3.2
Impact Factor
ISSN: 1837-9664
J Cancer 2018; 9(18):3303-3310. doi:10.7150/jca.25693 This issue Cite
Research Paper
Improved Long-Term Clinical Outcomes And Safety Profile Of Sunitinib Dosing Schedule With 4/2 Switched To 2/1 In Patients With Metastatic Renal Cell Carcinoma
Department of Urology, Institute of Urology, West China Hospital, Sichuan University, Chengdu, China, 610041
*These three authors are co-first authors of the manuscript
Received 2018-2-23; Accepted 2018-6-16; Published 2018-9-7
Abstract
Purpose: This study aimed to identify the survival benefit and safety of alternative dosage schedules for sunitinib in metastatic renal cell carcinoma.
Materials and Methods: Clinicopathologic and survival data of patients treated with sunitinib as first-line therapy were retrospectively reviewed. Patients were classified into three groups: a standard dosing schedule (4/2 schedule), alternative dosing schedule (2/1 schedule), and switched dosing schedule (4/2-2/1 schedule).
Results: Ninety-nine patients were retrospectively included. Seventy-five (75.8%) patients were initially administrated with a 4/2 schedule of sunitinib, while 24 were started with the 2/1 schedule. During treatment, 45 (60.0%) patients with an initial 4/2 schedule switched to a 2/1 schedule (4/2-2/1 schedule) due to severe adverse events (AEs) or poor tolerance. Compared to that with a 4/2 schedule, patients with a 2/1 schedule had a much lower incidence of grade 3/4 AEs (69.6% vs. 40.6%, p=0.001). Overall, the 4/2-2/1 schedule was associated with the best survival benefits. Among the 4/2, 2/1, and 4/2-2/1 schedule groups, the median PFS was 12.5, 11.0, and 25.0 months, respectively (p=0.003), and the median OS was 21.0, 28.0, and 52.0 months, respectively (p=0.03). Multivariate analysis identified the 4/2-2/1 schedule as an independent factor predicting favorable PFS. Although without statistical significance, 4/2-2/1 schedule could decrease 55% risk of death. Furthermore, patients with unfavorable IMDC risk seemed to have more opportunity to achieve better survival from the 4/2-2/1 dosing schedule.
Conclusion: Patients with a 4/2-2/1 schedule could minimize treatment-related toxicities; more importantly, patients with 4/2-2/1 schedule could achieve a superior survival benefit.
Keywords: clinical outcome, dosing schedule, metastatic renal cell carcinoma, sunitinib, safety profile
Introduction
Metastatic renal cell carcinoma (mRCC) accounts for 20-30% of all kidney cancer at the initial diagnosis. Additionally, 20-40% of localized renal cell carcinoma will develop distant metastases after radical surgery [1]. Sunitinib has been recommended as a standard first-line treatment for mRCC. According to pre-clinical and clinical studies, the suggested standard dosing schedule of sunitinib is a dose of 50 mg/day taken orally (4 weeks on and 2 weeks off, i.e., the '4/2 schedule'), and this schedule has a superior survival benefit [2-4]. Evidence has demonstrated that high exposure of sunitinib was associated with improved response, time to progression, and overall survival (OS) [5]. However, long-term and high exposure was concomitant with severe adverse events (AEs). Data on sunitinib, either from phase III trials, expanded-access trials, or even in the adjuvant setting, showed that, although patients could achieve better survival benefit, more severe AEs would occur and impact compliance and medication taken [3-4,6-8]. In Asian studies, compared to that in western countries, survival was greatly improved; however, the incidence of toxicities tended to be much higher, resulting in more frequent schedule modifications, dosage reduction, interruption, and even discontinuation [9-14].
To balance efficacy and AEs better, alternative schedules of sunitinib have been evaluated to improve patients' tolerability and survival outcomes. In a phase 2 trial conducted by Motzer in 2012, a comparison to the standard 4/2 schedule with 37.5 mg continuous daily dosage of sunitinib was performed; unfortunately, the results failed to show superior drug tolerance and clinical benefit of a continuous dosing schedule [15]. Subsequently, retrospective studies from different medical centers compared individualized dosing schedule modifications according to toxic severity with a standard 4/2 schedule; the results summarized the non-inferior or superior clinical benefit of a 2/1 dosing schedule [16-24]. Recently, a prospective phase 2 trial compared an initial 2/1 schedule with a standard 4/2 schedule, and confirmed the superiority of an alternative schedule of sunitinib [25].
However, in the real world, some patients would be originally treated with an alternative 2/1 schedule, more patients were switched to the 2/1 schedule due to severe toxicities occurred after the initial cycles of standard 4/2 schedule. So far, studies comparing a 4/2 schedule switched to a 2/1 schedule indicated that the 2/1 schedule with standard 4/2 schedules were rare and from the western population [17,20]. In this retrospective study, we compared three sunitinib dosing schedules in Asian patients with respective to tumor response, survival outcomes and safety, and analyzed potential risk factors guiding physician's decision-making for the optimal dosing schedule in the treatment of mRCC as first-line therapy.
Materials and Methods
Patients and sunitinib dosing schedules
We retrospectively reviewed the medical records of patients treated with sunitinib as first-line antiangiogenic therapy between 2008 and 2015 at West China Hospital, Sichuan University. Eligibility criteria were patients with the following characteristics: ≥18 years of age, and pathological evidence of RCC. Patients with no distant metastasis were excluded. Clinicopathologic data, survival outcomes, and AEs were collected. Patients were seen by physicians for regular evaluations (physical examination, radiologic assessment and laboratory tests) to assess AEs and efficacy every 4-6 weeks. Most patients were initially administered with a standard dosing schedule of sunitinib (50 mg per day, 4 weeks on and 2 weeks off, i.e., the '4/2 schedule'); the remaining patients were placed on an alternative dosing schedule (50 mg per day, 2 weeks on and 1 week off, i.e., the '2/1 schedule'), mainly due to poor performance status or the physicians' experience. During treatment, patients who initially began with a 4/2 schedule changed their dosing schedule to a 2/1 schedule (4/2-2/1 schedule) due to severe AEs and/or worsening intolerance.
