Impact Factor ISSN: 1837-9664
J Cancer 2026; 17(4):890-897. doi:10.7150/jca.126211 This issue Cite
Research Paper
Association between Oxidative Balance Score and Colorectal Cancer: Insights from NHANES 1999-2018
1. Department of Epidemiology and Health Statistics, Fujian Provincial Key Laboratory of Environment Factors and Cancer, School of Public Health, Fujian Medical University, Fuzhou, 350108, China.
2. Key Laboratory of Gastrointestinal Cancer (Fujian Medical University), Ministry of Education, School of Basic Medical Sciences, Fujian Medical University, Fuzhou 350108, China.
† These authors contributed equally to this work.
Received 2025-10-2; Accepted 2026-3-25; Published 2026-4-8
Abstract
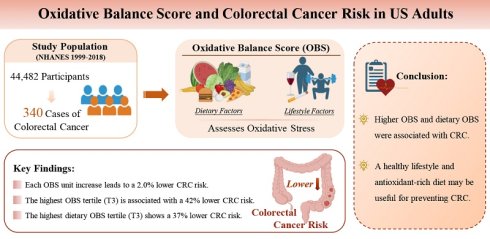
Background: Previous studies have indicated an association between high antioxidant exposure and colorectal cancer (CRC) risk in elderly populations. However, the relationship between the oxidative balance score (OBS) and CRC risk in the general population remains unclear.
Objective: In this study, we used the OBS, which is based on dietary and lifestyle factors, to assess the oxidative stress (OS) status of individuals and to explore the association between OBS and CRC risk in American adults.
Methods: Overall, 44,482 participants from the National Health and Nutrition Examination Survey (NHANES 1999-2018) were included in the cross-sectional study, of whom 340 had CRC. OBS, which consists of 20 dietary and lifestyle factors, was the exposure variable. Weighted multivariate logistic regression, subgroup analysis, and restricted cubic spline curves were used to assess the association between OBS and CRC. Sensitivity analyses were conducted to evaluate the robustness of the results.
Results: Multiple logistic regression showed that the CRC risk decreased by 2.0% for each OBS unit added (OR= 0.98, 95%CI: 0.96-1.00, P=0.019). Compared with the lowest OBS reference group (T1), OBS in the highest tertile of OBS (T3) was associated with a reduced CRC risk (OR= 0.58, 95%CI: 0.39-0.84). Similarly, individuals in the highest dietary OBS tertile (T3) had a reduced CRC risk (OR= 0.63, 95%CI: 0.44-0.91), whereas no significant association was observed in lifestyle OBS and CRC.
Conclusion: Higher OBS and dietary OBS were associated with CRC. A healthy lifestyle and antioxidant-rich diet may be useful for preventing CRC.
Keywords: colorectal cancer, oxidative stress, oxidative balance score, risk, cross-sectional study
Introduction
Colorectal cancer (CRC) is the third most common cancer worldwide and the second leading cause of cancer-related deaths(1, 2). Although progress has been made in the treatment of CRC, improving overall survival, its incidence continues to increase(3). It is estimated that there were more than 1.9 million new cases of CRC and 904,000 deaths in 2022, with a trend toward a younger population(1, 4). Research indicates that CRC is ranked as the third leading cause of cancer-related deaths in U.S. adults younger than 50 years old(5). Notably, CRC in the younger population is usually characterized by rapid progression and aggressiveness(6, 7). As a result, CRC has become an increasingly serious health problem.
Oxidative stress (OS) is normally thought to be caused by an imbalance between the production and removal of reactive oxygen species (ROS) and plays an important role in the development of various cancers, including gastric, breast and colorectal cancers(8-11). Several existing studies have found that higher levels of ROS can disrupt the function of biomolecules such as lipids, nucleic acids, and proteins, which can promote carcinogenesis(9, 12). Sharifi-Rad et al.(13) demonstrated that unhealthy dietary patterns and poor lifestyle habits may lead to the production of ROS, which can result in OS. Individual's oxidative balance is determined by the interaction of anti-oxidants and pro-oxidants. The oxidative balance score (OBS) is a way to measure exposure to anti-oxidants and pro-oxidants in diet and lifestyle, which reflect the overall balance between pro-oxidants and antioxidants(14, 15). Researchers have combined multiple dietary and lifestyle exposures to estimate the OBS, which has been extensively used to assess the relationship between overall oxidative status and the risk of chronic diseases(14). Recently, several epidemiologic studies have shown that a higher OBS is associated with a lower risk of chronic kidney disease, chronic obstructive pulmonary disease, non-alcoholic fatty liver disease, and other chronic diseases(16-18).
