Impact Factor ISSN: 1837-9664
J Cancer 2020; 11(6):1436-1445. doi:10.7150/jca.33720 This issue Cite
Research Paper
Triple-high expression of phosphatase and tensin homolog (PTEN), estrogen receptor (ER) and progesterone receptor (PR) may predict favorable prognosis for patients with Type I endometrial carcinoma
1. Department of Pathology, Dongguan Hospital Affiliated to Medical College of Jinan University, The Fifth People's Hospital of Dongguan, Dongguan 523905, China
2. Dongguan Key Laboratory of Medical Bioactive Molecular Developmental and Translational Research, Guangdong Provincial Key Laboratory of Medical Molecular Diagnostics, Guangdong Medical University, Dongguan, Guangdong 523808, China
3. Clinical Experimental Center, Jiangmen Central Hospital, Affiliated Jiangmen Hospital of Sun Yat-sen University, Jiangmen 529030, China.
Received 2019-1-31; Accepted 2019-11-19; Published 2020-1-13
Abstract
Endometrial carcinoma (EC) is the most common malignant tumors in female derived from the endometrial epithelium. Several previous studies have described estrogen receptors (ER), progesterone Receptor (PR) and phosphatase and tensin homolog (PTEN) are associated with clinicopathological factors and prognosis in EC patients. However, during EC patients follow-up, we found that some EC patients with down-regulation of PTEN, but up-regulation of ER or PR , and some EC patients with down-regulation of ER or PR, but up-regulation of PTEN also had a poor prognosis. Therefore, to reveal the prognosis of EC patients with different phenotypes based on PTEN, ER and PR expression, 120 cases formalin-fixed paraffin-embedded EC tissues and 543 cases uterine corpus endometrial carcinoma (UCEC) patients from the cancer genome atlas (TCGA) UCEC datasets were analyzed. Results showed that EC tissues can be classified to PTENLERLPRL, PTENHERLPRL, PTENHERHPRH, PTENLERHPRH, PTENHERHPRL, PTENHERLPRH, and PTENLERHPRL phenotypes basing on IHC analysis. Additionally, EC patients with PTENLERLPRL showed high malignancy, while patients with PTENHERHPRH showed low malignancy. Therefore, combined detection of PTEN, ER, PR may help identify a small subset of EC with more aggressive behavior and may aid in risk stratification.
Keywords: Endometrial carcinoma, estrogen receptors, progesterone receptor, phosphatase and tensin homolog, prognosis
Introduction
Endometrial carcinoma (EC) is the most common malignant tumors in female derived from the endometrial epithelium. Recently, the EC incidence is increased, while the 5-year survival rate is decreased [1]. The etiology of EC is not yet clear. Experimental and epidemiologic evidence showed that the risk factors of EC including BMI≥25kg/m2, hypertension, diabetes, smoking, taking tamoxifen, family history of cancer and non-pregnant women[2, 3]. EC can be broadly classified into two types. Majority (~80%) of EC patients are of Type I endometrioid histology, up to 15% are Type II EC patients, primarily serous carcinomas [3-5]. Most of Type I EC patients present with low-grade and early-stage disease and have a favorable prognosis. According to the previous study, 74-91% of International Federation of Gynecology and Obstetrics (FIGO) stages I-II type I EC patients have a 5-year OS time [6]. However, 10-20% of early stages I-II and 50-70% of advanced stage III-IV type I EC patients will recur after primary treatment [7]. Previous studies have revealed that clinicopathological parameters such as histological grade, FIGO clinical stage, myometrial tumor invasion, tumor size, lymph node metastasis, lymphovascular space invasion, and the patients' age and race has prognostic effect in type I EC patients[6, 8, 9]. However, these factors are usually obtained postoperatively and have proven to be insufficient to predict recurrence and estimate survival time. Therefore, it is necessary to identify more effective prognostic predictors to identify preoperative high-risk type I EC patients.
Several previous studies have described estrogen receptors (ER), progesterone Receptor (PR) and phosphatase and tensin homolog (PTEN) are associated with clinicopathological factors and prognosis in EC patients [10-16]. Both of ER and PR are members of the nuclear receptor family that release relevant related ligand-activated transcription factors to regulate cell gene expression through activation and suppression of transcription [17-19]. Low expression or deletion of ER and PR associated with increased malignancy, invasion, and non-hormone-dependent tumor transformation [20]. The absence of PR and ER indicates poor prognosis, poor histologic type, higher histological grade, higher frequency of lymph node metastasis, and higher clinical staging at diagnosis [18, 19]. PTEN located on chromosome 10q23.3, is known as a vital tumor suppressor gene. Recently, researchers found that PTEN has low expression during the development and progression of EC, and implies poor prognosis, higher histological grade and clinical staging, and shorter survival [21, 22]. Especially, the mutation rate of PTEN in EC is about 34% ~ 55%, which is higher than the mutation rate of K-ras and P53 [23, 24]. However, during follow-up of EC patients, we found that some EC patients with down-regulation of PTEN, but up-regulation of ER or PR, and some EC patients with down-regulation of ER or PR, but up-regulation of PTEN also had a poor prognosis. Therefore, to reveal the prognosis of EC patients with different phenotypes based on PTEN, ER and PR expression, 120 cases formalin-fixed paraffin-embedded EC tissues and 543 cases UCEC patients from TCGA-UCEC datasets were analyzed.
Material and Methods
Patients
A total of 120 formalin-fixed paraffin embedded EC tissue samples between March 2005 and April 2015 from Affiliated Hospital of Guangdong Medical University (China), Dongguan Fifth People's Hospital (China), and Affiliated Jiangmen Hospital of Sun Yat-sen University (China) were collected. Tumors were staged according to the FIGO 2009 system [25]. Histological grade was assessed based on the 2014 World Health Organization criteria [26]. Patients, who had history of other tumors, underwent chemotherapy, radiotherapy, radical surgery treatment, or other anticancer therapies prior to surgery were excluded in this study. The demographic and clinical characteristics for all the patients are shown in Table 1. Informed consent was obtained from all study subjects, and the studies were approved by the institutional ethics committee.