The primary endpoints were progression-free survival (PFS) and OS, and the second endpoints were tumor response and AEs. PFS was defined as the time from first administration of sunitinib to progression of disease (PD) or death. OS was defined as the time from first administration of sunitinib to all-cause death. AEs causing sunitinib schedule modification were recorded and the interval between the first and second schedule was measured. The Response Evaluation Criteria in Solid Tumors (RECIST) 1.1 [26] was applied to assess the treatment efficacy in patients. Severe AEs were defined as those of grade ≥3 as classified by the Common Terminology Criteria for Adverse Events (CTCAE), version 4.0 [27].
Statistical Analysis
We used the one-sample Kolmogorov-Smirnov test to evaluate normal distribution. Data were presented as categorical variables and continuous variables; chi-square (χ2) and Kruskal-Wallis tests were used to evaluate statistical differences, respectively. Kaplan-Meier survival curves were plotted and the log-rank test was used to analyze statistical significance. The GraphPad Prism 6.02 and SPSS version 16.0 (SPSS Inc., Chicago, IL, USA) software were used for all statistical analyses. A p<0.05 was considered statistically significant.
Results
Patient characteristics
A total of 99 patients with metastatic RCC treated with sunitinib were included in this retrospective study. The median follow-up time was 37 months. At the beginning, 75 (75.8%) patients were on a 4/2 schedule of sunitinib, while the remaining 24 were placed on a 2/1 schedule (mainly due to poor performance status or the physicians' experience). During treatment, 45 patients who initially began with a 4/2 schedule changed their dosing schedule to a 2/1 schedule (4/2-2/1 schedule) due to severe AEs and/or worsening intolerance. Only 30 were maintained with a 4/2 schedule throughout therapy. The median time from the initial 4/2 schedule switched to a 2/1 schedule was about 5 months (3.6 cycles). Table 1 summarized and compared the baseline characteristics of the three groups. Over two-third of the patients were men; most patients (>80%) underwent nephrectomy before systemic therapy, and over 60% of metastatic lesion sites were within a single organ. Although there were no statistical differences, more patients (41.7%) belonging to the 2/1 group had poor performance status, and patients in the 4/2-2/1 group (40.0%) had more opportunity for sequential therapy than the other two groups.
Baseline characteristics of patients treated with sunitinib stratified by different dosage schedules
| Characteristics | Schedule 4/2-2/1 (N=45) | Schedule 2/1 (N=24) | Schedule 4/2 (N=30) | P value |
|---|---|---|---|---|
| Age | 0.108 | |||
| Median(range) | 59.0(20.0-81.0) | 59.5(42-77) | 53.5(27.0-72.0) | |
| Gender, no. (%) | 0.524 | |||
| Female | 14(31.1%) | 9(37.5%) | 7(23.3%) | |
| Male | 31(68.9%) | 15(62.5%) | 23(76.7%) | |
| Histological type, no. (%) | 0.272 | |||
| Clear cell | 37(82.2%) | 15(62.5%) | 22(73.3%) | |
| Non-clear cell | 8(17.8%) | 9(37.5%) | 8(26.7%) | |
| Nephrectomy, no. (%) | 0.538 | |||
| Y | 40(87.0%) | 19 (79.2%) | 26(86.7%) | |
| N | 5(13.0%) | 5(20.8%) | 4(13.3%) | |
| ECOG score, no. (%) | 0.096 | |||
| 0-1 | 35(77.8%) | 14(58.3%) | 23(76.7%) | |
| ≥2 | 10(22.2%) | 10(41.7%) | 7(23.3%) | |
| IMDC, no. (%) | 0.571 | |||
| Low risk | 9(20.0%) | 4(16.7%) | 10(33.3%) | |
| Intermediate risk | 21(46.7%) | 13(54.2%) | 13(43.3%) | |
| High risk | 15(33.3%) | 7(29. 2%) | 7(23.3%) | |
| Interval from diagnosis to metastasis, no. (%) | 0.032 | |||
| Metachronous | 14(31.1%) | 14(58.3%) | 17(56.7%) | |
| Synchronous | 31(68.9%) | 10(41.7%) | 13(43.3%) | |
| Metastatic site, no. (%) | ||||
| Lung | 26(57.8%) | 13(54.2%) | 20(66.7%) | 0.557 |
| Bone | 13(28.9%) | 6(25.0%) | 6(20.0%) | 0.686 |
| Liver | 0(0%) | 1(4.2%) | 1(20.0%) | 0.417 |
| Brain | 2(4.4%) | 0(0%) | 1(3.3%) | 0.587 |
| Lymph node | 5(11.1%) | 4(16.7%) | 3(10.0%) | 0.728 |
| Other | 16(35.6%) | 10(41.7%) | 11(36.7%) | 0.879 |
| Metastatic organs, no. (%) | 0.71 | |||
| Single | 31(68.9%) | 15(62.5%) | 18(60.0%) | |
| Multiple(≥2 sites) | 14(31.1%) | 9(37.5%) | 12(40.0%) | |
| Sequential therapies after sunitinib failure, no. (%) | 0.06 | |||
| Without | 27(60.0%) | 20(83.3%) | 24(80.0%) | |
| With | 18(40.0%) | 4(16.7%) | 6(20.0%) | |
Abbreviations: IMDC, International Metastatic renal cell carcinoma Database Consortium; ECOG, Eastern Cooperative Oncology Group.