Furthermore, the association between OBS and cancer risk has also drawn significant attention. Research indicates that higher OBS is associated with a reduced risk of developing breast cancer, ovarian cancer, cervical cancer, and other types of cancer(19, 20). Chang Y et al. found that a higher dietary OBS was associated with a lower risk of gastrointestinal (GI) cancers (OR: 0.90, 95% CI: 0.76-0.99), indicating that oxidative stress imbalance may be involved in the development of GI cancers(21). Currently, studies have explored the association between OBS and the risk of colorectal tumors. Several available case-control studies have found that higher OBS is associated with a reduced risk of colorectal adenomas(15, 22-24). Meanwhile, researchers have explored the association between OBS and CRC in elderly populations. Dash C et al. found that higher antioxidant levels were negatively associated with CRC risk in people older than 50 years old(25). In addition, recent studies have shown that older women with higher antioxidant exposure have a lower risk of CRC(26, 27). However, existing research has primarily focused on elderly or specific gender populations. The relationship between OBS and CRC risk in the general population remains poorly understood, and further validation in larger, representative populations is warranted.
In this study, we analyzed a representative sample of adults from the United States (US), obtained from the National Health and Nutrition Examination Survey (NHANES) 1999-2018. We used the OBS(28), which is based on 20 pro-oxidant and antioxidant factors, to assess the OS status of individuals and to explore the association between OBS and CRC.
Methods
Sources of data and study population
The data were extracted from NHANES 1999-2018, which are all available on the NHANES website (https://wwwn.cdc.gov/nchs/nhanes/Default.aspx). Of the 101,316 participants in NHANES 1999-2018, a total of 44,482 participants were included in the final analysis (Supplementary Figure 1). Exclusion criteria were as follows: a) missing information on cancer questionnaire; b) participants with cancers other than colorectal cancer; c) missing information on more than 4 out of the 20 components of the OBS; d) participants with missing weighting data.
The NHANES methods were authorized by the Ethics Review Board of the U.S. Centers for Disease Control and Prevention (CDC) National Center for Health Statistics, and all participants provided written informed consent. Therefore, analysis of the anonymized data did not require additional ethical review board approval (29). This study was conducted in accordance with the guidelines of the 1975 Declaration of Helsinki, as revised in 2013, and followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for reporting of cross-sectional studies.
Calculation of the oxidation balance score
OBS was calculated based on 16 dietary nutrients and 4 lifestyle factors that have been shown to influence oxidative stress(14, 30). Dietary intake data were collected through 24-hour dietary recall interviews in NHANES, providing information on alcohol intake and 16 dietary nutrients such as fiber, carotenoids, riboflavin, niacin, total folate, vitamins B6, B12, C, and E, calcium, magnesium, zinc, copper, selenium, total fat, and iron. Lifestyle components considered included physical activity, body mass index (BMI), alcohol consumption, and smoking, with cotinine levels used to assess smoking. Physical activity was quantified by the total time spent walking, engaging in moderate-intensity and vigorous activity throughout the week. Pro-oxidant factors included total fat, iron consumption, smoking, alcohol intake, and obesity, whereas the remaining components were classified as antioxidants.
Each dietary component was categorized into three groups according to sex-specific tertiles. Antioxidants were scored from 0 to 2 and pro-oxidants from 2 to 0 (Supplementary Table 1). Participants with data available for at least 16 of the 20 OBS components were included in the study. For components with missing data, a score of 0 was assigned to the OBS. The total OBS, ranging from 0 to 40 in this study, was calculated by summing the scores across all 20 components, with higher values reflecting greater overall antioxidant exposure and lower oxidative stress potential.
Identification of CRC cases
In the Medical Conditions section of the NHANES questionnaire, participants were asked the questions “Have you ever been told by a doctor or other health professional that you had cancer?” and “What kind of cancer?” Participants were defined as CRC cases if they reported a previous diagnosis of colon or rectal cancer. CRC cases were identified using the International Classification of Diseases for Oncology (ICD-O) codes for colon cancer (C18) and rectal cancer (C19-C20).
Covariates
The study included confounding factors that may be associated with CRC, including age, sex, education, race, family income, hypertension, and diabetes. Education was grouped into three categories: less than high school, high school, or more than high school. Race was categorized as non-Hispanic White or others. Poverty income ratio (PIR) was classified into three categories: < 1.3, 1.3-3.5, or ≥ 3.5. Participants were classified as hypertensive if they had a self-reported history of hypertension, were currently taking antihypertensive medication, or had an average systolic blood pressure ≥ 140 mmHg or diastolic blood pressure ≥ 90 mmHg, based on at least three measurements. A self-reported history of diabetes or being on glucose-lowering medication or insulin were diagnosed as diabetes.
Statistical analysis
Due to NHANES' complex sampling design, all analyses accounted for sample weights, strata, and clusters. For continuous and categorical variables, distributions were compared using the Mann-Whitney U and χ2 tests, respectively. These variables were then expressed as medians, interquartile ranges (IQR) and numbers (percentages), respectively. Odds ratios (OR) and 95% confidence intervals (95%CI) were calculated using survey-weighted binary logistic regression models to estimate the association between OBS and CRC. A total of three models were developed with stepwise inclusion of covariates: Model 1, which was unadjusted for any covariates; Model 2, adjusted for sociodemographic factors such as sex, age, education, race, and PIR; and Model 3, which further included adjustments for diabetes and hypertension. To better understand the relationship between OBS and CRC, OBS was separated into dietary and lifestyle components. Stratified analyses were conducted based on sex, age, education, race, PIR, hypertension, and diabetes to further investigate the relationship between OBS and CRC in various subgroups. An interaction test was conducted by including the OBS subgroup product term in a weighted multivariate logistic regression model to assess the difference between models with and without the interaction term. To examine the nonlinear relationship between OBS and CRC, restricted cubic spline (RCS) analysis was employed. Finally, a sensitivity analysis was conducted to assess the robustness of the results. The relationship between OBS and CRC was re-evaluated using weighted multivariate logistic regression, excluding participants with fewer than 19 OBS score items. Additionally, a 1:4 propensity score matching method was used to further assess the relationship between OBS and CRC.
