Impact Factor ISSN: 1837-9664
J Cancer 2019; 10(22):5494-5503. doi:10.7150/jca.34029 This issue Cite
Research Paper
Prognostic value of the albumin-to-alkaline phosphatase ratio on urologic outcomes in patients with non-metastatic renal cell carcinoma following curative nephrectomy
1. Department of Hematology, The Third Clinical Institute Affiliated to Wenzhou Medical University, People's Hospital of Wenzhou, Wenzhou, Zhejiang province, 325006, P.R. China
2. Department of Urology, Affiliated Hospital of Yangzhou University, Yangzhou, Jiangsu province, 225001, P.R. China
3. Department of Anorectal surgery, sixth affiliated hospital of Wenzhou medical university (Lishui People's Hospital), Lishui, Zhejiang province, 323000, P.R. China
4. Department of Urology, The First Affiliated Hospital of Wenzhou Medical University, Wenzhou, Zhejiang province,325006, P.R. China
5. Department of Respiratory, Rui'an People's Hospital, The Third Affiliated Hospital of the Wenzhou Medical University, Wenzhou, Zhejiang province, 325200, P.R. China
6. Department of Urology, Changhai Hospital, Second Military Medical University, Shanghai, 200433, P.R. China
*These authors (Aidan Xia, Yuming Chen, Jingfeng Chen) equally contributed to this manuscript.
Received 2019-2-12; Accepted 2019-8-5; Published 2019-8-29
Abstract
Background: Few studies focused on the relationship between the albumin-to-alkaline phosphatase ratio (AAPR) and the urologic outcomes in patients with non-metastatic renal cell carcinoma (RCC) following curative surgery. The aim of this study was to evaluate the prognostic value of preoperative AAPR in non-metastatic RCC patients.
Methods: The prognostic value of AAPR was evaluated in a primary cohort with 419 non-metastatic RCC patients following curative radical or partial nephrectomy and then further validated in an independent cohort consisting of 204 patients. A nomogram was developed based on the independent predictors, and its predictive value was assessed.
Results: Kaplan-Meier survival analysis demonstrated that patients with low AAPR levels were significantly associated with worse overall survival (OS) and cancer-specific survival (CSS) compared with patients with high AAPR levels both in two cohorts. Univariate and multivariate analyses revealed that low AAPR was an independent risk factor for OS (HR = 2.745; 95%CI, 1.266-5.953; P = 0.011) and CSS (HR = 3.042; 95%CI, 1.278-7.243; P = 0.012). Moreover, subgroup analysis (Fuhrman grade G1+G2 and Fuhrman grade G3+G4; T1+T2 stage and T3+T4 stage) revealed that low AAPR was also related to worse urological outcomes. Although no significant differences between patients with low AAPR and patients with high AAPR can be observed with regard to CSS under Fuhrman grade G1+G2 (P=0.058) and T1+T2 stage (P=0.318), there was a worse CSS trend in low AAPR patients. The established nomograms for OS and CSS were well calibrated and had moderate discriminative ability (concordance index: 0.821 and 0.839, respectively)
Conclusions: Preoperative AAPR might be an independent prognostic factor in patients with non-metastatic RCC. The ratio should be applied in RCC patients for risk stratification and clinical decision-making.
Keywords: renal cell carcinoma, albumin-to-alkaline phosphatase ratio, prognosis, serum biomarker
Introduction
Renal cell carcinoma (RCC) is one of the most common cancers in urology, accounting for more than 90% of all kidney cancers [1-3]. The global incidence of RCC has increased by approximately 2% during the last two decades [2]. There were roughly 27,400 new cases and 117,000 kidney-cancer related deaths worldwide in 2008 [4]. In Asia, the number of cases of kidney cancer per 100,000 person years in Japan, Singapore, Hong Kong, Shanghai, China, and India were 6.5, 4.3, 3.8, 2.9, and 2.0, respectively, with Japan having the highest incidence [5]. At present, surgical resection remains the most effective therapy for clinically localized disease [2]. Nevertheless, there remain approximately 30% of patients who will inevitably suffer from local or distant recurrence following curative nephrectomy [2]. Therefore, an increasing number of researchers have paid attention to seeking useful pre- and post-operative predictors to categorize patients with worse outcomes at early time-points.
Several prognostic models have been developed and have been well-confirmed in external validation cohorts [6-9]. However, the prognostic value of these models can be potentially improved because none of them has been applied in clinical practice because of the time-consuming and expensive nature of assays, lack of standardization, and non-reproducibility [10]. Therefore, a new potential prognostic factor should be developed that is cheap and easily detected in a standardized manner. At the moment, some biomarkers from blood have been established, including the AST/ALT (De Ritis) ratio, the neutrophil-to-lymphocyte ratio, and the prognostic nutritional index [10-12], although their results remain inconsistent and controversial. Recently, the albumin-to-alkaline phosphate ratio (AAPR), a novel prognostic factor, has been demonstrated to be significantly associated with poorer urologic outcomes for hepatocellular carcinoma (HCC), metastatic nasopharyngeal carcinoma, and upper tract urothelial carcinoma (UTUC) [13-17]. However, it has not yet been studied in non-metastatic RCC patients following curative nephrectomy. Therefore, the aim of this study was to evaluate the potential prognostic impact of preoperative AAPR in patients with non-metastatic disease.
Materials and Methods
Patients. This retrospective study included 803 consecutive patients diagnosed with non-metastatic RCC (pathological T1-4N0M0) between January 2004 and July 2014 at the Urologic Department of The First Affiliated Hospital of Wenzhou Medical University, China. All underwent curative radical or partial nephrectomy and none received neo-adjuvant chemotherapy before surgery. The exclusion criteria were patients who: (1) underwent kidney transplantation before surgery or had only 1 kidney or hemodialysis therapy (n = 29); (2) had any history of other cancers or bilateral RCC or prior surgery for RCC (n = 51); (3) had liver diseases, including cirrhosis and chronic hepatitis B, that could affect AAPR levels (n = 42); and (4) had incomplete preoperative medical information on albumin and ALP (n = 21) or follow-up (n = 37) (Figure 1A). Finally, a total of 623 patients were enrolled in this study, among which 419 patients were randomly assigned to the primary cohort and 204 patients were assigned to the validation cohort. This study was approved by the ethics committee of The First Affiliated Hospital of Wenzhou Medical University. The informed consent was not required for this study.