Treatment efficacy
As shown in Table 2, we analyzed the best tumor response of the three groups. Complete response (CR) was achieved in two patients, exclusively from the 4/2-2/1 schedule group. A total of 27 patients achieved partial response (PR); 13 were in the 4/2-2/1 group, 10 in the 4/2 group, and 4 in the 2/1 group. Twenty-two patients showed PD, 8 were in the 4/2-2/1 group, and 7 each in the 2/1 and 4/2 groups.
The best therapeutic tumor response among three dosage schedules
| Schedule | Tumor Response (%) | ORR# (%) | DCR# (%) | |||
|---|---|---|---|---|---|---|
| CR | PR | SD | PD | |||
| 4/2-2/1 | 2(4.4) | 13(28.9) | 22(48.9) | 8(17.8) | 15(33.3) | 37 (82.2) |
| 2/1 | 0(0) | 4(16.7) | 13(51.2) | 7(29.2) | 4(16.7) | 17(70.8) |
| 4/2 | 0(0) | 10(33.3) | 13(43.3) | 7(23.3) | 10(33.3) | 23(76.7) |
| Total | 2(2.0) | 27(27.3) | 48(48.5) | 22(22.2) | 29(29.3) | 77 (77.8) |
Abbreviations: CR, complete response; PR, partial response; SD, stable disease; PD, progression of disease; ORR, objective response rate; DCR, disease control rate; #, p value>0.05.
Statistical analysis by chi-square test suggested that PR was significantly different among three groups (p=0.047), while no statistical differences were observed in CR and SD. Overall, the objective response rate (ORR) was achieved in 29.3% (29/99), and the disease control rate (DCR) reached to 77.8% (77/99). Although the ORR was relatively lower in the 2/1 schedule group (4/24, 16.7%), a statistical difference was not observed (p=0.383).
Survival outcomes
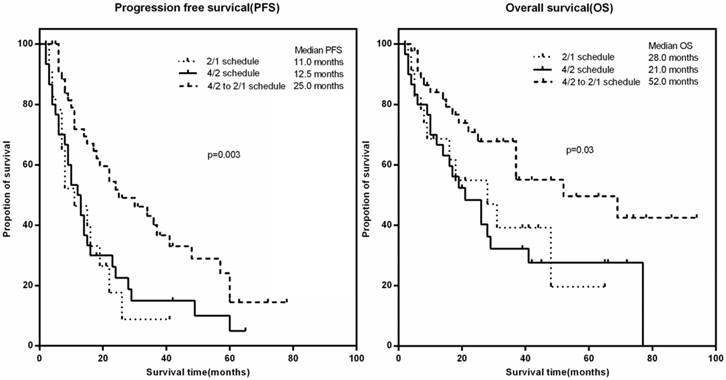
At the cut-off point, 74.4% (74/99) patients had disease progression or death with the median PFS of 15.0 months, while 51 patients had died at the time of the last follow-up. The median OS of the whole cohort reached 31.0 months. We sub-analyzed and compared PFS and OS among patients with different dosing schedules. In patients with a 4/2-2/1 schedule, the median PFS was much longer than that in the 2/1 and 4/2 groups (25.0 vs. 11.0 vs. 12.5 months, respectively; p=0.003) (Figure 1A). A similar result was observed in OS (Figure 1B); patients in the 4/2-2/1 group had longer OS compared with the other two groups (median OS: 53.0 vs. 28.0 vs. 21 months, p=0.03).
We then analyzed potential risk factors for PFS and OS in patients with mRCC treated with sunitinib (Table 3). Univariate analysis indicated that IMDC ≥3, ≥2 sites of metastatic organs, nephrectomy, and a 4/2-2/1 schedule were associated with PFS. Further multivariate analysis suggested that IMDC ≥3 and a 4/2-2/1 schedule were independent prognostic factors of PFS. A 4/2-2/1 schedule was strongly associated with a decreased risk of disease progression (hazard ratio [HR] 0.38, 95% confidence interval [Cl] 0.23-0.63, p=0.001), while IMDC ≥3 was associated with an increased risk of disease progression (HR 1.74, 95% Cl 1.03-2.95, p=0.004). In terms of OS, ECOG ≥2, IMDC ≥3, nephrectomy, and a 4/2-2/1 schedule were statistically associated with OS in univariate analysis. Cox's regression model demonstrated that nephrectomy before TKI was independent factor predicting a favorable prognosis (HR 0.13, 95% Cl 0.033-0.461, p=0.002), while IMDC ≥3 was still associated with an increased risk of death (HR 6.14, 95% Cl 1.31-28.87, p=0.022). In addition, although there was no statistical significance, a 4/2-2/1 schedule could definitely decrease the risk of death (4/2-2/1 schedule: HR 0.45, 95% Cl 0.11-1.84, p=0.268).
Subsequently, the association of clinical/pathological parameters with survival outcomes among three dosing schedules were further analyzed (Table 4). Compared to patients with favorable IMDC risk, patients with intermediate/poor IMDC risk could have much better survival benefit from a 4/2-2/1 schedule than the other two dosing schedules. Lung/lymph node metastasis and multiple sites (≥2) in metastatic organs were two parameters that could predominantly delay DP in patients with a 4/2-2/1 schedule. The median PFS of patients with lung/lymph node metastasis treated with a 4/2-2/1, 2/1, and 4/2 schedule were 22.0, 8.0, and 13.0 months, respectively (p=0.019). The median PFS of patients with multiple metastatic sites were 29.5, 7.0, and 11.0 months, respectively (p=0.002). However, in a subgroup of patients with lung/lymph node metastasis or multiple metastatic sites, OS differences among three dosing schedules were not significantly different. Patients stratified by prior nephrectomy, ECOG status, and sex showed no different survival outcomes among three dosing schedules.