Statistical analyses were performed using R software (version 4.3.3) and relevant R packages (“mice”, “survey”, “rms”, etc.). Statistical significance was set at a two-sided P value < 0.05.
Results
General characteristics of participants
A total of 44,482 participants from NHANES 1999-2018 were included in the cross-sectional study, of whom 340 had CRC. Participants with CRC were older, more likely to be non-Hispanic White and to have middle level of income. However, no significant differences were observed in the distribution of sex and education levels between the two groups (Table 1). The baseline characteristics comparison revealed that CRC patients had a higher prevalence of hypertension and diabetes. Compared with participants without CRC, participants with CRC showed significantly lower OBS, dietary OBS, and lifestyle OBS.
General characteristics of the study subjects
| Overall N = 444821 | Non-CRC N = 441421 | CRC N = 3401 | P-value2 | |
|---|---|---|---|---|
| Age(years) | < 0.001 | |||
| < 50 | 24261 (60%) | 24237 (60%) | 24 (9%) | |
| ≥ 50 | 20221 (40%) | 19905 (40%) | 316 (91%) | |
| Sex | 0.300 | |||
| Male | 21497 (49%) | 21324 (49%) | 173 (45%) | |
| Female | 22985 (51%) | 22818 (51%) | 167 (55%) | |
| Education | 0.200 | |||
| Less than high school | 12121 (17%) | 12005 (17%) | 116 (22%) | |
| High school diploma | 10351 (24%) | 10275 (24%) | 76 (22%) | |
| More than high school | 22010 (58%) | 21862 (58%) | 148 (55%) | |
| Race | < 0.001 | |||
| Other Hispanic | 18883 (67%) | 18667 (66%) | 216 (82%) | |
| Other Race | 25599 (33%) | 25475 (34%) | 124 (18%) | |
| Family PIR | 0.047 | |||
| < 1.3 | 14066 (23%) | 13962 (23%) | 104 (21%) | |
| 1.3-3.5 | 16912 (36%) | 16763 (36%) | 149 (44%) | |
| ≥ 3.5 | 13504 (41%) | 13417 (41%) | 87 (35%) | |
| Hypertension | < 0.001 | |||
| No | 26631(65%) | 26554(65%) | 77(25%) | |
| Yes | 17851 (35%) | 17588 (35%) | 263 (75%) | |
| Diabetes | < 0.001 | |||
| No | 38992(91%) | 38750(91%) | 242(71%) | |
| Yes | 5490 (9%) | 5392 (9%) | 98 (29%) | |
| OBS | 20 (14, 26) | 20 (14, 26) | 18 (13, 22) | < 0.001 |
| Dietary OBS | 16 (10, 22) | 16 (10, 22) | 15 (9, 18) | < 0.001 |
| Lifestyle OBS | 4 (3, 5) | 4.00 (3, 5) | 3 (2, 5) | 0.007 |
1N (unweighted) (%); Median (IQR)
2Chi-squared test with Rao & Scott's second-order correction; Wilcoxon rank-sum test for complex survey samples
Association between OBS and CRC
The weighted logistic regression analysis indicated a significant negative association between OBS and CRC (Table 2). After adjusting for age, sex, education, race, PIR, hypertension, and diabetes (Model 3), each 1-point increase in OBS was associated with 2.0% lower odds of CRC (OR = 0.98, 95%CI: 0.96-1.00, P = 0.019). Compared with the lowest tertile (T1), participants in the highest tertile (T3) were associated with lower odds of CRC (OR = 0.58, 95%CI: 0.39-0.84).
Odds ratio for the association between the OBS and CRC
| OBS | Categorical models OR (95%CI) | Continuous models OR (95%CI) | |||||
|---|---|---|---|---|---|---|---|
| T1 | T2 | T3 | P trend | Per 1 point increase in score | P value | ||
| Model 1 | Ref. | 1.03(0.74,1.44) | 0.46(0.32,0.66) | <0.001 | 0.97(0.95, 0.98) | <0.001 | |
| Model 2 | Ref. | 1.01(0.72,1.40) | 0.49(0.33,0.72) | <0.001 | 0.97(0.95, 0.99) | 0.001 | |
| Model 3 | Ref. | 1.09(0.79,1.50) | 0.58(0.39,0.84) | 0.005 | 0.98(0.96, 1.00) | 0.019 | |
Model 1: Adjusted with no covariates.