(A) The patient selection flowchart. (B) The ROC curve of AAPR for OS in primary cohort. (C) The ROC curve of AAPR for OS in validation cohort.
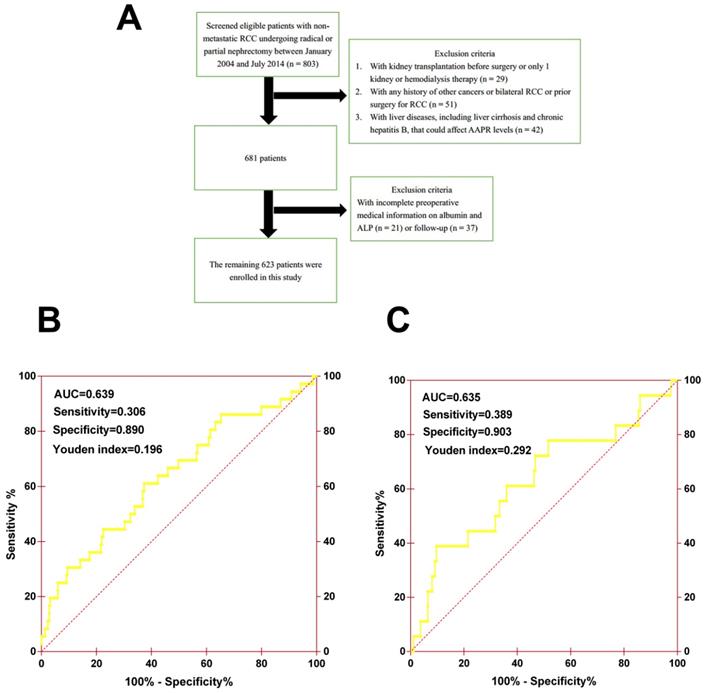
Methods. Clinicopathological records and data on laboratory assessments, including albumin (ALB) and alkaline phosphatase (ALP), were collected and retrospectively analyzed. The cutoff value of AAPR was determined by performing an ROC analysis for evaluating OS, and 0.39 was chosen as the final cutoff value because it had the maximum Youden index value (sensitivity: 30.6%; specificity: 89.0%; Youden index: 0.196) (Figure 1B). Patients were then generally followed up every 3 to 6 months for the first 2 years and annually after surgery for blood and urine tests, cystoscopy, and image examination. Information on death was obtained from outpatient medical records, telephone interviews, or the patient's social security death index.
Statistical analysis. Statistical analyses were performed using the SPSS software package version 25.0 (IBM, Armonk, NY), and two-sided P value < 0.05 were considered significant. The Pearson chi-square test was used to evaluate the association of clinicopathologic characteristics with AAPR. The overall survival (OS) rates and cancer-specific survival (CSS) rates were estimated using the Kaplan-Meier method in primary cohort and validated cohort. To determine the independent prognostic factors, univariate analysis and multivariate Cox regression analysis were performed. Variables with P < 0.05 in the univariate analysis were included in the subsequent multivariate model. Nomograms for probability of OS and CSS were established based on the results of the multivariate analysis using the R software (Version 3.6.0) with the packages rms, Hmisc, and ggplots. Calibration plot, concordance index (c-index), and ROC analysis were applied to evaluate the performance of nomograms.
Results
Patient characteristics
The patients' clinicopathological characteristics of both two cohorts are summarized in Table 1. In the primary cohort, there were 266 (63.5%) males and 153 (36.5%) females. The mean age was 61.0±12.9 years and 173 (41.3%) patients were 65 years of age or older. The median follow-up duration was 50.0 (30.4 - 83.0) months. During follow up, a total of 36 (8.6%) patients died, among which 27 (6.4%) patients died of cancer-specific causes. In the validation cohort, there were 127(62.3%) males and 84 (41.2%) patients were 65 years of age or older. The mean age was 62.4±11.7 years. The median follow-up duration was 50.2 (29.8 - 83.1) months. During follow up, a total of 18 (8.8%) patients died, among which 10 (4.9%) patients died of cancer-specific causes.