Survival outcomes among patients with different dosing schedules. A). progression-free survival (PFS), B) overall survival (OS)
Univariate and multivariate analyses for PFS and OS
| Factors | PFS | OS | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Univariate | Multivariate | Univariate | Multivariate | ||||||||
| HR(95%CI) | P | HR(95%CI) | P | HR(95%CI) | P | HR(95%CI) | P | ||||
| Age>50 | 0.85 (0.52-1.42) | 0.541 | - | - | 0.76 (0.42-1.39) | 0.375 | - | - | |||
| Male | 1.03 (0.63-1.67) | 0.920 | - | - | 1.32 (0.72-2.42) | 0.372 | - | - | |||
| ECOG≥2 | 2.06 (0.94-4.52) | 0.072 | - | - | 3.31 (1.04-10.57) | 0.043 | 1.57 (0.38-6.47) | 0.535 | |||
| IMDC≥3 | 1.66 (1.02-2.72) | 0.042 | 1.74 (1.03-2.95) | 0.040 | 2.52 (1.43-4.41) | 0.001 | 6.14 (1.31-28.87) | 0.022 | |||
| Clear cell | 0.99 (0.59-1.67) | 0.971 | - | - | 0.88 (0.47-1.62) | 0.674 | - | - | |||
| Metachronous | 0.93 (0.59-1.47) | 0.753 | - | - | 0.85 (0.49-1.49) | 0.572 | - | - | |||
| Lung and LN | 0.95 (0.47-1.90) | 0.874 | 0.87 (0.47-1.63) | 0.667 | 0.92 (0.51-1.64) | 0.765 | - | - | |||
| ≥2 sites | 1.62 (1.02-2.57) | 0.042 | 1.58 (0.85-2.92) | 0.149 | 1.58 (0.91-2.73) | 0.106 | - | - | |||
| Nephrectomy | 2.18 (1.14-4.19) | 0.019 | 1.81 (0.90-3.65) | 0.096 | 3.23 (1.64-6.39) | 0.001 | 8.56 (2.15-30.05) | 0.002 | |||
| schedule 4/2-2/1 | 0.46 (0.29-0.74) | 0.001 | 0.38 (0.23-0.63) | 0.001 | 0.47 (0.26-0.84) | 0.011 | 0.45 (0.11-1.84) | 0.268 | |||
Abbreviations: IMDC, International Metastatic renal cell carcinoma Database Consortium; LN, Lymph Node; ECOG, Eastern Cooperative Oncology Group.
Subgroup analysis of survival outcomes among three dosage schedules
| Factors | Progression free survival | P value | Overall survival | P value | ||||
|---|---|---|---|---|---|---|---|---|
| 4/2-2/1 | 2/1 | 4/2 | 4/2-2/1 | 2/1 | 4/2 | |||
| IMDC | ||||||||
| Favorable | 25.0(6.0-60.0) | 13.5(4.0-21.0) | 15.5(4.0-65.0) | 0.62 | 52.0(6.0-94.0) | 29.0(5.0-41.0) | 26.0(10.0-72.0) | 0.208 |
| Intermediate | 30.0(5.0-78.0) | 15.0(2.0-41.0) | 12.0(2.0-49.0) | 0.015 | NR | 29.5(2.0-41.0) | 41.0(2.0-77.0) | 0.047 |
| Poor | 14.0(4.0-63.0) | 8.0(3.0-26.0) | 6.0(3.0-14.0) | 0.004 | 37.0(4.0-69.0) | 18.0(3.0-65.0) | 9.0(3.0-28.0) | 0.03 |
| Histological patterns | ||||||||
| ccRCC | 25.0(4.0-78.0) | 16.0(2.0-26.0) | 11.0(3.0-65.0) | 0.006 | 52.0(4.0-86.0) | 48.0(2.0-65.0) | 21.0(3.0-77.0) | 0.186 |
| Non-ccRCC | 41.0(6.0-72.0) | 8.0(3.0-41.0) | 14.0(3.0-60.0) | 0.283 | NR | 18.0(4.0-41.0) | 22.0(2.0-72.0) | 0.151 |
| Interval from diagnosis to metastasis | ||||||||
| Metachronous | 24.0(6.0-78.0) | 26.0(2.0-22.6) | 13.0(2.0-65.0) | 0.26 | NR | 48.0(2.0-65.0) | 19.0(3.0-77.0) | 0.051 |
| Synchronous | 25.0(4.0-72.0) | 7.5(4.0-41.0) | 10.0(2.0-29.0) | 0.001 | 37.0(4.0-94.0) | 9.0(4.0-44.0) | 26.0(2.0-66.0) | 0.122 |
| Metastatic sites | ||||||||
| Lung/LN | 22.0(4.0-78.0) | 8.0(3.0-26.0) | 13.0(2.0-65.0) | 0.019 | 69.0(4.0-94.0) | 16.0(4.0-48.0) | 26.0(2.0-77.0) | 0.054 |
| Other sites | 30.0(5.0-63.0) | 16.0(2.0-41.0) | 9.5(3.0-60.0) | 0.095 | 37.0(5.0-74.0) | NR | 18.0(3.0-72.0) | 0.092 |
| Metastatic organs | ||||||||
| ≤1 sites | 25.0(4.0-78.0) | 15.0(2.0-41.0) | 14.0(3.0-65.0) | 0.155 | NR | 28.0(3.0-65.0) | 21.0(3.0-77.0) | 0.139 |
| ≥2 sites | 29.5(7.0-60.0) | 7.0(3.0-26.0) | 11.0(2.0-29.0) | 0.002 | 52.0(7.0-94.0) | 16.0(4.0-48.0) | 19.0(2.0-66.0) | 0.116 |
| Nephrectomy | ||||||||
| Y | 25.0(4.0-78.0) | 26.0(3.0-41.0) | 13.0(3.0-65.0) | 0.035 | 69.0(4.0-94.0) | 31.0(2.0-65.0) | 26.0(3.0-77.0) | 0.076 |
| N | 29.5(5.0-37.0) | 4.5(4.0-7.0) | 4.0(2.0-23.0) | 0.034 | 29.5(5.0-37.0) | 6.0(4.0-9.0) | 9.5(2.0-41.0) | 0.064 |
| ECOG score | ||||||||
| 0-1 | 30.0(5.0-78.0) | 11.0(2.0-11.0) | 23.0(9.0-42.0) | 0.155 | NR | NR | NR | 0.992 |
| ≥2 | 11.0(8.0-18.0) | 4.0(3.0-26.0) | 28.0(5.0-49.0) | 0.229 | NR | 5.0(4.0-48.0) | 45.0(5.0-77.0) | 0.282 |
| Gender | ||||||||
| M | 24.0(4.0-63.0) | 9.5(2.0-26.0) | 12.0(2.0-65.0) | 0.046 | 37.0(5.0-94.0) | 18.0(2.0-48.0) | 21.0(2.0-77.0) | 0.109 |
| F | 25.0(7.0-78.0) | 15.0(4.0-41.0) | 13.0(3.0-28.0) | 0.021 | NR | NR | 26.0(3.0-45.0) | 0.087 |
Notes: Data are presented as median (range).