Model 2: Adjusting for age, sex, education, race and PIR.
Model 3: Additionally, adjusted for hypertension and diabetes.
Association between dietary OBS/lifestyle OBS and CRC
Table 3 displays the association of dietary OBS and lifestyle OBS with CRC. In Model 3, compared with the lowest dietary OBS (T1), the highest tertile (T3) was associated with lower odds of CRC (OR= 0.63, 95%CI: 0.44-0.91). Meanwhile, dietary OBS was analyzed as a continuous variable. The results showed that every 1-point increase in dietary OBS was associated with 2.0% lower odds of CRC (P = 0.021). In contrast the lifestyle OBS showed no significant association with CRC, whether analyzed as categorical or continuous variable (P > 0.05). However, there was no multiplicative interaction between dietary and lifestyle OBS (P for interaction = 0.303).
Odds ratio for the association between dietary OBS/lifestyle OBS and CRC
| OBS | Categorical models OR (95%CI) | Continuous models OR (95%CI) | |||||
|---|---|---|---|---|---|---|---|
| T1 | T2 | T3 | P trend | Per 1 point increase in score | P value | ||
| Dietary OBS | |||||||
| Model 1 | Ref. | 0.96 (0.68,1.34) | 0.52 (0.37,0.73) | <0.001 | 0.97 (0.95, 0.99) | <0.001 | |
| Model 2 | Ref. | 0.95 (0.68,1.31) | 0.56 (0.39,0.81) | 0.002 | 0.97 (0.95, 0.99) | 0.003 | |
| Model 3 | Ref. | 1.00 (0.73,1.38) | 0.63 (0.44,0.91) | 0.013 | 0.98 (0.96, 1.00) | 0.021 | |
| Lifestyle OBS | |||||||
| Model 1 | Ref. | 0.77 (0.58,1.03) | 0.61 (0.38,0.99) | 0.046 | 0.90 (0.84, 0.98) | 0.009 | |
| Model 2 | Ref. | 0.77 (0.58,1.04) | 0.66 (0.41,1.06) | 0.083 | 0.91 (0.84, 0.99) | 0.022 | |
| Model 3 | Ref. | 0.86 (0.66,1.21) | 0.86 (0.52,1.41) | 0.600 | 0.97 (0.89, 1.06) | 0.500 | |
Model 1: Adjusted with no covariates.
Model 2: Adjusting for age, sex, education, race and PIR.
Model 3: Additionally, adjusted for hypertension and diabetes.
Strata analysis
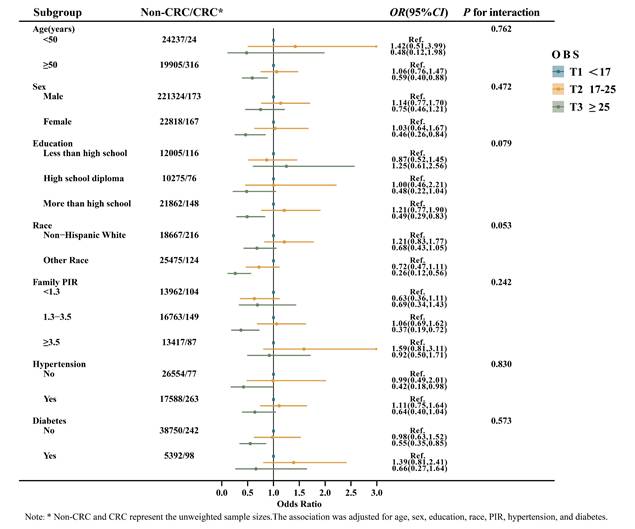
OBS was analyzed as a categorical variable across subgroups defined by age, sex, education, race, PIR, hypertension, and diabetes status (Figure 1). The subgroup analysis revealed a significant relationship between OBS and CRC among individuals aged ≥ 50, female, with more than a high school education, of other races, PIR between 1.3-3.5, and those without hypertension or diabetes (P < 0.05). Nevertheless, a multiplicative interaction between each of the above variables and OBS was not observed (P for interaction > 0.05). In addition, dietary OBS was further stratified according to variables of interest (Supplementary Figure 2). The results suggested a consistent negative association between dietary OBS and CRC among individuals aged ≥ 50, female, other race, PIR between 1.3-3.5, and those without diabetes (P < 0.05). No multiplicative interaction was observed between these variables and dietary OBS (P for interaction > 0.05).
Subgroup analyses of the association between OBS and CRC.
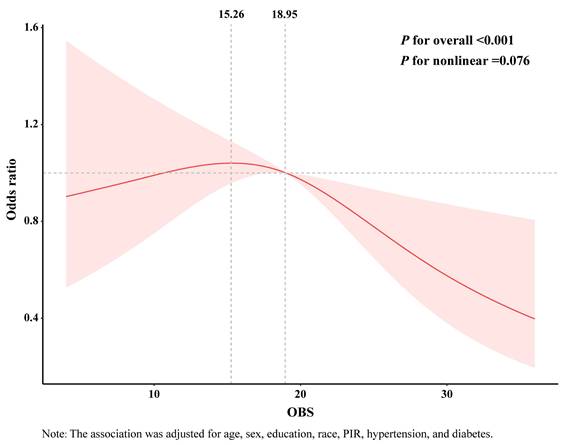
RCS analysis
RCS analysis revealed an inverted J-shaped relationship between OBS and CRC (P for nonlinear = 0.076) (Figure 2). When the OBS score exceeded 15.26, the curve showed a decreasing trend. Furthermore, when the score surpassed 18.95, higher OBS was associated with lower odds of CRC. Similarly, the RCS curves for the association of dietary OBS and CRC exhibited a comparable trend (Supplementary Figure 3).
