3.2
Impact Factor
ISSN: 1837-9664
J Cancer 2024; 15(4):955-965. doi:10.7150/jca.90391 This issue Cite
Research Paper
Unraveling the Action Mechanism of Tubeimoside-1 against Tumor Microvessels via Network Pharmacology and Experimental Validation
1. Department of Clinical Laboratory, Qilu Hospital, Shandong University (Qingdao), Qingdao, Shandong 266035, China.
2. Department of Clinical Laboratory, Qilu Hospital, Shandong University, Jinan, Shandong 250012, China.
3. Department of Clinical Laboratory, Shandong University School Hospital, Jinan, Shandong, 250012, China.
*These two corresponding authors contribute equally to this article.
Abstract
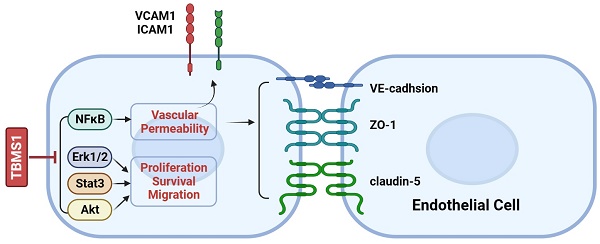
Objective: Tubeimoside-1 (TBMS1) is a plant-derived triterpenoid saponin that exhibits pharmacological properties and anti-tumor effects, but the anti-tumor microvessels of action of TBMS1 remains to be completely elucidated. This study aims to verify the effect of TBMS1 on tumor microvessels and its underlying mechanism.
Methods: A SKOV3 xenografted mouse model were constructed to evaluate the anti-tumor microvessels of TBMS1 in vivo, followed by function assays to verify the effects of TBMS1 on the proliferation, cell cycle, migration, and tubule formation of vascular endothelial cells in vitro. Next, based on network pharmacology, the drug/disease-target protein-protein interaction (PPI) networks, biological functions and gene enrichment analyses were performed to predict the underlying mechanism. Finally, molecules and pathways associated with tumor trans-endothelial migration were identified.
Results: TBMS1 treatment effectively reduced tumor microvessel density in ovarian cancer model and inhibited the proliferation, cell cycle, migration, and induced apoptosis of vascular endothelial cells in vitro. Network pharmacological data suggested that tumor cell adhesion and trans-endothelial migration may participate in antiangiogenic effects of TBMS1. By endothelial adhesion and permeability assay, we identified that tumor adhesion and the permeability of endothelial monolayers were reduced by TBMS1. Furthermore, adhesion protein (VCAM-1and ICAM-1) and tight junction (TJ) proteins (VE-cadhsion, ZO-1 and claudin-5) were found to be regulated. Finally, Akt, Erk1/2, Stat3 and NF-κB signaling were decreased by TBMS1 treatment.
Conclusion: To sum up, our findings strongly suggest that clinical application of TBSM1 may serve as a vasoactive drug treatment to suppress tumor progression.
Keywords: TBMS1, trans-endothelial migration, adhesion protein, tight junction, network pharmacology.

 Global reach, higher impact
Global reach, higher impact