Abbreviations: IMDC, International Metastatic renal cell carcinoma Database Consortium; ccRCC, clear cell Renal Cell Carcinoma; LN, Lymph Node; ECOG, Eastern Cooperative Oncology Group.
Treatment related adverse events among three dosage schedules
| Adverse event | 4/2-2/1(pre-switch) (n=45) | 4/2-2/1(post-switch) (n=45) | 2/1 (n=24) | 4/2 (n=30) | P* value | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| All grade | Grade 3/4 | All grade | Grade 3/4 | All grade | Grade 3/4 | All grade | Grade 3/4 | |||||
| All | 44(97.8%) | 33(73.3%) | 44(97.8%) | 17(37.8%) | 23(95.8%) | 11(45.8%) | 28(93.3%) | 19(63.3%) | 0.001 | |||
| Clinical observation | ||||||||||||
| Fatigue | 27(60.0%) | 13(28.8%) | 17(37.8%) | 6(13.3%) | 10(41.6%) | 3(12.5%) | 17(56.7%) | 7(23.3%) | 0.071 | |||
| Hypertension | 14(31.1%) | 8(17.8%) | 10(22.2%) | 2(4.4%) | 4(16.7%) | 2(8.3%) | 12(40.0%) | 4(13.3%) | 0.044 | |||
| Hand-foot syndrome | 25(55.6%) | 12(26.7%) | 15(33.3%) | 4(8.9%) | 10(41.6%) | 5(20.8%) | 15(33.3%) | 3(10.0%) | 0.027 | |||
| Diarrhea | 10(22.2%) | 5(11.1%) | 6(13.3%) | 0 | 7(29.2%) | 1(4.2%) | 11(36.7%) | 4(13.3%) | 0.021 | |||
| Mucositis (oral) | 16(35.6%) | 5(11.1%) | 6(13.3%) | 0 | 7(29.2%) | 1(4.2%) | 11(36.7%) | 4(13.3%) | 0.021 | |||
| Skin color change | 8(17.7%) | 0 | 7(15.6%) | 0 | 4(16.7%) | 0 | 6(20.0%) | 1(3.3%) | - | |||
| Anorexia | 16(35.6%) | 2(4.4%) | 6(13.3%) | 0 | 5(20.8%) | 0 | 12(40.0%) | 1(3.3%) | 0.153 | |||
| Abdominal pain | 2(4.4%) | 0 | 2(4.4%) | 0 | 0 | 0 | 2(6.7%) | 0 | - | |||
| Epistaxis | 4(8.9%) | 0 | 1(2.2%) | 0 | 0 | 0 | 3(10.0%) | 0 | - | |||
| Pain in extremities | 0 | 0 | 0 | 0 | 1(4.2%) | 0 | 0 | 0 | - | |||
| Heart failure | 1(2.2%) | 1(2.2%) | 1(2.2%) | 1(2.2%) | 0 | 0 | 0 | 0 | - | |||
| Laboratory tests | ||||||||||||
| Renal dysfunction | 15(33.3%) | 9(20.0%) | 15(33.3%) | 6(13.3%) | 9(37.5%) | 2(8.3%) | 6(20.0%) | 1(3.3%) | 0.396 | |||
| Thrombocytopenia | 23(51.1%) | 7(15.6%) | 22(48.9%) | 4(8.9%) | 8(33.3%) | 2(8.3%) | 16(53.3%) | 2(6.7%) | 0.334 | |||
| Leukopenia | 28(62.2%) | 14(31.1%) | 23(51.1%) | 4(8.9%) | 11(45.8%) | 2(8.3%) | 17(56.7%) | 9(30.0%) | 0.033 | |||
| Anemia | 18(40.0%) | 5(11.1%) | 10(22.2%) | 3(6.7%) | 6 (25.0%) | 1(4.2%) | 16(53.3%) | 2(6.7%) | 0.459 | |||
| Liver dysfunction | 12(26.6%) | 3(6.7%) | 8(17.8%) | 2(4.4%) | 8(33.3%) | 2(8.3%) | 8(26.7%) | 1(3.3%) | 0.654 | |||
| Neutropenia | 28(62.2%) | 14(31.1%) | 25(55.6%) | 4(8.9%) | 10(41.7%) | 2(8.3%) | 19(63.3%) | 7(23.3%) | 0.033 | |||
| Lymphopenia | 23(51.1%) | 8(17.8%) | 22(48.9%) | 7(15.6%) | 11(45.8%) | 4(16.7%) | 15(50.0%) | 6(20.0%) | 0.777 | |||
| Hypophosphatemia | 16(35.6%) | 0 | 16(35.6%) | 0 | 6(25.0%) | 0 | 10(33.3%) | 0 | - | |||
| Hypocalcemia | 19(42.2%) | 0 | 14(31.1%) | 0 | 9(37.5%) | 2(8.3%) | 13(43.3%) | 1(3.3%) | - | |||
| Hypothyroidism | 18(40.0%) | 3(6.7%) | 10(22.2%) | 1(2.2%) | 9(37.5%) | 0 | 17(56.7%) | 2(6.7%) | 0.306 | |||
| Hyponatremia | 9(20.0%) | 0 | 9(20.0%) | 0 | 8(33.3%) | 0 | 7(23.3%) | 0 | - | |||
Notes: All statistical analyses were compared between 4/2-2/1 schedule pre-switched and post-switched periods
Treatment-related toxicities associated with sunitinib
AEs are summarized in Table 5. Overall, the incidence of toxicities of all grades was 95.9%; the incidence of grade 3/4 AEs reached 63.6%. For patients receiving a standard dosing schedule (continuous 4/2 schedule plus 4/2-2/1 schedule before the switch period, n=75), the incidence of grade 3/4 AEs was as high as 69.6%, while for patients receiving an alternative dosing schedule (initial 2/1 schedule plus 4/2-2/1 schedule after switch period, n=69), the incidence of severe AEs was only 40.6%. The difference was statistically significant (χ2=12.03, p=0.001). According to each dosing schedule, the incidence of all grade AEs for 4/2-2/1, 2/1, and 4/2 schedules were 97.8%, 95.8%, and 93.3%, respectively. For the initial 2/1 schedule and maintained 4/2 schedule, the incidence of grade 3/4 AEs was 45.8% and 63.3%, respectively. It is noteworthy that for the 4/2-2/1 schedule, grade 3/4 AEs decreased from 73.3% to 37.8% between the