RCS analysis of the association between OBS and CRC. The association was adjusted for age, sex, education, race, hypertension, and diabetes.
Sensitivity analysis
We performed a sensitivity analysis to assess the robustness of the findings (Table 4). After excluding participants with fewer than 19 OBS components, weighted multivariable logistic regression analyses indicated that compared with the lowest tertile (T1) of OBS, the highest tertile (T3) of OBS was also associated with lower odds of CRC (OR = 0.64, 95%CI: 0.43-0.96). Additionally, a 1:4 propensity score matching approach was employed to reassess the association between OBS and CRC. The results consistently showed that individuals in the highest tertile (T3) of OBS had lower odds of CRC compared with those in the lowest tertile (T1) (OR= 0.64, 95%CI: 0.43-0.95). These analyses indicate that the inverse association between OBS and CRC remained consistent.
Odds ratio for the association between the OBS and CRC in sensitivity analysis
| OBS | Exclusion of OBS score items less than 19 | 1:4 PSM | ||||
|---|---|---|---|---|---|---|
| OR (95%CI) | P for trend* | OR (95%CI) | P for trend* | |||
| T1 | Ref. | 0.030 | Ref. | 0.026 | ||
| T2 | 1.25 (0.90,1.74) | 1.16 (0.79,1.70) | ||||
| T3 | 0.64 (0.43,0.96) | 0.64 (0.43,0.95) | ||||
* Adjusted for age, sex, education, race, PIR, hypertension, and diabetes.
Discussion
Results from this large-scale cross-sectional study using NHANES data indicated that higher categories of OBS and dietary OBS were strongly associated with lower odds of CRC when compared with the lowest categories. Additionally, the association between OBS and CRC was particularly evident among individuals aged ≥ 50 years, female, with more than a high school education, of other races, participants with a PIR of 1.3-3.5, and those without hypertension or diabetes. Overall, a higher OBS score was associated with lower odds of CRC.
An increase in OS is one of the main characteristics of cancer(31). Excessive ROS/RNS production causes OS, which damages DNA, resulting in cell cycle arrest, transcription factor induction, replication errors, and genomic instability, all of which are associated with CRC(32, 33). Meanwhile, population-based studies have shown that OS may be influenced by many modifiable lifestyle factors, including smoking, alcohol consumption, physical activity, and dietary factors (34, 35). Some foods and specific dietary components, such as red meat, fats, and refined carbohydrates, may act as potential pro-oxidants whereas vegetables, whole grains, tocopherols, carotenoids, and vitamin C may act as antioxidants (36-39). Lifestyle factors such as smoking and alcohol consumption will increase the production of reactive oxygen species(40-42). Currently, more than 20 OBSs have been widely used in the assessment of individual OS(43). OBS provides a convenient method a convenient way to assess pro-oxidant and antioxidant intake from combined diets and lifestyles, making it a promising tool.
In this study, an OBS score was constructed based on 16 dietary and 4 lifestyle indicators with regard to OS exposure to assess OS exposure and its association with CRC. The analysis revealed that a higher OBS was associated with lower odds of CRC, and this relationship remained consistent after controlling for potential confounders. Currently, the majority of evidence linking OS to CRC is derived from in vitro and animal studies, and the positive health effects of antioxidants on CRC are well documented(39). In addition, an Iranian case-control study found that a negative correlation existed between total antioxidant capacity (TAC) and CRC risk (ORQ3-Q1= 0.25; 95%CI: 0.13-0.46)(44). Another meta-analysis evaluated participants' oxidative stress status by derivatives of reactive oxygen metabolites (d-ROM) levels in blood samples, and showed that high oxidative stress was associated with an increased incidence of colorectal cancer (HR= 1.70; 95%CI: 1.15-2.51)(45). A recent large prospective cohort study of the UK biobank (UKB) explored the association between OBS and CRC and has found that higher OBS is negatively associated with the development of CRC (HRQ4-Q1= 0.713; 95%CI 0.636-0.799) (46). This evidence further confirms our research findings.
Similar to previous studies(26, 27), our study found that the association of OBS and dietary OBS with CRC was more pronounced (P < 0.05) in older individuals (≥ 50 years) and females. As with most cancers, CRC risk increases with age(47). Additionally, cellular senescence negatively affects tissues and organisms. In the elderly individuals, dysregulated clearance processes and the accumulation of senescent cells lead to chronic inflammation and oxidative stress(48, 49). These findings underscore the critical role of antioxidants in reducing CRC risk among the elderly population. Furthermore, estrogen exhibits antioxidant properties that enhance the body's defenses against oxidative stress(50). However, during menopause, the decline in estrogen levels renders women more reliant on external antioxidants to counteract oxidative stress and mitigate CRC risk.