Characteristics of primary and validation cohorts
| Variables | Primary cohort (n=419) | Validation cohort (n=204) |
|---|---|---|
| Age, years | ||
| >65 | 173(41.3%) | 84(41.2%) |
| ≤65 | 246(58.7%) | 120(58.8%) |
| Gender | ||
| Male | 266(63.5%) | 127(62.3%) |
| Female | 153(36.5%) | 77(37.7%) |
| ASA grade | ||
| ≥3 | 23(5.5%) | 17(8.3%) |
| <3 | 396(94.5%) | 187(91.7%) |
| BMI, kg/m2 | ||
| ≥25 | 92(22.0%) | 57(27.9%) |
| <25 | 327(78.0%) | 147(72.1%) |
| DM | ||
| Yes | 143(34.1%) | 67(32.8%) |
| No | 276(65.9%) | 137(67.2%) |
| Hypertension | ||
| Yes | 173(41.3%) | 85(41.7%) |
| No | 246(58.7%) | 119(58.3%) |
| Anemia | ||
| Yes | 65(15.5%) | 25(12.3%) |
| No | 354(84.5%) | 179(87.7%) |
| Surgical approach | ||
| Partial nephrectomy | 87(20.8%) | 45(22.1%) |
| Radical nephrectomy | 332(79.2%) | 159(77.9%) |
| CKD stage | ||
| CKD 1 | 292(69.7%) | 134(65.7%) |
| CKD 2 | 99(23.6%) | 60(29.4%) |
| CKD 3 | 17(4.1%) | 8(3.9%) |
| CKD 4 | 4(1.0%) | 0 |
| CKD 5 | 7(1.6%) | 2(1.0%) |
| Pathologic stage | ||
| pT1 | 322(76.8%) | 161(78.9%) |
| pT2 | 46(11.0%) | 25(12.3%) |
| pT3 | 45(10.7%) | 15(7.4%) |
| pT4 | 6(1.5%) | 3(1.4%) |
| Fuhrman grade | ||
| 1 | 132(31.5%) | 68(33.3%) |
| 2 | 177(42.2%) | 87(42.6%) |
| 3 | 95(22.7%) | 45 (22.1%) |
| 4 | 15(3.6%) | 4(2.0%) |
| Histologic subtype | ||
| Clear cell | 359(85.7%) | 186(91.2%) |
| Non-clear cell | 60(14.3%) | 18(9.8%) |
| Tumor necrosis | ||
| Yes | 17(4.1%) | 4(2.0%) |
| No | 402 (95.9%) | 200(98.0%) |
| Tumor size, cm | ||
| ≥7 | 73(17.4%) | 35(17.2%) |
| <7 | 346(82.6%) | 169(82.8%) |
| Albumin, g/l | ||
| ≤35 | 48(11.5%) | 29(14.2%) |
| >35 | 371(88.5%) | 175(85.8%) |
| ALP, U/l | ||
| ≥125 | 21(5.0%) | 11(5.4%) |
| <125 | 398(95.0%) | 193(94.6%) |
| AAPR | ||
| <0.39 | 54(12.9%) | 25(12.3%) |
| ≥0.39 | 365(87.1%) | 179(87.7%) |
| Globulin, g/l | ||
| ≤25 | 75(17.9%) | 41(20.1%) |
| >25 | 344(82.1%) | 163(79.9%) |
| ALT, IU/l | ||
| ≥40 | 51(12.2%) | 30(14.7%) |
| <40 | 368(87.8%) | 174(85.3%) |
| AST, IU/l | ||
| ≥35 | 63(15.0%) | 35(17.2%) |
| <35 | 356(85.0%) | 169(82.8%) |
Abbreviations: DM, diabetes mellitus; CKD stage, chronic kidney disease stage; ALP, alkaline phosphatase; AAPR, albumin-to-alkaline phosphatase; ALT, alanine aminotransaminase; AST, aspartate aminotransaminase.
Associations between AAPR and clinicopathological characteristics of the primary cohort
The clinicopathological characteristics of the cohort according to the preoperative values of AAPR are shown in Table 2. The median value of AAPR was 0.58 (0.46-0.71). Higher serum ALT (P=0.004) and AST (P=0.016) levels and anemia (P=0.001) can be more commonly observed in patients with low AAPR. Additionally, low AAPR patients were older (P=0.004) than high AAPR patients.
Associations between AAPR and clinicopathological characteristics in primary cohort.
| Variables | AAPR<0.39 (n=54) | AAPR≥0.39 (n=365) | P value |
|---|---|---|---|
| Age, years | 0.004* | ||
| >65 | 32(59.3%) | 141(38.6%) | |
| ≤65 | 22(40.7%) | 224(61.4%) | |
| Gender | 0.320 | ||
| Male | 31(57.4%) | 130(35.6%) | |
| Female | 23(42.6%) | 235(64.4%) | |
| ASA grade | 0.193 | ||
| ≥3 | 5(9.3%) | 18(4.9%) | |
| <3 | 49(90.7%) | 347(95.1%) | |
| BMI, kg/m2 | 0.087 | ||
| ≥25 | 7(13.0%) | 85(23.3%) | |
| <25 | 47(87.0%) | 280(76.7%) | |
| DM | 0.160 | ||
| Yes | 23(42.6%) | 120(32.9%) | |
| No | 31(57.4%) | 245(67.1%) | |
| Hypertension | 0.091 | ||
| Yes | 28(51.9%) | 145(39.7%) | |
| No | 26(48.1%) | 220(60.3%) | |
| Anemia | 0.001* | ||
| Yes | 17(31.5%) | 48(13.2%) | |
| No | 37(68.5%) | 317(86.8%) | |
| Surgical approach | 0.663 | ||
| Partial nephrectomy | 10(18.5%) | 77(21.1%) | |
| Radical nephrectomy | 44(81.5%) | 288(78.9%) | |
| CKD stage | 0.025* | ||
| CKD 1 | 37(68.5%) | 255(69.9%) | |
| CKD 2 | 9(16.7%) | 90(24.7%) | |
| CKD 3 | 3(5.6%) | 14(3.8%) | |
| CKD 4 | 1(1.8%) | 3(0.8%) | |
| CKD 5 | 4(7.4%) | 3(0.8%) | |
| Pathologic stage | 0.306 | ||
| pT1 | 37(68.5%) | 285(78.1%) | |
| pT2 | 6(11.1%) | 40(11.0%) | |
| pT3 | 10(18.5%) | 35(9.6%) | |
| pT4 | 1(1.9%) | 5(1.3%) | |
| Fuhrman grade | 0.531 | ||
| 1 | 17(31.5%) | 115(31.5%) | |