pre-switched and post-switched period, and the difference between these two periods was statistically significant (χ2=11.520, p=0.001). Although grade 3/4 AEs were most common in the 4/2-2/1 schedule at the pre-switched period, it reduced to the least incidence among the three schedules at the post-switched period. The most common grade 3/4 AEs resulting in a 4/2-2/1 schedule before dosage switch were neutropenia and leukopenia (31.1%), fatigue (28.8%), hand-foot syndrome (20.0%), thrombocytopenia (15.6%), hypertension (15.6%), mucositis (11.1%), and diarrhea (11.1%). After dosage adjustment within this group, the following common grade 3/4 AEs dramatically and statistically reduced: neutropenia and leukopenia (8.9%), fatigue (13.3%), hand-foot syndrome (8.9%), hypertension (4.4%), and the mucositis and diarrhea were not present. Compared to those in the 2/1 schedule, dose reduction and interruption rates were higher in the 4/2 and 4/2-2/1 schedules.
Discussion
In the current study, we firstly analyzed three sunitinib dosing schedules—4/2-2/1, 2/1 and 4/2 schedules—in Asian patients with mRCC with respect to clinical outcomes and drug toxicities. Among these three dosing schedules, patients with a 4/2-2/1 schedule seemed to achieve the best survival benefits and experienced an improved safety profile.
Clinical data showed that patients treated with sunitinib would frequently suffer from kinds of drug-related toxicities [3-4, 6-7]. These AEs could result in a 49-50% of dose reduction or interruption; 19-24% of patients discontinued therapy due to severe AEs. It is noteworthy that, among patients of Asian ethnicity, the incidence of AEs, especially myelosuppression, appeared to be much higher than that in other races [10-12]. Pharmacokinetic analysis revealed a relatively higher bioavailability in the Asian population, which could at least partially explain why higher AE incidence and better survival benefits were seen in Asian patients [28].
In the present study, the safety profile was similar to that in previous studies. An alternating 2/1 schedule, including the initial 2/1 and 4/2-2/1 schedules, could undoubtedly minimize treatment-related toxicities. Although the total incidence of all grades of AEs between the alternative 2/1 schedule and standard 4/2 schedule was not significantly different, the incidence of grade ¾ AEs in patients on a 2/1 schedule was considerably lower than in those on a standard regimen. It is worth noting that the incidence of severe AEs in patients with an initial 2/1 schedule appeared to be slightly higher than that in those with a 4/2-2/1 schedule after schedule modification; this suggested that AEs, especially severe AEs, tend to occur in the early cycles of TKI therapy, and patients showed probable drug tolerance and adaption to TKI treatment.
Currently, different alternative dosing schedules for sunitinib have been investigated in clinical practice. Although there is a lack of validation with well-designed prospective randomized trials, the 2/1 schedule seemed to have the highest probability of replacing the current standard 4/2 schedule. A phase I study has showed that the 2/1 schedule provided prolonged drug exposure compared with that of the 4/2 schedule, which suggested the reliable survival benefits from the 2/1 schedule [29].
In the present study, some patients were initially treated with the 2/1 schedule, while some were finally placed on a 2/1 schedule after a period of being on the 4/2 schedule. We suspect there is difference in efficacy between the two types of 2/1 schedules. The MD Anderson cancer center and the RAINBOW group have compared the initial 2/1 schedule and 4/2 to 2/1 switch (4/2-2/1) schedule with standard 4/2 schedule in the Western population [17,20]. Paradoxically and interestingly, two studies from the Asian population showed evidence to support the initial 2/1 schedule as the optimal regimen in the treatment of mRCC [23,25]. However, these two studies lacked long-term outcomes of OS, included a small sample size, and/or focused on the safety profile or health-related quality of life.