The major strengths of our study include investigating the relationship between OBS and CRC for the first time in a nationally representative survey. In addition, we employed the OBS proposed by Zhang W et al.(28) based on NHANES dietary data, which may provide a more comprehensive assessment of overall oxidative stress compared with previous OBS methods(51). Furthermore, this study considered multivariable confounding factors and employed cross-validation using continuous and multiple logistic regression models, thereby enhancing the reliability of the findings. Sensitivity analyses were also performed to further confirm the reliability of the results.
Nevertheless, this study has several limitations. First, due to its cross-sectional design, causal inference cannot be made, and reverse causation cannot be excluded, as CRC status may have influenced dietary and lifestyle habits. Second, the relatively small number of CRC cases may limit statistical power. Therefore, although our study observed a significant association between OBS, dietary OBS, and CRC, further validation in large prospective studies is warranted.
In summary, this cross-sectional study of U.S. adults suggests that a higher OBS is associated with lower odds of CRC. The incidence of CRC continues to rise and has shown a trend toward younger populations. Therefore, a healthy lifestyle and a diet rich in antioxidants may be beneficial for overall health, although causal effects on CRC cannot be established. However, further prospective studies are required to confirm these findings.
Supplementary Material
Supplementary figures and tables.
Acknowledgements
The authors would like to express their gratitude to all participants for their cooperation.
Funding
This work was supported by the Natural Science Foundation of Fujian Province (No. 2023J01628) and the Natural Science Foundation Project (Master Youth Program, No. 2023J06030).
Author contributions
CDJ and XBQ contributed to conception, drafted the initial manuscript, prepared tables and figures. CDJ and FH contributed to data acquisition, performed the statistical analyses. CHJ, QWX and FJW contributed to critically reviewed the results and provided valuable feedback on the manuscript. PXE and WYL contributed to conception, design, and critically revised the manuscript. All authors gave their final approval and agree to be accountable for all aspects of the work.
Availability of data and materials
Data could be publicly available from the website (https://www.cdc.gov/nchs/nhanes/index.htm, access on 10 March 2024).
Competing Interests
The authors have declared that no competing interest exists.
References
1. Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, Jemal A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74(3):229-263
2. Siegel RL, Giaquinto AN, Jemal A. Cancer statistics, 2024. CA Cancer J Clin. 2024;74(1):12-49
3. Brouwer NPM, Bos A, Lemmens V, Tanis PJ, Hugen N, Nagtegaal ID. et al. An overview of 25 years of incidence, treatment and outcome of colorectal cancer patients. Int J Cancer. 2018;143(11):2758-2766
4. Done JZ, Fang SH. Young-onset colorectal cancer: A review. World J Gastrointest Oncol. 2021;13(8):856-866
5. Bhandari A, Woodhouse M, Gupta S. Colorectal cancer is a leading cause of cancer incidence and mortality among adults younger than 50 years in the USA: a SEER-based analysis with comparison to other young-onset cancers. J Investig Med. 2017;65(2):311-315
6. Medici B, Ricco B, Caffari E, Zaniboni S, Salati M, Spallanzani A. et al. Early Onset Metastatic Colorectal Cancer: Current Insights and Clinical Management of a Rising Condition. Cancers (Basel). 2023;15(13):3509
7. Lingas EC. Early-Onset Colon Cancer: A Narrative Review of Its Pathogenesis, Clinical Presentation, Treatment, and Prognosis. Cureus. 2023;15(9):e45404
8. Acevedo-Leon D, Monzo-Beltran L, Perez-Sanchez L, Naranjo-Morillo E, Gomez-Abril SA, Estan-Capell N. et al. Oxidative Stress and DNA Damage Markers in Colorectal Cancer. Int J Mol Sci. 2022;23(19):11664
9. Sawai K, Goi T, Sakamoto S, Matsunaka T, Maegawa N, Koneri K. Oxidative Stress as a Biomarker for Predicting the Prognosis of Patients with Colorectal Cancer. Oncology. 2022;100(11):612-619
10. Bel'skaya LV, Dyachenko EI. Oxidative Stress in Breast Cancer: A Biochemical Map of Reactive Oxygen Species Production. Curr Issues Mol Biol. 2024;46(5):4646-4687