| 2 | 21(38.9%) | 156(42.8%) | |
| 3 | 12(22.2%) | 83(22.7%) | |
| 4 | 4(7.4%) | 11(3.0%) | |
| Histologic subtype | 0.911 | ||
| Clear cell | 46(85.2%) | 313(85.8%) | |
| Non-clear cell | 8(14.8%) | 52(14.2%) | |
| Tumor necrosis | 0.888 | ||
| Yes | 2(3.7%) | 15(4.1%) | |
| No | 52(96.3%) | 350(95.9%) | |
| Tumor size, cm | 0.820 | ||
| ≥7 | 10(18.5%) | 63(17.3%) | |
| <7 | 44(81.5%) | 302(82.7%) | |
| Globulin, g/l | 0.163 | ||
| ≤25 | 6(11.1%) | 69(18.9%) | |
| >25 | 48(88.9%) | 296(81.1%) | |
| ALT, IU/l | 0.004* | ||
| ≥40 | 13(24.1%) | 38(10.4%) | |
| <40 | 41(75.9%) | 327(89.6%) | |
| AST, IU/l | 0.016* | ||
| ≥35 | 14(25.9%) | 49(13.4%) | |
| <35 | 40(74.1%) | 316(86.6%) |
*Statistically significant
Prognostic significance of AAPR
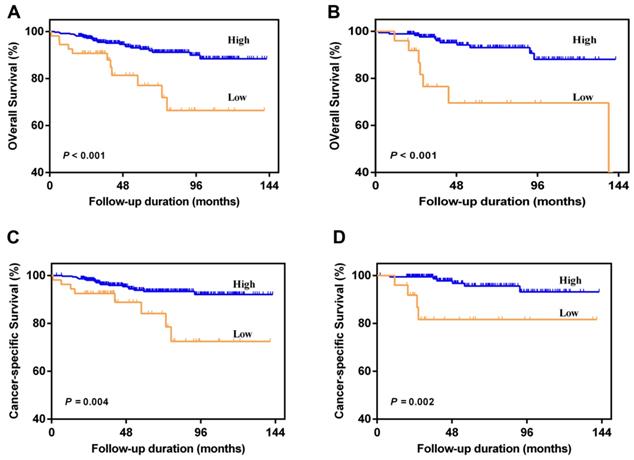
Patients with AAPR < 0.39 were significantly associated with worse OS and CSS compared with patients with AAPR ≥ 0.39 in primary cohort (Figure 2A and 2B) and validation cohort (Figure 2C and 2D). In primary cohort, the 5-year OS and CSS rates were 92.5% and 93.3% in patients with AAPR ≥ 0.39, and 77.1% and 84.1% in patients with AAPR < 0.39, respectively. In validation cohort, the 5-year OS and CSS rates were 93.1% and 95.6% in patients with AAPR ≥ 0.39, and 69.6% and 81.6% in patients with AAPR < 0.39, respectively.
Kaplan-Meier survival curves for OS and CSS of patients with non-metastatic RCC according to AAPR levels. Patients with low AAPR were associated with worse OS and CSS in primary cohort (A and C) and validation cohort (B and D).
In the univariate analysis, AAPR < 0.39 was significantly associated with poorer OS and CSS. Variables with P < 0.05 in the univariate analysis were included in the subsequent multivariate analysis and the results revealed that AAPR < 0.39 was identified as an independent risk factor of OS (HR = 2.745; 95%CI, 1.266-5.953; P = 0.011) and CSS (HR = 3.042; 95%CI, 1.278-7.243; P = 0.012) (Table 3). Other variables were also determined as independent predictors, including old age, higher pathologic T stage, and tumor necrosis.
Multivariable regression analysis of clinicopathological parameters for the prediction of OS and CSS in primary cohort
| Variables | OS | CSS | ||
|---|---|---|---|---|
| HR(95%CI) | P value | HR(95%CI) | P value | |
| Age (>65 vs ≤65 years) | 2.862(1.321-6.201) | 0.008* | 2.834(1.190-6.746) | 0.019* |
| ASA grade (≥3 vs <3) | 2.413(0.911-6.390) | 0.076 | 1.623(0.481-5.479) | 0.436 |
| BMI (≥25 vs <25) | 0.435(0.099-1.900) | 0.268 | - | - |
| Anemia (Yes vs No) | 1.418(0.625-3.218) | 0.404 | 1.464(0.566-3.788) | 0.432 |
| CKD stage | ||||
| CKD 1 | 1.000 | 1.000 | ||
| CKD 2-3 vs CKD 1 | 1.555(0.440-5.493) | 0.493 | - | - |
| CKD 4-5 vs CKD 1 | 3.052(0.744-12.508) | 0.121 | ||
| Pathologic T stage | ||||
| pT1 | 1.000 | 1.000 | 1.000 | 1.000 |
| pT2 vs pT1 | 1.771(0.557-5.633) | 0.333 | 1.559(0.432-5.630) | 0.498 |
| pT3 vs pT1 | 3.153(1.227-8.101) | 0.017* | 4.258(1.487-12.192) | 0.007* |
| pT4 vs pT1 | 32.425(6.683-157.335) | <0.001* | 32.617(5.226-203.590) | <0.001* |
| Fuhrman grade (≥3 vs <3) | 1.742(0.815-3.723) | 0.152 | 2.403(0.996-5.798) | 0.051 |
| Tumor necrosis (Yes vs No) | 2.687(0.885-8.159) | 0.081 | 3.672(1.166-11.560) | 0.026* |
| Tumor size (≥7 vs <7) | 2.549(0.965-6.736) | 0.059 | 2.619(0.973-7.054) | 0.057 |
| Albumin (<35 vs ≥35) | 1.684(0.741-3.828) | 0.213 | 1.417(0.520-3.861) | 0.495 |
| AAPR (<0.39 vs ≥0.39) | 2.745(1.266-5.953) | 0.011* | 3.042(1.278-7.243) | 0.012* |
*Statistically significant
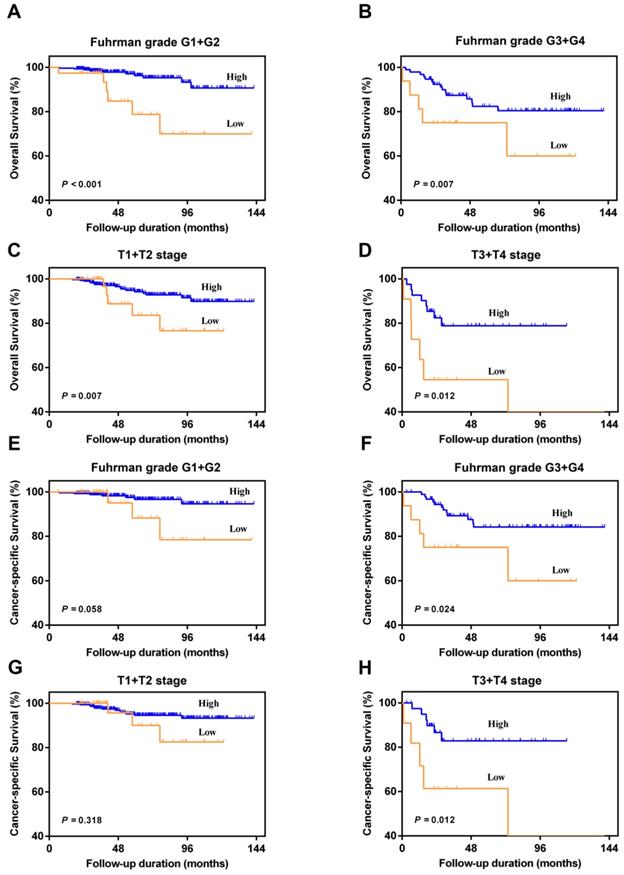
In the subgroup analysis, we divided patients into Fuhrman grade G1+G2 group and Fuhrman grade G3+G4 group, or T1+T2 stage group and T3+T4 stage group. The results showed that AAPR < 0.39 was also significantly associated with poorer urologic outcomes among four different subgroups (P < 0.05) (Figure 3). Although no significant differences between patients with AAPR < 0.39 and patients with AAPR ≥0.39 can be observed with regard to CSS under Fuhrman grade G1+G2 (P=0.058) and T1+T2 stage (P=0.318) (Figure 3E and 3G), there was a worse CSS trend in low AAPR patients.