The demographic and clinical characteristics for all EC phenotypes
| Group | EC phenotypes [n(%)] | ||||||
|---|---|---|---|---|---|---|---|
| PTENlowERlowPRlow (n = 48) | PTENhighERlowPRlow (n = 30) | PTENhighERhighPRhigh (n = 20) | PTENlowERhighPRhigh (n = 12) | PTENhighERhighPRlow (n = 4) | PTENhighERlowPRhigh (n = 4) | PTENlowERhighPRlow (n = 2) | |
| Histological grade | |||||||
| G1 | 10(20.83) | 18(60.00) | 12(60.00) | 5(41.67) | 1(25.00) | 1(25.00) | 1(50.00) |
| G2 | 20(41.67) | 6(20.00) | 5(25.00) | 5(41.67) | 1(25.00) | 1(25.00) | 1(50.00) |
| G3 | 18(37.50) | 6(20.00) | 3(15.00) | 2(16.67) | 2(50.00) | 2(50.00) | 0(-) |
| FIGO clinical stage | |||||||
| I | 12(25.00) | 20(66.67) | 9(45.00) | 5(41.67) | 1(25.00) | 2(50.00) | 0(-) |
| II | 18(37.50) | 4(13.33) | 7(35.00) | 3(25.00) | 1(25.00) | 1(25.00) | 1(50.00) |
| III | 13(27.08) | 4(13.33) | 3(15.00) | 3(25.00) | 1(25.00) | 1(25.00) | 1(50.00) |
| IV | 5(10.42) | 2(6.67) | 1(5.00) | 1(8.33) | 1(25.00) | 0(-) | 0(-) |
| Myometrial tumor invasion | |||||||
| Yes | 22(45.83) | 15(50.00) | 8(40.00) | 7(58.33) | 1(25.00) | 2(50.00) | 0(-) |
| No | 26(54.17) | 15(50.00) | 12(60.00) | 5(41.67) | 3(75.00) | 2(50.00) | 2(50.00) |
| Age (years) | |||||||
| > 45y | 30(62.50) | 17(56.67) | 12(60.00) | 7(58.33) | 2(50.00) | 3(75.00) | 1(50.00) |
| ≤ 45y | 18(37.50) | 13(43.33) | 8(40.00) | 5(41.67) | 2(50.00) | 1(25.00) | 1(50.00) |
Immunohistochemistry (IHC)
Tissue sections (4-μM) were prepared from formalin-fixed paraffin embedded tissue blocks and then subjected to stain with hematoxylin and eosin as our previously described [27-29]. The results were assessed by two pathologists to demonstrate the presence of tumor and the proportion of tumor cells in each section. Tissue sections were subjected to incubate in 0.3% Hydrogen peroxide solution for 10 min at room temperature to block the endogenous peroxidase activity, and then washed by Phosphate Buffered Saline (PBS) solution. Antigenic epitopes were next retrieved by heating for 2 min in 10 mmol/L citrate buffer (pH 6.0). The slides were then first incubated with antibodies against PTEN (ZSGB-BIO, China), ER (ZSGB-BIO, China), PR (ZSGB-BIO, China), Ki-67 (Thermofisher,USA), p53 (Thermofisher,USA), CEA (Thermofisher,USA) and CA125 (Thermofisher,USA) for 30 min at room temperature. Next, the sections were washed with PBS for three times and followed by a goat anti-rabbit and mouse IgG-HRP (Kit-0015, Maixin Biotech, Fuzhou, China) secondary antibody for one hour at room temperature at 1:500 dilutions. The slides were visualized using DAB Detection Kit (Enhanced Polymer) (Kit-0015, Maixin Biotech, Fuzhou, China) and chromogenic reaction was controlled under a microscope (Nikon). After immunostaining, sections were immersed into hematoxylin for nuclear staining, then dehydrated through gradient concentrations of ethanol, cleared with xylene, and covered with neutral balsam.
Score of immunohistochemical sections
The score of immunohistochemical sections were assessed by two pathologists in a blinded fashion to the clinical status of the patients, as our previously reported [27-29]. The immunoreactive area (percentage of positive staining cells) and intensity scores of PTEN, ER and PR were evaluated. In brief, according to the immunoreactive area, 0-5% scored 0, 6-25% scored 1, 26-50% scored 2, 51~75% scored 3 and more than 75% scored 4. If the scores was 0~2, the section was defined as low expression, and if the final scores was 3~6 was defined as high expression. Two specialists who were blinded to the clinical status of the patients evaluated the staining independently.
mRNA expression
All available mRNA expression data were collected from 543 UCEC tumors and 23 adjacent non-EC tissues in TCGA (https://cancergenome.nih.gov/). According to the average value of mRNA expression of each gene, EC patients were divided into 8 phenotypes (PTENLESR1LPGRL, PTENLESR1HPGRH, PTENHESR1LPGRL, PTENHESR1HPGRH, PTENLESR1LPGRH, PTENLESR1HPGRL, PTENHESR1HPGRL, and PTENHESR1LPGRH), according to high and low PTEN, PGR and ESR1 mRNA expression.
Statistical analysis
Statistical analysis was performed using SPSS 19.0 Software (SPSS, Chicago, IL, USA). Chi-square test or Fisher's exact test was employed for analysis the differences of categorical variables. For survival analysis, overall survival (OS) or disease-free survival (DFS) was calculated using Kaplan-Meier method and evaluated by log-rank test, as our previously reported [30, 31]. Multivariate analysis was based on the Cox proportional hazard regression model. A p value <0.05 was considered with statistical significance.