It was the first time to compare these three different dosing schedules in the Asian population with long-term outcomes. Undoubtedly, based on the results from ours and the two previous retrospective studies, the 4/2-2/1 schedule seemed to have the highest efficacy among the three dosing schedules, while the superiority of the initial 2/1 schedule compared to the standard 4/2 schedule is undetermined [17,20]. Although we also had a small sample size, we compared long-term clinical outcomes among the three schedules and attempted to find potential factors that may affect clinical decision making on a suitable sunitinib dosing schedule. In addition, patients in our study had relatively poor ECOG status and the ratio of poor IMDC risk was relatively high. Furthermore, we analyzed more comprehensive AE data than the previous Asian reports.
Providing reasonable explanations for these inconsistent conclusions was worthy of thorough consideration. Thus, we acknowledge the limitations of our study. First, our results were limited by the retrospective study design itself. Patient selection and decision-making for dosing schedule were unavoidable biases. Second, the collection and evaluation of AEs could also be biased, due to limited medical records and recall or reporting bias from physicians and patients. Third, patient compliance could not be precisely assessed during treatment. In addition, the development of AEs could help identify patients with inherent pharmacokinetic and pharmacodynamics characteristics that predispose to clinical benefit, which could at least partially explain why the patients with a 4/2-2/1 schedule had much longer PFS and OS [30-32]. Herein, ongoing prospective trials are currently collecting more data, which will ultimately help in eliminating this bias (NCT02060370, NCT01499121).
Overall, the 4/2-2/1 schedule was an independent factor predicting favorable prognosis in patients treated with sunitinib as first-line therapy. Interestingly and surprisingly, patients with unfavorable IMDC risk (intermediate/poor) or higher tumor burden (multiple sites of metastatic organs) seemed to achieve much better survival benefit from the 4/2-2/1 schedule. Ultimately, the risk factors mentioned above together with severe AEs occurred during treatment might help clinicians to find optimal candidates for the 4/2-2/1 dosing schedule. Again, whether these risk factors together with AEs could be considered as criteria for the physician's decision to place a patient on the 4/2-2/1 schedule, needs to be validated with a large-scale prospective clinical trial.
Totally, compared to other two schedules, the safety profile and clinical outcomes of patients with mRCC treated with sunitinib were improved significantly with the 4/2-2/1 schedule. Patients with unfavorable IMDC risk or higher tumor burden seemed to be candidates who could get better survival benefit from switched dosing schedules of sunitinib. Further prospective studies are required to verify the current conclusion; however, our results provide evidence that this alternative dosing schedule may offer some insights into the improvement of clinical practice, particularly regarding personalized patient management and achieving individualized therapy.
Abbreviations
AEs: Adverse events; RCC: renal cell carcinoma; mRCC: Metastatic RCC; OS: overall survival; IMDC: International Metastatic Renal-Cell Carcinoma Database Consortium; PFS: progression-free survival; RECIST: the Response Evaluation Criteria in Solid Tumors; PD: progression of disease; CR: Complete response; PR: partial response; ORR: objective response rate; DCR: disease control rate; CTCAE: the Common Terminology Criteria for Adverse Events.
Acknowledgements
This work was supported by Natural Science Foundation of China (NSFC 81402110 and 81672547), Science and Technology Support Program of Sichuan Province (2015SZ0230-3) and 1.3.5 project for disciplines of excellence, West China Hospital, Sichuan University (No.0040205301E21).
Competing Interests
The authors have declared that no competing interest exists.
References
1. Janzen NK, Kim HL, Figlin RA. et al. Surveillance after radical or partial nephrectomy for localized renal cell carcinoma and management of recurrent disease. Urol Clin North Am. 2003;30:843-852
2. Faivre S, Delbaldo C, Vera K. et al. Safety, pharmacokinetic, and antitumor activity of SU11248, a novel oral multitarget tyrosine kinase inhibitor, in patients with cancer. J Clin Oncol. 2006;24:25-35
3. Motzer RJ, Hutson TE, Tomczak P. et al. Sunitinib versus interferon alfa in metastatic renal-cell carcinoma. N Engl J Med. 2007;356:115-124
4. Motzer RJ, Hutson TE, Tomczak P. et al. Overall survival and updated results for sunitinib compared with interferon alfa in patients with metastatic renal cell carcinoma. J Clin Oncol. 2009;27:3584-3590
5. Houk BE, Bello CL, Poland B. et al. Relationship between exposure to sunitinib and efficacy and tolerability endpoints in patients with cancer: results of a pharmacokinetic/pharmacodynamic meta-analysis. Cancer Chemother Pharmacol. 2010;66:357-371
6. Motzer RJ, Hutson TE, Cella D. et al. Pazopanib versus sunitinib in metastatic renal-cell carcinoma. N Engl J Med. 2013;369:722-731
7. Gore ME, Szczylik C, Porta C. et al. Final results from the large sunitinib global expanded-access trial in metastatic renal cell carcinoma. Br J Cancer. 2015;113:12-19
8. Ravaud A, Motzer RJ, Pandha HS. et al. Adjuvant Sunitinib in High-Risk Renal-Cell Carcinoma after Nephrectomy. N Engl J Med. 2016;375:2246-2254