Kaplan-Meier curves for OS and CSS according to AAPR in the Fuhrman grade G1+G2 (A and E), Fuhrman grade G3+G4 (B and F), pathological T1+T2 stage (C and G), and pathological T3+T4 stage (D and H).
The nomogram and its performance
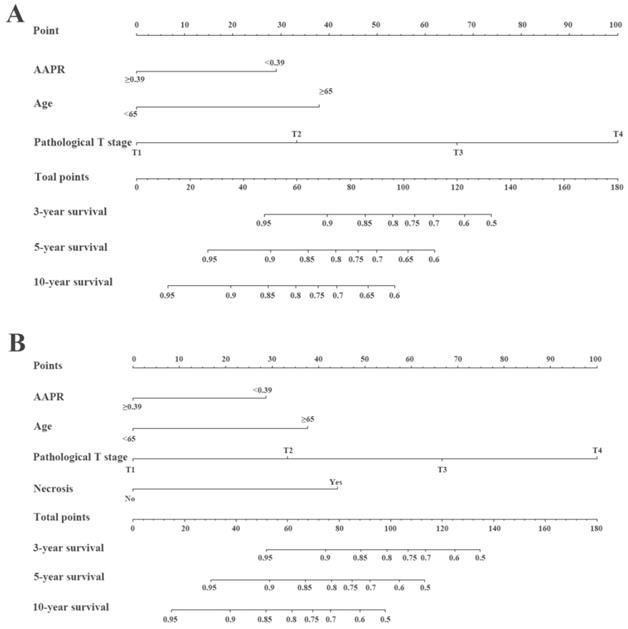
The prognostic nomograms for OS (Figure 4A) and CSS (Figure 4B) were depicted by independent indicators in the multivariate analysis. Each predictor in the nomogram was assigned a score (top scale). Thereafter, the sum of these scores implied the probability of 3-, 5-, and 10-year OS or CSS (bottom scale). The c-indexes for the nomogram of OS and CSS were 0.821 (95%CI, 0.750-0.892) and 0.839 (95%CI, 0.7557-0.922), respectively (Table 4), indicating moderate discriminative ability of these two models (low discriminative ability: 0.50-0.70; moderate discriminative ability: 0.71-0.90; high discriminative ability: 0.90-1). The calibration plots of the nomograms were developed (Figure 5), which demonstrated that the nomograms were well-calibrated. As shown in Figure 6 and Table 5, the AUC of the nomogram for OS was 0.806 (95% CI, 0.728-0.884), with the sensitivity, specificity, and Youden index of 66.67%, 84.60%, and 0.513, respectively. The AUC of the nomogram for CSS was 0.811 (95% CI, 0.729-0.892), with the sensitivity, specificity, and Youden index of 74.07%, 79.85%, and 0.539, respectively.
Construction of preoperative nomogram with AAPR and other significant factors that predicted the probability of non-metastatic RCC for OS (A) and CSS (B). To use the nomogram, the value of individual patients with RCC is located on each variable axis, and a line is depicted upward to determine the number of points received for each variable value. Subsequently, the sum of these numbers is located on the Total Point axis, and a line is drawn downward to the survival axes to determine the likelihood of 3-, 5-, and 10-year survival of OS or CSS.
Predictive ability comparison of models for OS and CSS with 1000 bootstraps
| Model | c-index | 95%CI |
|---|---|---|
| Nomogram for OS | ||
| Model A = pT+Age+AAPR | 0.821 | 0.750-0.892 |
| Model B = pT+Age | 0.772 | 0.690-0.854 |
| Nomogram for CSS | ||
| Model C = pT+Age+AAPR+Necrosis | 0.839 | 0.756-0.922 |
| Model D = pT+Age +Necrosis | 0.809 | 0.723-0.895 |
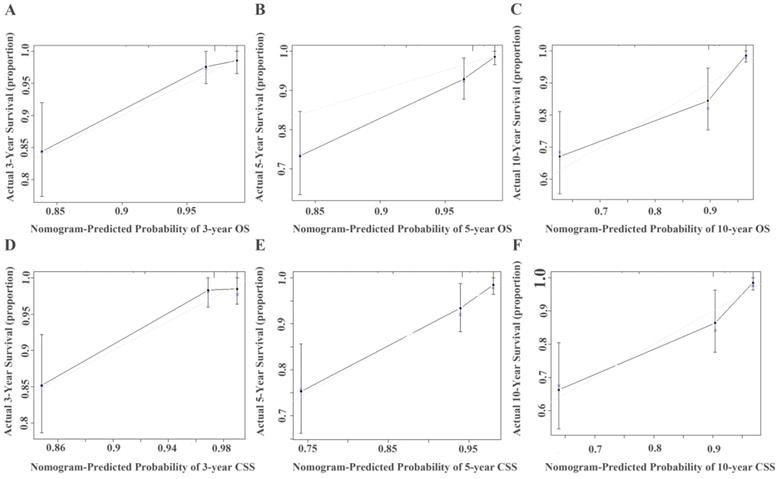
Calibration curve for predicting 3-, 5-, and 10-year survival of OS (A, B, and C) or CSS (D, E, and F) in non-metastatic RCC patients. The actual OS or CSS rates are plotted on the y-axis and nomogram-predicted OS or CSS rates are plotted on the x-axis.