Results
Classification of EC tissues based on PTEN, ER and PR expression
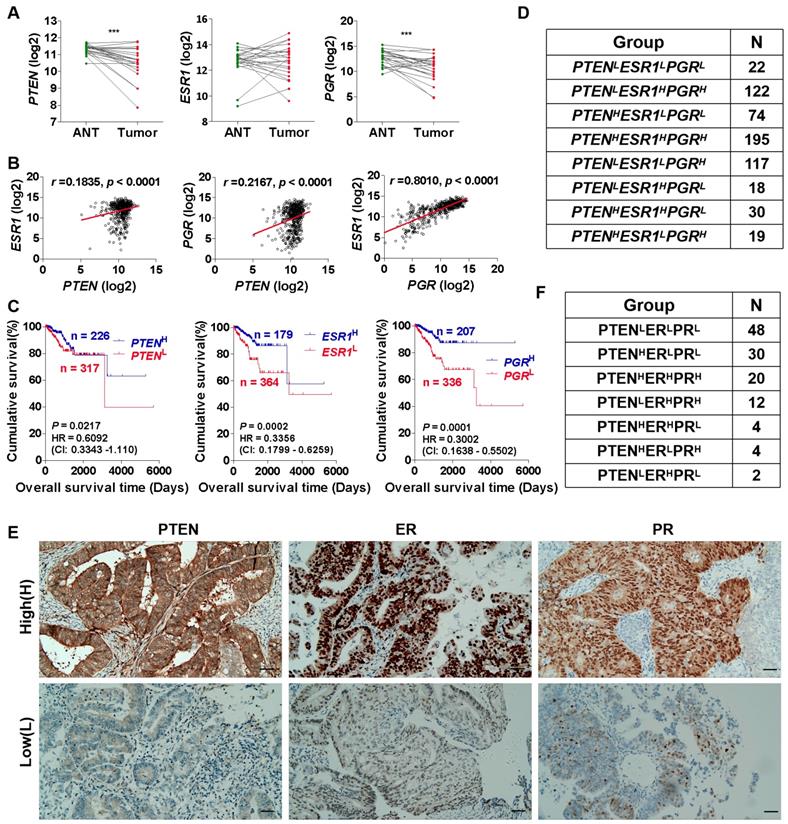
It has been reported that the tumor suppressor gene PTEN is down-regulated in a variety of cancers, including breast cancer [32], prostate cancer [32] and EC [33], etc. PTEN deficiency accelerates tumuor progression and invasiveness [34], promotes macrophage infiltration [35], and plays a significant role in the pathogenesis of carcinogenesis [36]. Herein, we first analyzed the cancer genome atlas uterine corpus endometrial carcinoma (TCGA-UCEC) datasets and found that PTEN mRNA expression was down-regulated in EC tumor tissues compared with adjacent normal tissues (ANT) (Fig. 1A). Prognostic factors of EC include the presence of ER and PR. We also found that the mRNA expression of PGR encoding PR, but not ESR1 encoding ER, down-regulated in EC tissues compared with ANT in TCGA-UCEC datasets (Fig. 1A). Furthermore, correlation analysis showed that there was a significant correlation among PTEN, PGR and ESR1 mRNA expression (Fig. 1B), and they all associated with the prognosis of EC (Fig. 1C). This was also consistent with the results reported in most previous studies [18, 19, 21, 22]. To further reveal the relationship between differential expression of PTEN, ER and PR, and EC prognosis, EC patients were divided into 8 phenotypes (PTENLESR1LPGRL, PTENLESR1HPGRH, PTENHESR1LPGRL, PTENHESR1HPGRH, PTENLESR1LPGRH, PTENLESR1HPGRL, PTENHESR1HPGRL, and PTENHESR1LPGRH), according to high (H) and low (L) PTEN, PGR and ESR1 mRNA expression (Fig. 1D). Additionally, we collected 120 formalin-fixed paraffin-embedded EC tissues and examined PTEN, ER and PR expression by IHC analysis (Fig. E). Based on PTEN, ER and PR expression, EC tissues can be classified to PTENLERLPRL (48/120), PTENHERLPRL (30/120), PTENHERHPRH (20/120), PTENLERHPRH (12/120), PTENHERHPRL (4/120), PTENHERLPRH (4/120), and PTENLERHPRL (2/120) phenotype (Fig. 1F). The demographic and clinical characteristics for all EC phenotypes are shown in Table 1. 60% of EC patients with PTENHERLPRL and PTENHERHPRH phenotype were G1 histological grading, respectively, while 20.83% of EC patients with PTENLERLPRL phenotype were G1 histological grading (Table 1). Similarly, in FIGO clinical staging, most EC patients with PTENHERLPRL (66.7%) and PTENHERHPRH (45.0%) phenotype were stage I, while 25.00% of EC patients with PTENLERLPRL phenotype were stage I (Table 1). These results suggest that different EC phenotypes which classified by PTEN, ER and PR expression may be associated with clinical pathological and histological grading.
Classification of EC tissues based on PTEN, ER and PR expression. (A) Comparison of PTEN, PGR and ESR1 mRNA expression between EC tumor tissues and adjacent normal tissues (ANT) based on TCGA database, respectively. ***: p < 0.001. (B) Correlation analysis among PTEN, PGR and ESR1 mRNA expression. (C) Kaplan-Meier analysis of overall survival time of EC patients with high PTEN mRNA expression versus low PTEN mRNA expression, high PGR mRNA expression versus low PGR mRNA expression and high ESR1 mRNA expression versus low ESR1 mRNA expression, respectively, based on TCGA database. (D) EC patients were divided into 8 phenotypes according to high and low PTEN, PGR and ESR1 mRNA expression based on TCGA database. (E) Detection of PTEN, ER and PR expression in EC tissues by IHC analysis. (F) EC patients were divided into 7 phenotypes according to high and low PTEN, ER and PR expression based on IHC analysis. H: high, L: low.
