3.2
Impact Factor
ISSN: 1837-9664
J Cancer 2019; 10(2):416-417. doi:10.7150/jca.29461 This issue Cite
Letter To The Editor
Bevacizumab Added to Neoadjuvant Chemotherapy in HER2-Negative Non-Metastatic Breast Cancer
Department of Oncology, Renmin Hospital, Hubei University of Medicine, Shiyan 442000, China.
Received 2018-8-25; Accepted 2018-11-1; Published 2019-1-1
To the Editor,
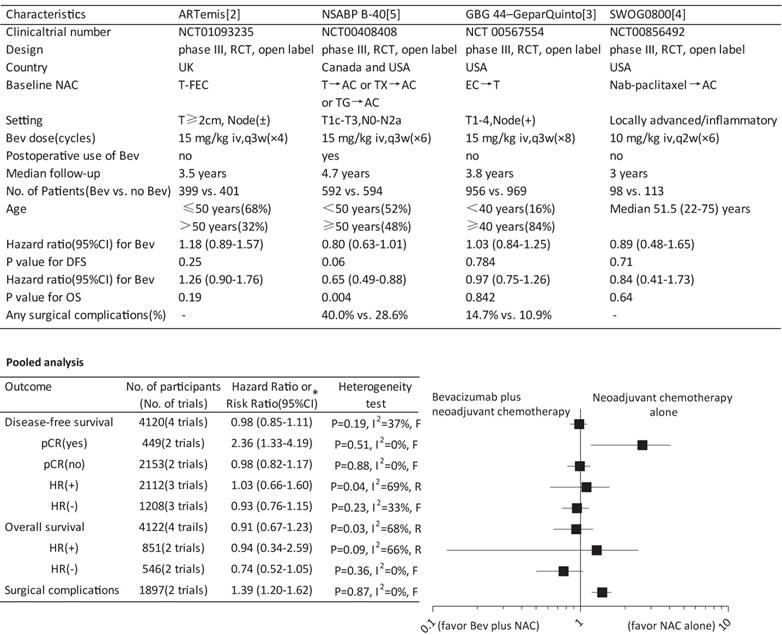
Recombinant humanized monoclonal antibody bevacizumab binding and inactivating VEGF-A has recently been a particularly attractive focus of research in anti-angiogenic treatment strategy for breast cancer especially the HER-2 negative subtype [1]. Three recently published phase III randomized controlled trials-ARTemis [2], GBG 44- GeparQuinto [3] and SWOG S0800 trial [4]-have showed that adding bevacizumab to neoadjuvant chemotherapy (NAC) increased the pathological complete response (pCR) but did not favor the survival of HER-2 negative non-metastatic breast cancer (NMBC), while NSABP B-40 trial [5] contradicted the findings by demonstrating an improved survival particularly disease-free survival (DFS). To settle the disputes, we therefore performed a meta-analysis to evaluate the survival benefit and risk of this treatment strategy.
Involving 4122 participants with a median follow-up from 3 to 4.7 year, the pooled data suggested no significant effect of bevacizumab on either DFS or overall survival (OS). Similar results on DFS were also found according to the hormone receptor (HR) status. Unexpectedly, a significantly reduced DFS was indicated in patients achieving a pCR (hazard ratio, 2.36; 95%CI, 1.33-4.19) while not in patients without a pCR. The stratification analyses by HR status regarding OS showed no significant effect as well (Figure 1). Involving two trials with a total of 1897 participants, the result showed significantly increased risk of any surgical complications (risk ratio, 1.39; 95%CI, 1.20-1.62) in patients receiving NAC and neoadjuvant bevacizumab (Figure 1).