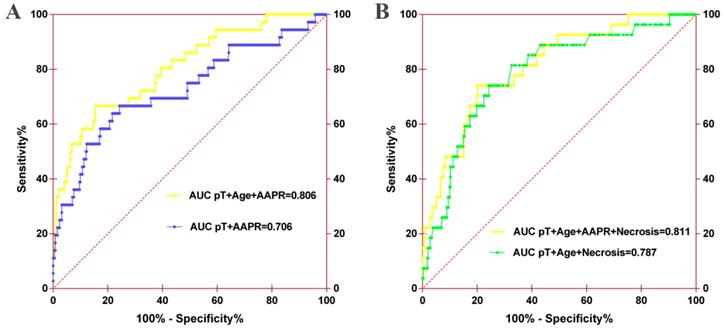
ROC analysis of the nomogram model for OS (A) or CSS (B). For OS, the AUC of the nomogram model was 0.706 when assessed by pT and age, which increased to 0.806 when the AAPR was added. For CSS, the AUC of the nomogram model increased from 0.787 to 0.811 when AAPR was added.
Predictive ability comparison of models for OS and CSS with ROC analysis
| Model | Sensitivity (%) | Specificity (%) | Accuracy (%) | Youden index | Positive predictive value (%) | Negative predictive value (%) | Positive likelihood ratio | Negative likelihood ratio |
|---|---|---|---|---|---|---|---|---|
| Nomogram for OS | ||||||||
| Model A = pT+Age+AAPR | 66.67 | 84.60 | 83.05 | 0.513 | 74.20 | 75.83 | 4.33 | 0.39 |
| Model B = pT+Age | 66.67 | 74.15 | 73.51 | 0.408 | 70.56 | 75.80 | 2.58 | 0.45 |
| Nomogram for CSS | ||||||||
| Model C = pT+Age+AAPR+Necrosis | 74.07 | 79.85 | 79.47 | 0.539 | 73.57 | 80.79 | 3.68 | 0.32 |
| Model D = pT+Age +Necrosis | 74.07 | 75.77 | 75.66 | 0.498 | 70.88 | 80.71 | 3.06 | 0.34 |
Evaluation of the predictive ability of AAPR for OS and CSS
By incorporating AAPR into the developed models, the c-indexes of the nomograms for OS and CSS increased from 0.772 (95% CI, 0.690-0.854) to 0.821 (95% CI, 0.750-0.892), and from 0.809 (95% CI, 0.723-0.895) to 0.839 (95%CI, 0.756-0.922), respectively (Table 4).
Furthermore, AUC comparison demonstrated that the AUC of the nomogram for OS increased from 0.706 (95% CI, 0.601-0.812) to 0.806 (95% CI, 0.728-0.884) when AAPR was incorporated into the model (Figure 6A). The AUC of the nomogram for CSS also improved from 0.787 (95% CI, 0.698-0.875) to 0.811 (95% CI, 0.729-0.892) when AAPR was added (Figure 6B). Other predictive parameters of nomograms for OS and CSS also were improved (Table 5). These findings revealed that AAPR could be a useful indicator of urologic outcomes in patients with non-metastatic RCC, which should be applied in patients with RCC for risk stratification and clinical decision-making.
Discussion
Various risk assessment models have been previously developed to predict the prognosis in patients with or without RCC patients after surgery, including the Leibovich prognosis score [7], Mayo Clinic SSIGN (stage, size, grade, and necrosis) [18], and UISS (UCLA Integrated Staging System) [19]. Several studies have identified several immunohistochemical biomarkers, genomic approaches, and nomograms as significantly independent factors for postoperative survival in RCC patients [20, 21]. In the present study, AAPR, a novel risk factor, was introduced and its prognostic value was evaluated and validated in this study. The cutoff value of AAPR was determined to be 0.39 by performing ROC analysis, and AAPR < 0.39 was found to be statistically correlated with old age, anemia, and higher pathological T stage. Subsequently, AAPR < 0.39 was revealed to be associated with poorer OS and CSS in primary cohort and confirmed in validation cohort. Furthermore, decreased AAPR was identified as an independent risk predictor for OS and CSS in non-RCC patients after surgery according to multivariate analysis. The predictive abilities of the developed nomograms also increased when AAPR were incorporated into these models.
AAPR was first reported by Anthony et al. in 2015. They found that AAPR was an independent prognostic indicator for hepatocellular carcinoma (HCC) with the highest c-index and χ2 among other liver biochemical parameters [15]. Researchers then demonstrated that AAPR was also an independent predictor of advanced HCC and metastatic nasopharyngeal carcinoma, and its predictive ability was significantly better than that of ALB or ALP alone [13, 14]. Recently, Tan et al. reported that AAPR was significantly associated with worse survival outcomes in UTUC with relative high AUC [17]. With respect to its superior predictive accuracy for cancer outcomes and absence of studies evaluating the prognostic role in RCC patients, we performed this study and found the same results as mentioned previously.