EC patients with triple-high expression of PTEN, ER and PR showed a lower degree of malignancy and proliferative activity
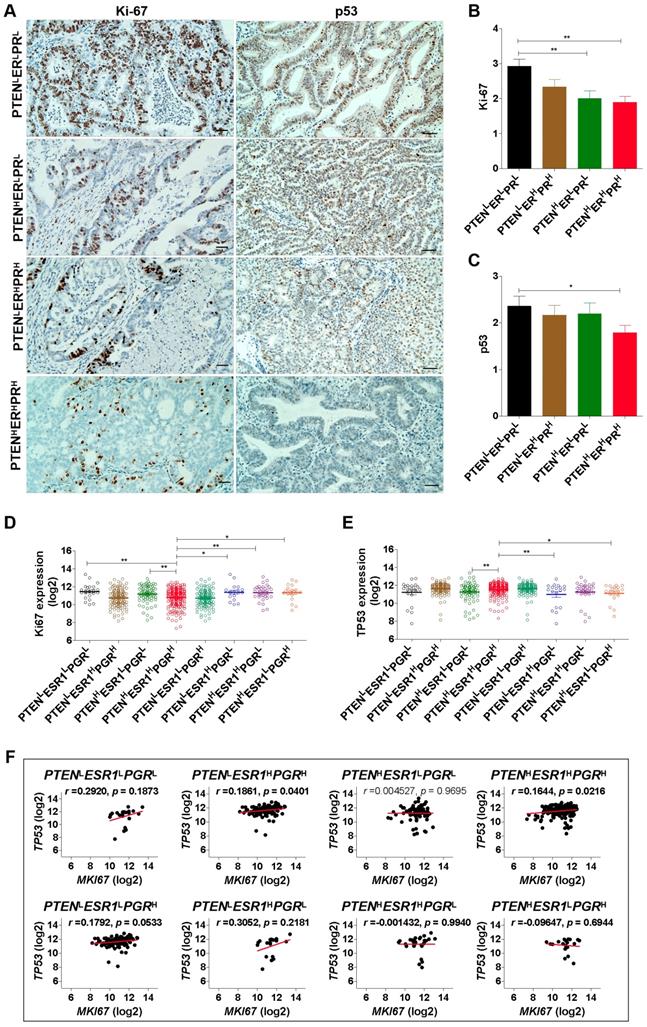
To reveal the proliferative activity of EC patients with different phenotypes, Ki-67 and p53 were detected by IHC analysis (Fig. 2A). Results showed that Ki-67 was low expressed in EC patients with PTENHERLPRL and PTENHERHPRH phenotype, while high expressed in EC patients with PTENLERLPRL phenotype (Fig. 2B). Indeed, based on TCGA-UCEC datasets, we also found that EC patients with PTENLESR1LPGRL phenotype had high expression of Ki67 mRNA, while patients with PTENHESR1HPGRH phenotype had low expression of Ki67 mRNA (Fig. 2C). Simultaneously, we also found p53 was low expressed in EC patients with PTENHERHPRH phenotype, and TP53 (encoding p53) mRNA was low expressed in EC patients with PTENHESR1HPGRH phenotype (Fig. 2D, 2E). Interestingly, there was a positive correlation between Ki67 and TP53 mRNA expression in patients with PTENHESR1HPGRH phenotype (r =0.1644; p = 0.0216) and PTENLESR1HPGRH phenotype (r =0.1861; p = 0.0401), respectively, but not in PTENLESR1LPGRL, PTENHESR1LPGRL, PTENLESR1LPGRH, PTENLESR1HPGRL, PTENHESR1HPGRL, and PTENHESR1LPGRH phenotype (Fig. 2F). These results suggest that EC patients with triple-high expression of PTEN, ER and PR showed a lower degree of malignancy and proliferative activity.
EC patients with triple-H expression of PTEN, ER and PR showed a Ler expression of Ki-67 and p53. (A) Detection of Ki-67 and p53 expression in EC tissues with PTENLERLPRL, PTENHERLPRL, PTENHERHPRH and PTENLERHPRH phenotypes by IHC analysis. (B) Comparison of Ki-67 expression among PTENLERLPRL, PTENHERLPRL, PTENHERHPRH and PTENLERHPRH phenotypes. **: p < 0.01. (C) Comparison of Ki67 mRNA expression among PTENLESR1LPGRL, PTENLESR1HPGRH, PTENHESR1LPGRL, PTENHESR1HPGRH, PTENLESR1LPGRH, PTENLESR1HPGRL, PTENHESR1HPGRL, and PTENHESR1LPGRH phenotypes based on TCGA database. *: p < 0.05; **: p < 0.01. (D) Comparison of p53 expression among PTENLERLPRL, PTENHERLPRL, PTENHERHPRH and PTENLERHPRH phenotypes. **: p < 0.01. (E) Comparison of TP53 mRNA expression among PTENLESR1LPGRL, PTENLESR1HPGRH, PTENHESR1LPGRL, PTENHESR1HPGRH, PTENLESR1LPGRH, PTENLESR1HPGRL, PTENHESR1HPGRL, and PTENHESR1LPGRH phenotypes based on TCGA database. *: p < 0.05; **: p < 0.01. (F) Correlation analysis between Ki67 and TP53 mRNA expression in PTENLESR1LPGRL, PTENLESR1HPGRH, PTENHESR1LPGRL, PTENHESR1HPGRH, PTENLESR1LPGRH, PTENLESR1HPGRL, PTENHESR1HPGRL, and PTENHESR1LPGRH phenotypes based on TCGA database. H: high, L: low.