9. Tomita Y, Shinohara N, Yuasa T. et al. Overall survival and updated results from a phase II study of sunitinib in Japanese patients with metastatic renal cell carcinoma. Jpn J Clin Oncol. 2010;40:1166-1172
10. Yoo C, Kim JE, Lee JL. et al. The efficacy and safety of sunitinib in korean patients with advanced renal cell carcinoma: high incidence of toxicity leads to frequent dose reduction. Jpn J Clin Oncol. 2010;40:980-985
11. Kim HS, Hong MH, Kim K. et al. Sunitinib for Asian patients with advanced renal cell carcinoma: a comparable efficacy with different toxicity profiles. Oncology. 2011;80:395-405
12. Li XS, Wu X, Zhao PJ. et al. Efficacy and safety of sunitinib in the treatment of metastatic renal cell carcinoma. Chin Med J (Engl). 2011;124:2920-2924
13. Motzer RJ, Hutson TE, Tomczak P. et al. Sunitinib versus interferon alfa in metastatic renal-cell carcinoma. N Engl J Med. 2007;356:115-124
14. Motzer RJ, Hutson TE, Tomczak P. et al. Overall survival and updated results for sunitinib compared with interferon alfa in patients with metastatic renal cell carcinoma. J Clin Oncol. 2009;27:3584-3590
15. Motzer RJ, Hutson TE, Olsen MR. et al. Randomized phase II trial of sunitinib on an intermittent versus continuous dosing schedule as first-line therapy for advanced renal cell carcinoma. J Clin Oncol. 2012;30:1371-1377
16. Najjar YG, Mittal K, Elson P. et al. A 2 weeks on and 1 week off schedule of sunitinib is associated with decreased toxicity in metastatic renal cell carcinoma. Eur J Cancer. 2014;50:1084-1089
17. Atkinson BJ, Kalra S, Wang X. et al. Clinical outcomes for patients with metastatic renal cell carcinoma treated with alternative sunitinib schedules. J Urol. 2014;191:611-618
18. Kondo T, Takagi T, Kobayashi H. et al. Superior tolerability of altered dosing schedule of sunitinib with 2-weeks-on and 1-week-off in patients with metastatic renal cell carcinoma-comparison to standard dosing schedule of 4-weeks-on and 2-weeks-off. Jpn J Clin Oncol. 2014;44:270-277
19. Ohzeki T, Fukasawa S, Komaru A. et al. Efficacy of traditional and alternative sunitinib treatment schedules in Japanese patients with metastatic renal cell carcinoma. Int J Urol. 2014;21:1065-1068
20. Bracarda S, Iacovelli R, Boni L. et al. Sunitinib administered on 2/1 schedule in patients with metastatic renal cell carcinoma: the RAINBOW analysis. Ann Oncol. 2015;26:2107-2113
21. Neri B, Vannini A, Brugia M. et al. Biweekly sunitinib regimen reduces toxicity and retains efficacy in metastatic renal cell carcinoma: a single-center experience with 31 patients. Int J Urol. 2013;20:478-483
22. Bjarnason GA, Khalil B, Hudson JM. et al. Outcomes in patients with metastatic renal cell cancer treated with individualized sunitinib therapy: correlation with dynamic microbubble ultrasound data and review of the literature. Urol Oncol. 2014;32:480-487
23. Pan X, Huang H, Huang Y. et al. Sunitinib dosing schedule 2/1 improves tolerability, efficacy, and health-related quality of life in Chinese patients with metastatic renal cell carcinoma. Urol Oncol. 2015;33:268.e9-15
24. Miyake H, Harada K, Miyazaki A. et al. Improved health-related quality of life of patients with metastatic renal cell carcinoma treated with a 2 weeks on and 1 week off schedule of sunitinib. Med Oncol. 2015;32:78
25. Lee JL, Kim MK, Park I. et al. Randomized phase II trial of Sunitinib four weeks on and two weeks off versus Two weeks on and One week off in metastatic clear-cell type REnal cell carcinoma: RESTORE trial. Ann Oncol. 2015;26:2300-2305
26. Eisenhauer EA, Therasse P, Bogaerts J. et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 2009;45:228-247
27. National Cancer Institute (U.S). Common Terminology Criteria for Adverse Events v4.0. Bethesda, Md: U.S Dept of Health and Human Services, National Institutes of Health, National Cancer Institute; 2009.
28. Houk BE, Bello CL, Kang D. et al. A population pharmacokinetic meta-analysis of sunitinib malate (SU11248) and its primary metabolite (SU12662) in healthy volunteers and oncology patients. Clin Cancer Res. 2009;15:2497-2506
29. Britten CD, Kabbinavar F, Hecht JR. et al. A phase I and pharmacokinetic study of sunitinib administered daily for 2 weeks, followed by a 1-week off period. Cancer Chemother. Pharmacol. 2008;61:515-524
30. Rini BI, Cohen DP, Lu DR. et al. Hypertension as a biomarker of efficacy in patients with metastatic renal cell carcinoma treated with sunitinib. J Natl Cancer Inst. 2011;103:763-773
31. Donskov F, Michaelson MD, Puzanov I. et al. Sunitinib-associated hypertension and neutropenia as efficacy biomarkers in metastatic renal cell carcinoma patients. Br J Cancer. 2015;113:1571-1580
32. Rautiola J, Donskov F, Peltola K. et al. Sunitinib-induced hypertension, neutropaenia and thrombocytopaenia as predictors of good prognosis in patients with metastatic renal cell carcinoma. BJU Int. 2016;117:110-117
Author contact
![]() Corresponding authors: Jia Wang and Hao Zeng. Department of Urology, Institute of Urology, West China Hospital, Sichuan University, No. 37 Guoxue Xiang, Chengdu, Sichuan, China, 610041; Tel.: +86 18980602129; E-mail: cdhx510com(Hao zeng); Fax: +86 2885422451.
Corresponding authors: Jia Wang and Hao Zeng. Department of Urology, Institute of Urology, West China Hospital, Sichuan University, No. 37 Guoxue Xiang, Chengdu, Sichuan, China, 610041; Tel.: +86 18980602129; E-mail: cdhx510com(Hao zeng); Fax: +86 2885422451.

 Global reach, higher impact
Global reach, higher impact