IL-6 secretion, which finally leading to decrease MDM2-mediated proteasomal degradation of P53 as aforementioned mechanism. In addition, we also found that KL-treated CM from senescent fibroblasts can effectively inhibit LC cells migration with E-cadherin increase and α-SMA decrease, which are characteristic of EMT. Thus, Klotho could indirectly inhibit LC cells growth and invasion by regulating extracellular secreted factors of SLF, which finally influences STAT3 activation and P53 expression of LC cells.
In conclusion, our results support that aging-related changes (SASP, such as IL-6 and IL-8) in the lung microenvironment could contribute to the genesis and progression of LC, while Klotho can regulate SLF release of IL-6 and IL-8, which can influence STAT3 activation and P53 expression of LC cells, finally inhibiting LC cells proliferation and migration indirectly. Thus, Klotho may represent a novel therapeutic candidate for the treatment of LC.
Abbreviations
LC: lung cancer; SASP: senescence-associated secretory phenotype (SASP); SLF: senescent lung fibroblasts; CM: conditioned medium; EMT: epithelial-mesenchymal transition; NSCLC: non-small cell lung cancer; FGF: fibroblast growth factor; PKC: protein kinase C; MMP: matrix metalloproteinase; HELF: human embryonic lung fibroblast; PBS: phosphate-buffered saline; CAF: cancer-associated fibroblasts; SMS: senescence-messaging secretome; SA-β-gal: senescence-associated beta-galactosidase activit; ELISA: enzyme-linked immunosorbent assay.
Acknowledgements
This study was supported by the International Science & Technology Cooperation Program of China (No. 2014DFA31940); The National Natural Science Foundation of China (Nos. 81572259 and 81302011); The Jiangsu Province's Youth Medical Talents Program (QNRC2016593); The Jiangsu Provincial Key Discipline of Medicine (ZDXKA2016003).
Competing Interests
The authors have declared that no competing interest exists.
References
1. Didkowska J, Wojciechowska U, Manczuk M. et al. Lung cancer epidemiology: contemporary and future challenges worldwide. Ann Transl Med. 2016;4:150
2. Kuro-o M, Matsumura Y, Aizawa H. et al. Mutation of the mouse klotho gene leads to a syndrome resembling ageing. Nature. 1997;390:45-51
3. Kurosu H, Yamamoto M, Clark JD. et al. Suppression of aging in mice by the hormone Klotho. Science. 2005;309:1829-33
4. Zhou X, Wang X. Klotho: a novel biomarker for cancer. J Cancer Res Clin Oncol. 2015;141:961-9
5. Elkhattouti A, Hassan M, Gomez CR. Stromal Fibroblast in Age-Related Cancer: Role in Tumorigenesis and Potential as Novel Therapeutic Target. Front Oncol. 2015;5:158
6. Bhowmick NA, Neilson EG, Moses HL. Stromal fibroblasts in cancer initiation and progression. Nature. 2004;432:332-7
7. Coppe JP, Patil CK, Rodier F. et al. Senescence-associated secretory phenotypes reveal cell-nonautonomous functions of oncogenic RAS and the p53 tumor suppressor. PLoS Biol. 2008;6:2853-68
8. Krtolica A, Campisi J. Cancer and aging: a model for the cancer promoting effects of the aging stroma. Int J Biochem Cell Biol. 2002;34:1401-14
9. Krtolica A, Parrinello S, Lockett S. et al. Senescent fibroblasts promote epithelial cell growth and tumorigenesis: a link between cancer and aging. Proc Natl Acad Sci U S A. 2001;98:12072-7
10. Lasry A, Ben-Neriah Y. Senescence-associated inflammatory responses: aging and cancer perspectives. Trends Immunol. 2015;36:217-28
11. Chen B, Ma X, Liu S. et al. Inhibition of lung cancer cells growth, motility and induction of apoptosis by Klotho, a novel secreted Wnt antagonist, in a dose-dependent manner. Cancer Biol Ther. 2012;13:1221-8
12. Chen B, Wang X, Zhao W. et al. Klotho inhibits growth and promotes apoptosis in human lung cancer cell line A549. J Exp Clin Cancer Res. 2010;29:99