EC patients with triple-high expression of PTEN, ER and PR showed a lower expression of CA125
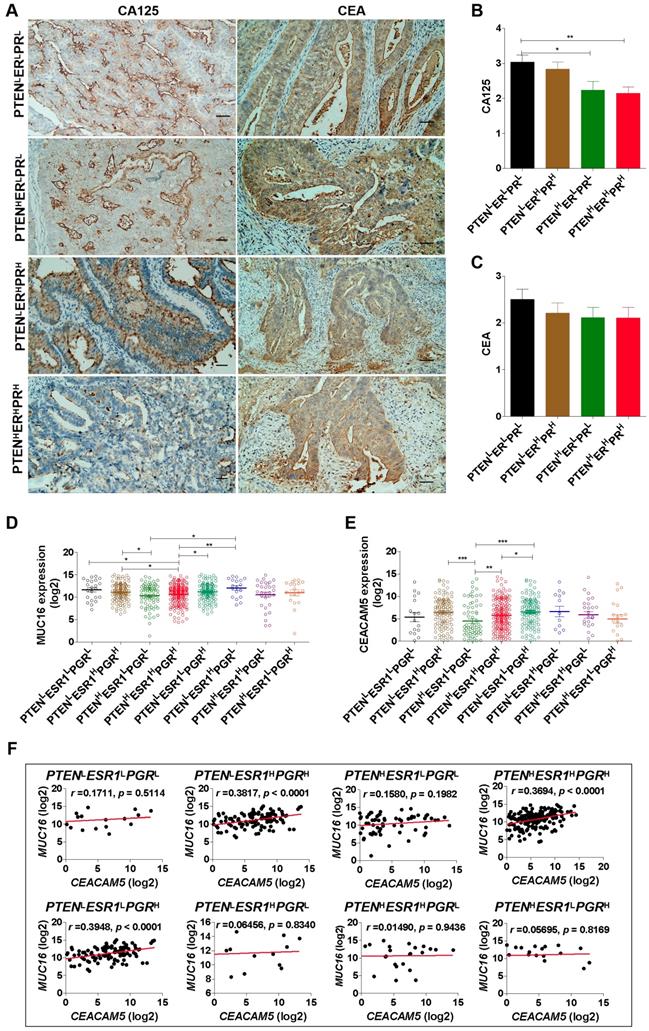
Carcinoma-associated antigens are considered to be useful markers for the detection of recurrent disease in EC patients [37]. Herein, we detected CA125 and CEA expression in EC tissues by IHC analysis (Fig. 3A). The results show that EC patients with PTENHERHPRH phenotype had low expression of CA125 (Fig. 3B) and EC patients with PTENHESR1HPGRH phenotype had low expression of MUC16 (encoding CA125) mRNA (Fig. 3C). Notably, there was no difference in the expression of CEA among patients with PTENLERLPRL, PTENHERLPRL, PTENHERHPRH, and PTENLERHPRH phenotypes (Fig. 3D). However, the mRNA expression of CEACAM5 (encoding CEA) was down-regulated in patients with PTENHESR1HPGRH and PTENHESR1LPGRL phenotype compared to patients with PTENLESR1LPGRH phenotype based on TCGA-UCEC datasets (Fig. 3E). Furthermore, we observed MUC16 mRNA expression was positively related to CEACAM5 mRNA expression in EC patients with PTENHESR1HPGRH (r=0.3694; p < 0.0001), PTENLESR1HPGRH (r=0.3817; p < 0.0001) and PTENLESR1LPGRH (r =0.3948; p < 0.0001) phenotypes, respectively (Fig. 3F). These studies suggest that there are differences in CEA and CA125 expression in EC patients with different phenotypes. In particular, EC patients with triple-high expression of PTEN, ER and PR showed low expression of CA125, this was positively correlated with CEA.
EC patients with triple-H expression of PTEN, ER and PR showed a Ler expression of CA125. (A) Detection of CA125 and CEA expression in EC tissues with PTENLERLPRL, PTENHERLPRL, PTENHERHPRH and PTENLERHPRH phenotypes by IHC analysis. (B) Comparison of CA125 expression among PTENLERLPRL, PTENHERLPRL, PTENHERHPRH and PTENLERHPRH phenotypes. **: p < 0.01. (C) Comparison of MUC16 mRNA expression among PTENLESR1LPGRL, PTENLESR1HPGRH, PTENHESR1LPGRL, PTENHESR1HPGRH, PTENLESR1LPGRH, PTENLESR1HPGRL, PTENHESR1HPGRL, and PTENHESR1LPGRH phenotypes based on TCGA database. *: p < 0.05; **: p < 0.01. (D) Comparison of CEA expression among PTENLERLPRL, PTENHERLPRL, PTENHERHPRH and PTENLERHPRH phenotypes. **: p < 0.01. (E) Comparison of CEACAM5 mRNA expression among PTENLESR1LPGRL, PTENLESR1HPGRH, PTENHESR1LPGRL, PTENHESR1HPGRH, PTENLESR1LPGRH, PTENLESR1HPGRL, PTENHESR1HPGRL, and PTENHESR1LPGRH phenotypes based on TCGA database. *: p < 0.05; **: p < 0.01. (F) Correlation analysis between MUC16 and CEACAM5 mRNA expression in PTENLESR1LPGRL, PTENLESR1HPGRH, PTENHESR1LPGRL, PTENHESR1HPGRH, PTENLESR1LPGRH, PTENLESR1HPGRL, PTENHESR1HPGRL, and PTENHESR1LPGRH phenotypes based on TCGA database. H: high, L: low.
Triple-high expression of PTEN, ER and PR may predict favorable prognosis in EC patients
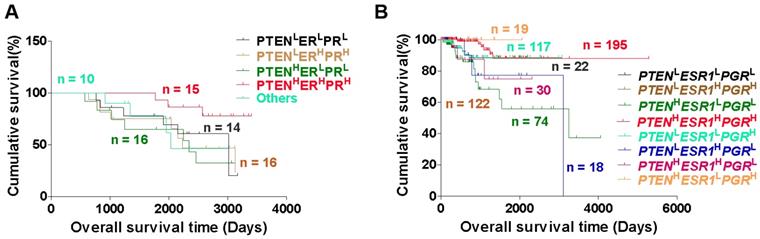
Subsequently, we analyzed the relationship between patients with different EC phenotypes and prognosis, and found that EC patients with PTENHERHPRH phenotype showed a favorable overall survival (OS) time, compared to patients with PTENLERLPRL and PTENHERLPRL phenotype (Fig. 4A). Similarly, EC patients with PTENHESR1HPGRH, PTENLESR1HPGRH, and PTENHESR1LPGRH phenotypes showed a favorable OS time than patients with PTENLESR1LPGRL, and PTENHESR1LPGRL phenotype (Fig. 4B). These studies suggest that triple-high expression of PTEN, ER and PR may predict favorable prognosis in EC patients.
Triple-H expression of PTEN, ER and PR may predict favorable prognosis in EC patients. (A) Kaplan-Meier analysis of overall survival time of EC patients with PTENLERLPRL, PTENHERLPRL, PTENHERHPRH and PTENLERHPRH phenotypes. (B) Kaplan-Meier analysis of overall survival time of EC patients with PTENLESR1LPGRL, PTENLESR1HPGRH, PTENHESR1LPGRL, PTENHESR1HPGRH, PTENLESR1LPGRH, PTENLESR1HPGRL, PTENHESR1HPGRL, and PTENHESR1LPGRH phenotypes based on TCGA database. H: high, L: low.
