Impact Factor ISSN: 1837-9664
J Cancer 2024; 15(3):764-775. doi:10.7150/jca.90051 This issue Cite
Research Paper
Exploring the biological behavior and underlying mechanism of KITLG in triple-negative breast cancer
1. Department of Gastrointestinal Gland Surgery, The First Affiliated Hospital of Guangxi Medical University, Nanning 530021, Guangxi, China.
2. The Thyroid and Breast Surgery Department, The First People's Hospital of Qinzhou, Qinzhou 535000, Guangxi, China.
†Liuqing Qin, Yuchao Li, Yifei Huang contributed equally to this work and should be considered as co-first authors.
Abstract
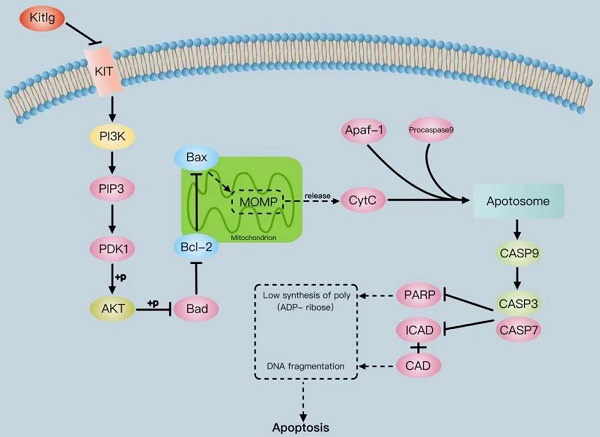
The tyrosine-kinase receptor that is specified by the KIT locus is demarcated by KITLG. This multifaceted factor is instrumental during in-utero germ and neural cell maturation and hematopoiesis, ostensibly reflecting its role in facilitating cell migration. Concurrently, KITLG is prone to a mutation in germ cell tumors, entailing a presumed connection to tumorigenesis. Despite this, the intricacies of its function in breast cancer and the relevant mechanisms remain elusive. Multiple independent databases depict a consistently low expression of KITLG within tissues affected by triple-negative breast cancers (TNBC), a trend strongly coupled with reduced survival rates. Interestingly, non-triple-negative breast cancers exhibit a markedly high expression of KITLG compared to the norm. An initial analysis of the GEO database speculates that KITLG may serve as an oncogene suppressor in TNBC, hinting at varied roles for KITLG isoforms within this disease context. In conclusion, our preliminary analysis offers valuable insights into the role and expression pattern of KITLG in TNBC. We provide evidence supporting its consideration as a promising new prognostic marker, thereby potentially enriching therapeutic strategies for TNBC. Indeed, given the limited advances in molecularly targeted therapy for TNBC, a significant need exists for a more precise therapeutic approach and a comprehensive understanding of its inherent mechanisms of action.
Keywords: triple-negative breast cancer, KITLG, biological behavior, mechanism

 Global reach, higher impact
Global reach, higher impact