13. Wang X, Chen B, Xu W. et al. Combined effects of klotho and soluble CD40 ligand on A549 lung cancer cells. Oncol Rep. 2011;25:1465-72
14. Wang Y, Chen L, Huang G. et al. Klotho sensitizes human lung cancer cell line to cisplatin via PI3k/Akt pathway. PLoS One. 2013;8:e57391
15. Brighenti E, Calabrese C, Liguori G. et al. Interleukin 6 downregulates p53 expression and activity by stimulating ribosome biogenesis: a new pathway connecting inflammation to cancer. Oncogene. 2014;33:4396-406
16. DePinho RA. The age of cancer. Nature. 2000;408:248-54
17. Rodier F, Campisi J. Four faces of cellular senescence. J Cell Biol. 2011;192:547-56
18. Davalos AR, Coppe JP, Campisi J. et al. Senescent cells as a source of inflammatory factors for tumor progression. Cancer Metastasis Rev. 2010;29:273-83
19. Kelly J, Ali Khan A, Yin J. et al. Senescence regulates macrophage activation and angiogenic fate at sites of tissue injury in mice. J Clin Invest. 2007;117:3421-6
20. Toso A, Revandkar A, Di Mitri D. et al. Enhancing chemotherapy efficacy in Pten-deficient prostate tumors by activating the senescence-associated antitumor immunity. Cell Rep. 2014;9:75-89
21. Xu Y, Sun Z. Molecular basis of Klotho: from gene to function in aging. Endocr Rev. 2015;36:174-93
22. Utsugi T, Ohno T, Ohyama Y. et al. Decreased insulin production and increased insulin sensitivity in the klotho mutant mouse, a novel animal model for human aging. Metabolism. 2000;49:1118-23
23. Tatar M, Bartke A, Antebi A. The endocrine regulation of aging by insulin-like signals. Science. 2003;299:1346-51
24. Liu S, Gupta A, Quarles LD. Emerging role of fibroblast growth factor 23 in a bone-kidney axis regulating systemic phosphate homeostasis and extracellular matrix mineralization. Curr Opin Nephrol Hypertens. 2007;16:329-35
25. Yamamoto M, Clark JD, Pastor JV. et al. Regulation of oxidative stress by the anti-aging hormone klotho. J Biol Chem. 2005;280:38029-34
26. van Deursen JM. The role of senescent cells in ageing. Nature. 2014;509:439-46
27. Bartling B, Demling N, Silber RE. et al. Proliferative stimulus of lung fibroblasts on lung cancer cells is impaired by the receptor for advanced glycation end-products. Am J Respir Cell Mol Biol. 2006;34:83-91
28. Lugo R, Gabasa M, Andriani F. et al. Heterotypic paracrine signaling drives fibroblast senescence and tumor progression of large cell carcinoma of the lung. Oncotarget. 2016;7:82324-37
29. Papadopoulou A, Kletsas D. Human lung fibroblasts prematurely senescent after exposure to ionizing radiation enhance the growth of malignant lung epithelial cells in vitro and in vivo. Int J Oncol. 2011;39:989-99
30. Liu F, Wu S, Ren H. et al. Klotho suppresses RIG-I-mediated senescence-associated inflammation. Nat Cell Biol. 2011;13:254-62
31. Eren M, Boe AE, Murphy SB. et al. PAI-1-regulated extracellular proteolysis governs senescence and survival in Klotho mice. Proc Natl Acad Sci U S A. 2014;111:7090-5
32. Lopez-Andres N, Calvier L, Labat C. et al. Absence of cardiotrophin 1 is associated with decreased age-dependent arterial stiffness and increased longevity in mice. Hypertension. 2013;61:120-9
33. Lee KW, Yeo SY, Sung CO. et al. Twist1 is a key regulator of cancer-associated fibroblasts. Cancer Res. 2015;75:73-85
34. Alfaro C, Sanmamed MF, Rodriguez-Ruiz ME. et al. Interleukin-8 in cancer pathogenesis, treatment and follow-up. Cancer Treat Rev. 2017;60:24-31
35. Guo Y, Xu F, Lu T. et al. Interleukin-6 signaling pathway in targeted therapy for cancer. Cancer Treat Rev. 2012;38:904-10
36. Schafer ZT, Brugge JS. IL-6 involvement in epithelial cancers. J Clin Invest. 2007;117:3660-3
Author contact
![]() Corresponding author: Jianqing Wu; email: jwunyedu.cn; Tel/Fax: +86 25 68135113
Corresponding author: Jianqing Wu; email: jwunyedu.cn; Tel/Fax: +86 25 68135113

 Global reach, higher impact
Global reach, higher impact