Discussion
EC is a type of female reproductive malignant tumor, the incidence of which is generally 20~30%. Exploring effective molecular prognosis targets for EC patients has become a hot topic in current research. In 1991, Raju KS, et al. found that post medroxyprogesterone acetate (MPA) dehydrogenase enzyme levels predicted survival more accurately than pre MPA receptor status of the tumors in EC patients [38]. Then, some researchers found lack of bcl-2 [39, 40], PTEN [41], TIMP-2 [42], ER and PR [43], decrease of hemoglobin [44], EpCAM [45], up-regulation of serum CA125 [46], serum TIMP-1 [47], ulex europeus agglutinin-I (UEA-I) [48], HIF-1α [49], aurora B [50], MMP-7 [51], homeobox (HOX) transcript antisense intergenic RNA (HOTAIR) [52], CHRM3 [53], AAA+ (ATPases associated with various cellular activities) nuclear coregulator cancer-associated (ANCCA) [54], glycogen synthase kinase-3β (GSK-3β) [55] can serve as a poor prognostic marker for EC patients. Herein, we also found that decrease of PTEN, PGR and ESR1 mRNA expression were associated with poor prognosis. However, it was not accurate during follow-up. Especially, we found that some EC patients with decrease of PTEN, but up-regulation of ER or PR, and some EC patients with decrease of ER or PR, but up-regulation of PTEN also have a poor prognosis (data not shown).
These characteristics can also be observed when analyzing PTEN, PGR and ESR1 mRNA expression using the TCGA database. Indeed, we found that EC patients can be divided into different phenotypes based on PTEN, ER and PR expression. EC patients were divided into PTENLESR1LPGRL, PTENLESR1HPGRH, PTENHESR1LPGRL, PTENHESR1HPGRH, PTENLESR1LPGRH, PTENLESR1HPGRL, PTENHESR1HPGRL, and PTENHESR1LPGRH phenotypes basing on TCGA-UCEC datasets. Similarly, EC tissues can be classified to PTENLERLPRL, PTENHERLPRL, PTENHERHPRH, PTENLERHPRH, PTENHERHPRL, PTENHERLPRH, and PTENLERHPRL phenotypes basing on IHC analysis.
PTEN is a tumor suppressor gene with double phosphatase activity discovered in 1997. It is a homologous gene of phosphatase and tensin which often associated with the deletion of chromosome allele 10q site [23]. PTEN plays an important role in the inhibition of tumorigenesis by regulating PTEN/PI3K/AKT, PTEN/FAK/P130cas, PTEN/ERK, p53/MDM2, and FRAP/mTOR signaling pathways. These signal pathways through its lipid phosphatase and protein phosphatase activity, induce apoptosis, block cell cycle, to inhibit tumor cell invasion and metastasis and tumor angiogenesis [13, 14]. At present, abnormal expression of PTEN in EC patients has been confirmed by most researchers. In the previous study, we also found that the loss rate of PTEN in EC patients was 58.8% (60/102), and the loss expression of PTEN was closely related to the grade of histological in EC patients, suggesting that the absent expression of PTEN plays an important role on EC occurrence and development [56, 57]. Additionally, the expression of ER and PR may be related to the increase of malignancy, the increase of invasion and the transformation of non-hormone-dependent tumors in EC patients [10-16]. Therefore, the detection of ER and PR may have important value for EC patients' prognosis and treatment. In particular, it is of great significance to detect ER, PR as endocrine therapy marker in EC patients. To reveal the prognosis of EC patients with different phenotypes based on PTEN, ER and PR expression, 120 cases formalin-fixed paraffin-embedded EC tissues and 543 cases UCEC patients from TCGA-UCEC datasets were analyzed. Results showed that EC patients with triple-high expression of PTEN, ER and PR showed a lower expression of Ki-67, p53 and CA125. Furthermore, triple-high expression of PTEN, ER and PR may predict favorable prognosis in EC patients.
Fiorillo, et al. found that the ER-α mutation Y537S, which associated with the over-expression of a number of protein markers of poor clinical outcome (COL6A3, ERBB2, STAT3, AFP, TFF1, CDK4 and CD44) can confer tamoxifen-resistance via enhanced mitochondrial metabolism, glycolysis and Rho-GDI/PTEN signaling [58]. Moreover, PTEN insufficiency stimulated ER+ breast cancer cell growth [59]. Additionally, PI3K/PTEN/AKT pathway was crucial for many aspects of cell growth and survival, feedback regulation on PTEN/AKT pathway by the ER stress kinase PERK mediated by interaction with the Vault complex[60]. ER-activating ability of breast cancer stromal fibroblasts was regulated independently of alteration of PTEN [61]. Herein, basing on IHC analysis, EC tissues can be classified to PTENLERLPRL, PTENHERLPRL, PTENHERHPRH, PTENLERHPRH, PTENHERHPRL, PTENHERLPRH, and PTENLERHPRL phenotypes. And, patients with PTENLERLPRL showed high malignancy, while patients with PTENHERHPRH showed low malignancy. Therefore, combined detection of PTEN, ER, PR may help identify a small subset of EC with more aggressive behavior and may aid in risk stratification.
In summary, our findings indicate that EC patients with triple-high expression of PTEN, ER and PR showed a lower degree of malignancy and proliferative activity and predicted a favorable prognosis. However, the physiological and pathological features of EC patients with different phenotypes basing on PTEN, ER and PR expression still require further investigation.
Abbreviations
PTEN: phosphatase and tensin homolog; ER: estrogen receptor; PR: progesterone receptor; EC: Endometrial carcinoma; UCEC: uterine corpus endometrial carcinoma; TCGA: the cancer genome atlas; BMI: Body Mass Index; OS: Overall survival; IHC: Immunohistochemistry; GSK-3β: glycogen synthase kinase-3β; ANT: adjacent normal tissues.
Acknowledgements
This study was supported by grants from the Medical Science Foundation of Guangdong Province (A2018123), the Science and Technology Project of Dongguan (201750715005451), and Science and Technology Project of Guangdong Province (2014A020212298).
Author Contributions
LY and ZJ conceived and designed the experiments. LY, LB and CC performed the experiments. CS, ZX and YH analyzed the data. LY, ZJ and ZK participated in writing manuscript. The final version of the manuscript has been reviewed and approved for publication by every author.