Although an individual patient-level meta-analysis would be ideal, our results highlighted that adding bevacizumab to NAC for HER-2 negative NMBC did not provide survival benefit but significantly increased the risk of surgical complications. Also, the results did not support the role of HR status in discriminating the effect of bevacizumab, which contradicted the finding from NSABP-B40 that addition of bevacizumab resulted in improved survival especially in HR-positive patients. In NSABP B40 but not in other trials, patients received bevacizumab not just preoperatively but also postoperatively, such substantial differences may have contributed to the discordant results because bevacizumab was able to affect not only the primary tumor but also dormant micrometastases. Although most previous trials incorporating the currently included trials showed consistently an increased pCR rate with neoadjuvant bevacizumab, reduced DFS was revealed in patients achieving pCR in the present study, suggesting that the pCR advantage seemed not always to be translated into a survival advantage. Our analysis failed to demonstrate this benefit while confirmed increased toxicity, supporting utmost cautions against the adoption of neoadjuvant bevacizumab in this setting.
Acknowledgements
GXW contributed to the initial concept, protocol writing, data interpretation and manuscript writing; FJC contributed to data collection and interpretation, manuscript review; XBW and XS helped with protocol writing, data collection and interpretation.
Competing Interests
The authors have declared that no competing interest exists.
Survival and surgical complications for bevacizumab plus neoadjuvant chemotherapy versus neoadjuvant chemotherapy alone. Bev, bevacizumab; NAC, neoadjuvant chemotherapy; RCT, randomized controlled trial; T, docetaxel; FEC, fluorouracil plus epirubicin plus cyclophosphamide; AC, doxorubicin plus cyclophosphamide; TX, docetaxel plus capecitabine; TG, docetaxel plus gemcitabine; EC, epirubicin plus cyclophosphamide; q3w, every third week; iv, intravenously; DFS, disease-free survival; OS, overall survival; pCR, pathological complete response; HR, hormone receptor * A hazard ratio or risk ratio less than 1 favors the outcome by Bev plus NAC, while a hazard ratio or risk ratio greater than 1 favors the outcome by NAC alone. F, fixed-effect model; R, random-effect model.
References
1. Kumler I, Christiansen OG, Nielsen DL. A systematic review of bevacizumab efficacy in breast cancer. Cancer Treat Rev. 2014;40(8):960-73
2. Earl HM, Hiller L, Dunn JA. et al. Disease-free and overall survival at 3.5 years for neoadjuvant bevacizumab added to docetaxel followed by fluorouracil, epirubicin and cyclophosphamide, for women with HER2 negative early breast cancer: ARTemis Trial. Ann Oncol. 2017;28(8):1817-24
3. von Minckwitz G, Loibl S, Untch M. et al. Survival after neoadjuvant chemotherapy with or without bevacizumab or everolimus for HER2-negative primary breast cancer (GBG 44-GeparQuinto) dagger. Ann Oncol. 2014;25(12):2363-72
4. Nahleh ZA, Barlow WE, Hayes DF. et al. SWOG S0800 (NCI CDR0000636131): addition of bevacizumab to neoadjuvant nab-paclitaxel with dose-dense doxorubicin and cyclophosphamide improves pathologic complete response (pCR) rates in inflammatory or locally advanced breast cancer. Breast Cancer Res Treat. 2016;158(3):485-95
5. Bear HD, Tang G, Rastogi P. et al. Neoadjuvant plus adjuvant bevacizumab in early breast cancer (NSABP B-40 [NRG Oncology]): secondary outcomes of a phase 3, randomised controlled trial. Lancet Oncol. 2015;16(9):1037-48
Author contact
![]() Corresponding author: Guoxing Wan, MD, PhD, Department of Oncology, Renmin Hospital, Hubei University of Medicine, 39 Chaoyang Road, 442000 Shiyan, China. Phone: +86-719-8637385; Fax: +86-719-8637385; Email: 15gxwanedu.cn
Corresponding author: Guoxing Wan, MD, PhD, Department of Oncology, Renmin Hospital, Hubei University of Medicine, 39 Chaoyang Road, 442000 Shiyan, China. Phone: +86-719-8637385; Fax: +86-719-8637385; Email: 15gxwanedu.cn

 Global reach, higher impact
Global reach, higher impact