11. Braga-Neto MB, Costa DVS, Queiroz DMM, Maciel FS, de Oliveira MS, Viana-Junior AB. et al. Increased Oxidative Stress in Gastric Cancer Patients and Their First-Degree Relatives: A Prospective Study from Northeastern Brazil. Oxid Med Cell Longev. 2021;2021:6657434
12. Jones DP. Radical-free biology of oxidative stress. Am J Physiol Cell Physiol. 2008;295(4):C849-868
13. Sharifi-Rad M, Anil Kumar NV, Zucca P, Varoni EM, Dini L, Panzarini E. et al. Lifestyle, Oxidative Stress, and Antioxidants: Back and Forth in the Pathophysiology of Chronic Diseases. Front Physiol. 2020;11:694
14. Goodman M, Bostick RM, Dash C, Flanders WD, Mandel JS. Hypothesis: oxidative stress score as a combined measure of pro-oxidant and antioxidant exposures. Ann Epidemiol. 2007;17(5):394-399
15. Goodman M, Bostick RM, Dash C, Terry P, Flanders WD, Mandel J. A summary measure of pro- and anti-oxidant exposures and risk of incident, sporadic, colorectal adenomas. Cancer Causes Control. 2008;19(10):1051-1064
16. Yin Y, Zhao C, Niu Y, Qi J, Zhang Y, Lu B. Associations between oxidative balance score and chronic kidney disease events in US adults: a population-based study. Sci Rep. 2024;14(1):13743
17. Liu Z, Zeng H, Zhang H. Association of the oxidation balance score with the prevalence of chronic obstructive pulmonary disease from the NHANES 2007-2012: A large-scale cross-sectional study. Heart Lung. 2024;65:84-92
18. Liu X, Wang Y, Liu X, Zeng B, Zhu B, Zhang Y. et al. Higher oxidative balance scores are associated with lower nonalcoholic fatty liver disease and not with fibrosis in US adults. Nutr Metab Cardiovasc Dis. 2023;33(12):2488-2496
19. Sohouli MH, Baniasadi M, Hernández-Ruiz Á, Melekoglu E, Zendehdel M, José Soto-Méndez M. et al. Adherence to Oxidative Balance Scores is Associated with a Reduced Risk of Breast Cancer; A Case-Control Study. Nutr Cancer. 2023;75(1):164-173
20. Yan Y, Zhang L, Yuan S, Zhang T, Shi C. Association between the oxidative balance score and gynecologic cancers from the National Health and Nutrition Examination Survey: An observational study. Medicine (Baltimore). 2025;104(39):e44782
21. Chang Y, Yu C, Dai X, Sun H, Tang T. Association of dietary inflammatory index and dietary oxidative balance score with gastrointestinal cancers in NHANES 2005-2018. BMC Public Health. 2024;24(1):2760
22. Goodman M, Bostick RM, Gross M, Thyagarajan B, Dash C, Flanders WD. Combined measure of pro- and anti-oxidant exposures in relation to prostate cancer and colorectal adenoma risk: an update. Ann Epidemiol. 2010;20(12):955-957
23. Dash C, Goodman M, Flanders WD, Mink PJ, McCullough ML, Bostick RM. Using pathway-specific comprehensive exposure scores in epidemiology: application to oxidative balance in a pooled case-control study of incident, sporadic colorectal adenomas. Am J Epidemiol. 2013;178(4):610-624
24. Kong SY, Bostick RM, Flanders WD, McClellan WM, Thyagarajan B, Gross MD. et al. Oxidative balance score, colorectal adenoma, and markers of oxidative stress and inflammation. Cancer Epidemiol Biomarkers Prev. 2014;23(3):545-554
25. Dash C, Bostick RM, Goodman M, Flanders WD, Patel R, Shah R. et al. Oxidative balance scores and risk of incident colorectal cancer in a US prospective cohort study. Am J Epidemiol. 2015;181(8):584-594
26. Mao Z, Prizment AE, Lazovich D, Gibbs DC, Bostick RM. Dietary and Lifestyle Oxidative Balance Scores and Incident Colorectal Cancer Risk among Older Women; the Iowa Women's Health Study. Nutr Cancer. 2021;73(11-12):2323-2335
27. Gu H, Li B, Xiang L, Xu Z, Tang Y, Zhu Z. et al. Association between oxidative stress exposure and colorectal cancer risk in 98,395 participants: results from a prospective study. Front Nutr. 2023;10:1284066
28. Zhang W, Peng SF, Chen L, Chen HM, Cheng XE, Tang YH. Association between the Oxidative Balance Score and Telomere Length from the National Health and Nutrition Examination Survey 1999-2002. Oxid Med Cell Longev. 2022;2022:1345071
29. NHANES. NCHS Research Ethics Review Board Approval 2022 [Available from: https://www.cdc.gov/nchs/nhanes/irba98.htm]
30. Schnell K, Weiss CO, Lee T, Krishnan JA, Leff B, Wolff JL, Boyd C. The prevalence of clinically-relevant comorbid conditions in patients with physician-diagnosed COPD: a cross-sectional study using data from NHANES 1999-2008. BMC Pulm Med. 2012;12:26