Ethics Committee Approval and Patient Consent
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Research Ethics Committee of Dongguan Fifth People's Hospital (China).
Competing Interests
The authors have declared that no competing interest exists.
References
1. Ren Y, Huang X, Shan B. et al. Adjuvant concurrent chemoradiation followed by chemotherapy for high-risk endometrial cancer. Gynecologic oncology. 2016;140:58-63
2. Beavis AL, Smith AJ, Fader AN. Lifestyle changes and the risk of developing endometrial and ovarian cancers: opportunities for prevention and management. International journal of women's health. 2016;8:151-67
3. Morice P, Leary A, Creutzberg C. et al. Endometrial cancer. Lancet. 2016;387:1094-108
4. Amant F, Moerman P, Neven P. et al. Endometrial cancer. Lancet. 2005;366:491-505
5. Kandoth C, Schultz N, Cherniack AD. et al. Integrated genomic characterization of endometrial carcinoma. Nature. 2013;497:67-73
6. Murali R, Soslow RA, Weigelt B. Classification of endometrial carcinoma: more than two types. The Lancet. Oncology. 2014;15:e268-78
7. Lewin SN, Wright JD. Comparative Performance of the 2009 International Federation of Gynecology and Obstetrics' Staging System for Uterine Corpus Cancer. Obstetrics and gynecology. 2011;117:1226
8. Söderlin E. Factors affecting prognosis of endometrial carcinoma. Acta obstetricia et gynecologica Scandinavica. Supplement. 1974;38:1-37
9. Beckner ME, Mori T, Silverberg SG. Endometrial carcinoma: nontumor factors in prognosis. International journal of gynecological pathology: official journal of the International Society of Gynecological Pathologists. 1985;4:131-45
10. Tian W, Teng F, Zhao J. et al. Estrogen and insulin synergistically promote type 1 endometrial cancer progression. Cancer biology & therapy. 2017;18:1000-10
11. Abd ENM, El-Gelany S. Differential Expression Patterns of PTEN in Cyclic, Hyperplastic and Malignant Endometrium: Its Relation with ER, PR and Clinicopathological Parameters. Journal of the Egyptian National Cancer Institute. 2009;21:323-31
12. Kappes H, Goemann C, Bamberger AM. et al. PTEN expression in breast and endometrial cancer: correlations with steroid hormone receptor status. Pathobiology: journal of immunopathology, molecular and cellular biology. 2001;69:136-42
13. Pieczyńska B, Wojtylak S, Zawrocki A. et al. Analysis of PTEN, estrogen receptor α and progesterone receptor expression in endometrial hyperplasia using tissue microarray. Polish journal of pathology: official journal of the Polish Society of Pathologists. 2011;62:133-8
14. Peevey JF, Seagle BL, Maniar KP. et al. Association of body mass index with ER, PR and 14-3-3σ expression in tumor and stroma of type I and type II endometrial carcinoma. Oncotarget. 2017;8:42548-59
15. NCM V, van der Wurff AAM, JMA P. et al. Tissue microarray is suitable for scientific biomarkers studies in endometrial cancer. Virchows Archiv: an international journal of pathology. 2018;472:407-13
16. Stoenescu VE, Niculescu M, Novac L. et al. Immunohistochemical reaction of the glandular epithelium in endometrial hyperplasia compared to endometrial carcinoma. Romanian journal of morphology and embryology = Revue roumaine de morphologie et embryologie. 2017;58:791-800
17. Kininis M, Kraus WL. A global view of transcriptional regulation by nuclear receptors: gene expression, factor localization, and DNA sequence analysis. Nuclear receptor signaling. 2008;6:e005
18. Ethier JL, Desautels DN, Amir E. et al. Is hormonal therapy effective in advanced endometrial cancer? A systematic review and meta-analysis. Gynecologic oncology. 2017;147:158-66
19. Zhang Y, Zhao D, Gong C. et al. Prognostic role of hormone receptors in endometrial cancer: a systematic review and meta-analysis. World journal of surgical oncology. 2015;13:208
20. Huvila J, Talve L, Carpén O. et al. Progesterone receptor negativity is an independent risk factor for relapse in patients with early stage endometrioid endometrial adenocarcinoma. Gynecologic oncology. 2013;130:463-9
21. Estrella JS, Broaddus RR, Mathews A. et al. Progesterone receptor and PTEN expression predict survival in patients with low- and intermediate-grade pancreatic neuroendocrine tumors. Archives of pathology & laboratory medicine. 2014;138:1027-36
22. Skrzypczak M, Lattrich C, Häring J. et al. Expression of SCUBE2 gene declines in high grade endometrial cancer and associates with expression of steroid hormone receptors and tumor suppressor PTEN. Gynecological endocrinology: the official journal of the International Society of Gynecological Endocrinology. 2013;29:1031-5
23. Bilbao C, Rodríguez G, Ramírez R. et al. The relationship between microsatellite instability and PTEN gene mutations in endometrial cancer. International journal of cancer. 2006;119:563-70
24. Huang P, Zhang H, Zhang XF. et al. Evaluation of Intraductal Ultrasonography, Endoscopic Brush Cytology and K-ras, P53 Gene Mutation in the Early Diagnosis of Malignant Bile Duct Stricture. Chinese medical journal. 2015;128:1887-92
25. Werner HM, Trovik J, Marcickiewicz J. et al. Revision of FIGO surgical staging in 2009 for endometrial cancer validates to improve risk stratification. Gynecologic oncology. 2012;125:103-8
26. Lu Z, Chen J. Introduction of WHO classification of tumours of female reproductive organs, fourth edition. Zhonghua bing li xue za zhi = Chinese journal of pathology. 2014;43:649-50
27. Liang Y, Chen Q, Du W. et al. Epstein-Barr Virus-Induced Gene 3 (EBI3) Blocking Leads to Induce Antitumor Cytotoxic T Lymphocyte Response and Suppress Tumor Growth in Colorectal Cancer by Bidirectional Reciprocal-Regulation STAT3 Signaling Pathway. Mediators of inflammation. 2016;2016:3214105