It remains unclear why lower AAPR increases the risk of tumor relapses and mortality; however there is one possibility that should be addressed: AAPR is calculated from serum ALB concentration divided by serum ALP concentration, indicating that nutritional deficiency and systemic inflammatory response might be involved in the development and progress of RCC when AAPR performed for its prognostic impact on tumor recurrence and metastasis. ALB is specifically synthesized by liver, where it is not only an important nutritional index, but also it is associated with systemic immunological response to inflammatory or tumor [22]. ALB has the ability to stabilize cell growth and proliferation, modulate immune reactions, and exert antioxidant effects against carcinogens [23]. As a result, the presence of low ALB or hypoalbuminemia may lead to impairment of immunity and poor anti-cancer responses [24]. Previous studies have reported that ALB was a reliable predictive tool in various cancers, including HCC, RCC, and prostate cancer [25-27]. ALP is a hydrolase enzyme that is primarily located in the kidney, liver, and bone. Serum ALP levels commonly increase in patients with HCC, kidney disease, and bone metastasis [13]. Furthermore, ALP was also identified as independent risk factor in various cancers, including HCC, nasopharyngeal carcinoma, and RCC [28-30].
In the present study, the optimal cutoff value was obtained from the maximal Youden index value by performing ROC analysis. This cutoff value, 0.39 for AAPR, was suggested as a superior prognostic level according to HR. Nevertheless, the cutoff-point was not consistent with those from previous studies [15-17, 27] and was close to that of one study [14]. This common problem can also be observed in studies of other prognostic biomarkers, including the platelet-lymphocyte ratio and neutrophil-lymphocyte ratio [31]. Several points are needed to be taken into account: the application in various cancers, cohorts with varying sample sizes, follow-up periods, survival end-points, and assay methods for AAPR, as well as absence of standardized methods to determine optimal cutoff value. Furthermore, although AAPR was still identified as an independent predictor, the proportion of patients with lower AAPR and pathologic T3 and T4 stage in the primary cohort of our study were only 54 (12.9%) and 54 (12.2%), relatively lower than those of other studies. For example, Tan et. al. retrospectively assessed 692 patients with UTUC after surgery in 2003 and 2016. The cutoff value of AAPR was determined to be 0.58 by performing ROC analysis, and they found that lower AAPR was significantly associated with worse prognosis in UTUC patients. However, there were 443 (64.0%) patients with AAPR < 0.58, and 342 (49.4%) patients with pathologic T3 and T4. Therefore, further studies are needed to identify the best cutoff value according to particular cancers.
There were some limitations to this study. First, our study was retrospectively designed in a single institution, possibly giving rise to selection bias. Nevertheless, our department is the largest urologic center with the largest sample size for patients with RCC in the south of Zhejiang Province; therefore, our data were representative and reliable. Second, we were unable to include C-reactive protein due to deficiency in some patients. We included preoperative hemoglobin instead because it is a similar predictor of urologic outcomes. Third, the optimal cutoff for AAPR also requires prospective validation. Fourth, the effects of dynamic changes in AAPR on long-term survival remain to be evaluated. Last, the prognostic value and mechanisms of AAPR are required to be evaluated in further prospective studies and basic researches. Fifth, our results revealed that low AAPR was also related to worse urological outcomes in subgroup analysis; however, Tan et al. found no relationship between AAPR and urologic outcomes in low-grade UTUC patients [17]. Therefore, the impact of AAPR on urologic outcomes in different subgroups remains to be investigated.
Conclusion
To the best of our knowledge, this was first study to assess the prognostic impact of AAPR in non-metastatic RCC patients following curative surgery. AAPR< 0.39 was an independent predictor of worse OS and CSS in RCC patients. Prospective studies and investigation of potential mechanisms regarding the close correlation between lower AAPR and inferior survival outcomes in non-metastatic RCC are required.
Abbreviations
AAPR, albumin-to-alkaline phosphatase ratio; ALB, albumin; ALP, alkaline phosphatase; RCC, renal cell carcinoma; HCC, hepatocellular carcinoma; UTUC, upper tract urothelial carcinoma; OS, overall survival; MFS, metastasis-free survival; CSS, cancer-specific survival; CKD, chronic kidney disease; SSIGN, Stage, Size, Grade and Necrosis Score; UISS, UCLA Integrated Staging System.
Acknowledgements
The authors thank Editage for English language editing. This study was supported by the 2018 Zhejiang medical and health science and technology program (2018KY930), and the 2017 Lishui science and technology planning program (2017GYX14).
Competing Interests
The authors have declared that no competing interest exists.