31. Gorrini C, Harris IS, Mak TW. Modulation of oxidative stress as an anticancer strategy. Nat Rev Drug Discov. 2013;12(12):931-947
32. Valko M, Rhodes CJ, Moncol J, Izakovic M, Mazur M. Free radicals, metals and antioxidants in oxidative stress-induced cancer. Chem Biol Interact. 2006;160(1):1-40
33. Dharshini LCP, Rasmi RR, Kathirvelan C, Kumar KM, Saradhadevi KM, Sakthivel KM. Regulatory Components of Oxidative Stress and Inflammation and Their Complex Interplay in Carcinogenesis. Appl Biochem Biotechnol. 2023;195(5):2893-2916
34. Clinton SK, Giovannucci EL, Hursting SD. The World Cancer Research Fund/American Institute for Cancer Research Third Expert Report on Diet, Nutrition, Physical Activity, and Cancer: Impact and Future Directions. J Nutr. 2020;150(4):663-671
35. Baniyash M, Sade-Feldman M, Kanterman J. Chronic inflammation and cancer: suppressing the suppressors. Cancer Immunol Immunother. 2014;63(1):11-20
36. Malesza IJ, Malesza M, Walkowiak J, Mussin N, Walkowiak D, Aringazina R. et al. High-Fat, Western-Style Diet, Systemic Inflammation, and Gut Microbiota: A Narrative Review. Cells. 2021;10(11):3164
37. Shi H, Schweren LJS, Ter Horst R, Bloemendaal M, van Rooij D, Vasquez AA. et al. Low-grade inflammation as mediator between diet and behavioral disinhibition: A UK Biobank study. Brain Behav Immun. 2022;106:100-110
38. Keshteli AH, Valcheva R, Nickurak C, Park H, Mandal R, van Diepen K. et al. Anti-Inflammatory Diet Prevents Subclinical Colonic Inflammation and Alters Metabolomic Profile of Ulcerative Colitis Patients in Clinical Remission. Nutrients. 2022;14(16):32394
39. Carini F, Mazzola M, Rappa F, Jurjus A, Geagea AG, Al Kattar S. et al. Colorectal Carcinogenesis: Role of Oxidative Stress and Antioxidants. Anticancer Res. 2017;37(9):4759-4766
40. Wu D, Zhai Q, Shi X. Alcohol-induced oxidative stress and cell responses. J Gastroenterol Hepatol. 2006;21(Suppl 3):S26-S29
41. Das SK, Vasudevan DM. Alcohol-induced oxidative stress. Life Sci. 2007;81(3):177-187
42. Kim M, Han CH, Lee MY. NADPH oxidase and the cardiovascular toxicity associated with smoking. Toxicol Res. 2014;30(3):149-157
43. Hernandez-Ruiz A, Garcia-Villanova B, Guerra-Hernandez EJ, Carrion-Garcia CJ, Amiano P, Sanchez MJ, Molina-Montes E. Oxidative Balance Scores (OBSs) Integrating Nutrient, Food and Lifestyle Dimensions: Development of the NutrientL-OBS and FoodL-OBS. Antioxidants (Basel). 2022;11(2):300
44. Rafiee P, Jafari Nasab S, Bahrami A, Rezaeimanesh N, Jalali S, Hekmatdoost A. et al. Dietary total antioxidant capacity and colorectal cancer and colorectal adenomatous polyps: a case-control study. Eur J Cancer Prev. 2021;30(1):40-45
45. Gào X, Wilsgaard T, Jansen EH, Holleczek B, Zhang Y, Xuan Y. et al. Pre-diagnostic derivatives of reactive oxygen metabolites and the occurrence of lung, colorectal, breast and prostate cancer: An individual participant data meta-analysis of two large population-based studies. Int J Cancer. 2019;145(1):49-57
46. Chang Y, Li F, Wang Z, Zhao Q, Wang Z, Han X. et al. Oxidative balance score: a potential tool for reducing the risk of colorectal cancer and its subsites incidences. Front Endocrinol (Lausanne). 2024;15:1397512
47. Giardina C, Kuo A, Nito K, Kurkcu S. Early onset colorectal cancer: Cancer promotion in young tissue. Biochem Pharmacol. 2024;226:116393
48. Collado M, Blasco MA, Serrano M. Cellular senescence in cancer and aging. Cell. 2007;130(2):223-233
49. Muñoz-Espín D, Serrano M. Cellular senescence: from physiology to pathology. Nat Rev Mol Cell Biol. 2014;15(7):482-496
50. Chainy GBN, Sahoo DK. Hormones and oxidative stress: an overview. Free Radic Res. 2020;54(1):1-26
51. Hernández-Ruiz Á, García-Villanova B, Guerra-Hernández E, Amiano P, Ruiz-Canela M, Molina-Montes E. A Review of A Priori Defined Oxidative Balance Scores Relative to Their Components and Impact on Health Outcomes. Nutrients. 2019;11(4):774
Author contact
Corresponding authors: Xian-E Peng, Mail address: Department of Epidemiology and Health Statistics, Xuefu North Road 1st, Shangjie Town, Minhou Country, Fuzhou, Fujian, 350108, China; Tel and Fax: +86-591-22862648; Email: fmuxecom. Yunli Wu, M.D., Ph, Mail address: Department of Epidemiology and Health Statistics, Xuefu North Road 1st, Shangjie Town, Minhou Country, Fuzhou, Fujian, 350108, China; Tel and Fax: +86-591-22862648.

 Global reach, higher impact
Global reach, higher impact