References
1. Siegel RL, Miller KD, Jemal A. Cancer Statistics, 2017. CA Cancer J Clin. 2017;67:7-30
2. Ljungberg B, Bensalah K, Canfield S. et al. EAU guidelines on renal cell carcinoma: 2014 update. Eur Urol. 2015;67:913-24
3. Capitanio U, Montorsi F. Renal cancer. Lancet. 2016;387:894-906
4. Chiong E, Tay MH, Tan MH. et al. Management of kidney cancer in Asia: resource-stratified guidelines from the Asian Oncology Summit 2012. Lancet Oncol. 2012;13:e482-e91
5. Mathew A, Devesa SS, Fraumeni JF Jr. et al. Global increases in kidney cancer incidence, 1973-1992. Eur J Cancer Prev. 2002;11:171-8
6. Leibovich BC, Blute ML, Cheville JC. et al. Prediction of progression after radical nephrectomy for patients with clear cell renal cell carcinoma: a stratification tool for prospective clinical trials. Cancer. 2003;97:1663-71
7. Leibovich BC, Han KR, Bui MH. et al. Scoring algorithm to predict survival after nephrectomy and immunotherapy in patients with metastatic renal cell carcinoma: a stratification tool for prospective clinical trials. Cancer. 2003;98:2566-75
8. Pichler M, Hutterer GC, Chromecki TF. et al. External validation of the Leibovich prognosis score for nonmetastatic clear cell renal cell carcinoma at a single European center applying routine pathology. J Urol. 2011;186:1773-7
9. Zhou W, Huang C, Yuan N. Prognostic nomograms based on log odds of positive lymph nodes for patients with renal cell carcinoma: A retrospective cohort study. Int J Surg. 2018;60:28-40
10. Bezan A, Mrsic E, Krieger D. et al. The Preoperative AST/ALT (De Ritis) Ratio Represents a Poor Prognostic Factor in a Cohort of Patients with Nonmetastatic Renal Cell Carcinoma. J Urol. 2015;194:30-5
11. Jeon HG, Choi DK, Sung HH. et al. Preoperative Prognostic Nutritional Index is a Significant Predictor of Survival in Renal Cell Carcinoma Patients Undergoing Nephrectomy. Ann Surg Oncol. 2016;23:321-7
12. Boissier R, Campagna J, Branger N. et al. The prognostic value of the neutrophil-lymphocyte ratio in renal oncology: A review. Urol Oncol. 2017;35:135-41
13. Nie M, Sun P, Chen C. et al. Albumin-to-Alkaline Phosphatase Ratio: A Novel Prognostic Index of Overall Survival in Cisplatin-based Chemotherapy-treated Patients with Metastatic Nasopharyngeal Carcinoma. J Cancer. 2017;8:809-15
14. Cai X, Chen Z, Chen J. et al. Albumin-to-Alkaline Phosphatase Ratio as an Independent Prognostic Factor for Overall Survival of Advanced Hepatocellular Carcinoma Patients without Receiving Standard Anti-Cancer Therapies. J Cancer. 2018;9:189-97
15. Chan AW, Chan SL, Mo FK. et al. Albumin-to-alkaline phosphatase ratio: a novel prognostic index for hepatocellular carcinoma. Dis Markers. 2015;2015:564057
16. Chen ZH, Zhang XP, Cai XR. et al. The Predictive Value of Albumin-to-Alkaline Phosphatase Ratio for Overall Survival of Hepatocellular Carcinoma Patients Treated with Trans-Catheter Arterial Chemoembolization Therapy. J Cancer. 2018;9:3467-78
17. Tan P, Xie N, Ai J. et al. The prognostic significance of Albumin-to-Alkaline Phosphatase Ratio in upper tract urothelial carcinoma. Sci Rep. 2018;8:12311
18. Frank I, Blute ML, Cheville JC. et al. An outcome prediction model for patients with clear cell renal cell carcinoma treated with radical nephrectomy based on tumor stage, size, grade and necrosis: the SSIGN score. J Urol. 2002;168:2395-400
19. Zisman A, Pantuck AJ, Dorey F. et al. Improved prognostication of renal cell carcinoma using an integrated staging system. J Clin Oncol. 2001;19:1649-57
20. Cindolo L, Patard JJ, Chiodini P. et al. Comparison of predictive accuracy of four prognostic models for nonmetastatic renal cell carcinoma after nephrectomy: a multicenter European study. Cancer. 2005;104:1362-71
21. Karakiewicz PI, Briganti A, Chun FK. et al. Multi-institutional validation of a new renal cancer-specific survival nomogram. J Clin Oncol. 2007;25:1316-22
22. Tanriverdi O. A discussion of serum albumin level in advanced-stage hepatocellular carcinoma: a medical oncologist's perspective. Med Oncol. 2014;31:282
23. Arroyo V, Garcia-Martinez R, Salvatella X. Human serum albumin, systemic inflammation, and cirrhosis. J Hepatol. 2014;61:396-407
24. Ahluwalia N. Aging, nutrition and immune function. J Nutr Health Aging. 2004;8:2-6
25. Chi KN, Kheoh T, Ryan CJ. et al. A prognostic index model for predicting overall survival in patients with metastatic castration-resistant prostate cancer treated with abiraterone acetate after docetaxel. Ann Oncol. 2016;27:454-60
26. Chen Z, Shao Y, Fan M. et al. Prognostic significance of preoperative C-reactive protein: albumin ratio in patients with clear cell renal cell carcinoma. Int J Clin Exp Pathol. 2015;8:14893-900
27. Ho SY, Hsu CY, Liu PH. et al. Albumin-bilirubin (ALBI) grade-based nomogram to predict tumor recurrence in patients with hepatocellular carcinoma. Eur J Surg On. 2019;45:776-81
28. Yu MC, Chan KM, Lee CF. et al. Alkaline phosphatase: does it have a role in predicting hepatocellular carcinoma recurrence? J Gastrointest Sur. 2011;15:1440-9
29. Jin Y, Yuan MQ, Chen JQ. et al. Serum alkaline phosphatase predicts survival outcomes in patients with skeletal metastatic nasopharyngeal carcinoma. Clinics (Sao Paulo). 2015;70:264-72
30. Chen XY, Lan M, Zhou Y. et al. Risk factors for bone metastasis from renal cell cancer. J Bone Oncol. 2017;9:29-33
31. Toyokawa T, Kubo N, Tamura T. et al. The pretreatment Controlling Nutritional Status (CONUT) score is an independent prognostic factor in patients with resectable thoracic esophageal squamous cell carcinoma: results from a retrospective study. BMC cancer. 2016;16:722
Author contact
Corresponding authors: Dr Xiaomin Gao, Department of Urology, Changhai Hospital, Second Military Medical University, 168 Changhai Road, Yangpu District, Shanghai 200433, P.R. China; And Dr Lianmin Bao, Department of Respiratory, Rui'an People's Hospital, The Third Affiliated Hospital of the Wenzhou Medical University, Wenzhou, Zhejiang, 325200, P.R. China; And Dr Yue Pan, Department of Urology, The First Affiliated Hospital of Wenzhou Medical University, Wenzhou, Zhejiang province, 325006, P.R. China. E-mail: doctorgaoxiaominedu.cn or baolianmin1990com or pxy_628com

 Global reach, higher impact
Global reach, higher impact