28. Liu XJ, Feng JS, Xiang WY. et al. Clinicopathological features of an ascending colon mixed adenoneuroendocrine carcinoma with clinical serosal invasion. International journal of clinical and experimental pathology. 2014;7:6395-8
29. Zeng JC, Zhang Z, Li TY. et al. Assessing the role of IL-35 in colorectal cancer progression and prognosis. International journal of clinical and experimental pathology. 2013;6:1806-16
30. Zhang X, Zhang L, Lin B. et al. Phospholipid Phosphatase 4 promotes proliferation and tumorigenesis, and activates Ca2+-permeable Cationic Channel in lung carcinoma cells. Molecular cancer. 2017;16:147
31. Ren D, Lin B, Zhang X. et al. Maintenance of cancer stemness by miR-196b-5p contributes to chemoresistance of colorectal cancer cells via activating STAT3 signaling pathway. Oncotarget. 2017;8:49807-23
32. Li J, Yen C, Liaw D. et al. PTEN, a putative protein tyrosine phosphatase gene mutated in human brain, breast, and prostate cancer. Science. 1997;275:1943-7
33. Tashiro H, Blazes MS, Wu R. et al. Mutations in PTEN are frequent in endometrial carcinoma but rare in other common gynecological malignancies. Cancer research. 1997;57:3935-40
34. Guigon CJ, Zhao L, Willingham MC. et al. PTEN deficiency accelerates tumour progression in a mouse model of thyroid cancer. Oncogene. 2009;28:509-17
35. Armstrong CW, Maxwell PJ, Ong CW. et al. PTEN deficiency promotes macrophage infiltration and hypersensitivity of prostate cancer to IAP antagonist/radiation combination therapy. Oncotarget. 2016;7:7885-98
36. Petrocelli T, Slingerland JM. PTEN deficiency: a role in mammary carcinogenesis. Breast cancer research: BCR. 2001;3:356-60
37. Bruns F, Micke O, Halek G. et al. Carcinoembryonic antigen (CEA)-a useful marker for the detection of recurrent disease in endometrial carcinoma patients. Anticancer research. 2003;23:1103-6
38. Raju KS, King RJ, Taylor RW. Biochemical changes in endometrial carcinoma and their correlation to clinical outcome and prognosis. European journal of gynaecological oncology. 1991;12:331-9
39. Sakuragi N, Ohkouchi T, Hareyama H. et al. Bcl-2 expression and prognosis of patients with endometrial carcinoma. International journal of cancer. 1998;79:153-8
40. Geisler JP, Geisler HE, Wiemann MC. et al. Lack of bcl-2 persistence: an independent prognostic indicator of poor prognosis in endometrial carcinoma. Gynecologic oncology. 1998;71:305-7
41. Kanamori Y, Kigawa J, Itamochi H. et al. PTEN expression is associated with prognosis for patients with advanced endometrial carcinoma undergoing postoperative chemotherapy. International journal of cancer. 2002;100:686-9
42. Honkavuori-Toivola M, Talvensaari-Mattila A, Soini Y. et al. Immunoreactivity for TIMP-2 is associated with a favorable prognosis in endometrial carcinoma. Tumour biology: the journal of the International Society for Oncodevelopmental Biology and Medicine. 2012;33:935-41
43. Li M, Wang Z, Zhao L. et al. Predictors of recurrence and prognosis in patients with stage I and II endometrial carcinoma. Zhonghua fu chan ke za zhi. 2014;49:455-9
44. Tamussino KF, Gücer F, Reich O. et al. Pretreatment hemoglobin, platelet count, and prognosis in endometrial carcinoma. International journal of gynecological cancer: official journal of the International Gynecological Cancer Society. 2001;11:236-40
45. Wen KC, Sung PL, Chou YT. et al. The role of EpCAM in tumor progression and the clinical prognosis of endometrial carcinoma. Gynecologic oncology. 2018;148:383-92
46. Xie Z, Zhang J, Tan A. Relationship between serum CA125 level and prognosis in the patients with uterine endometrial carcinoma. Zhonghua yi xue za zhi. 2001;81:1456-7
47. Honkavuori M, Talvensaari-Mattila A, Puistola U. et al. High serum TIMP-1 is associated with adverse prognosis in endometrial carcinoma. Anticancer research. 2008;28:2715-9
48. Ookuma Y, Hachisuga T, Iwasaka T. et al. Assessment of lectin binding for prognosis in endometrial carcinoma. Pathology. 1994;26:225-9
49. Sivridis E, Giatromanolaki A, Gatter KC. et al. Association of hypoxia-inducible factors 1alpha and 2alpha with activated angiogenic pathways and prognosis in patients with endometrial carcinoma. Cancer. 2002;95:1055-63
50. Kurai M, Shiozawa T, Shih HC. et al. Expression of Aurora kinases A and B in normal, hyperplastic, and malignant human endometrium: Aurora B as a predictor for poor prognosis in endometrial carcinoma. Human pathology. 2005;36:1281-8
51. Misugi F, Sumi T, Okamoto E. et al. Expression of matrix metalloproteinases and tissue inhibitors of metalloproteinase in uterine endometrial carcinoma and a correlation between expression of matrix metalloproteinase-7 and prognosis. International journal of molecular medicine. 2005;16:541-6
52. He X, Bao W, Li X. et al. The long non-coding RNA HOTAIR is upregulated in endometrial carcinoma and correlates with poor prognosis. International journal of molecular medicine. 2014;33:325-32
53. Wang Y, Li J, Wen S. et al. CHRM3 is a novel prognostic factor of poor prognosis in patients with endometrial carcinoma. American journal of translational research. 2015;7:902-11
54. Shang P, Meng F, Liu Y. et al. Overexpression of ANCCA/ATAD2 in endometrial carcinoma and its correlation with tumor progression and poor prognosis. Tumour biology: the journal of the International Society for Oncodevelopmental Biology and Medicine. 2015;36:4479-85
55. Chen S, Sun KX, Liu BL. et al. The role of glycogen synthase kinase-3β (GSK-3β) in endometrial carcinoma: A carcinogenesis, progression, prognosis, and target therapy marker. Oncotarget. 2016;7:27538-51
56. Liang YF, Zeng JC, Kang DP. et al. The expression and clinical significance of PTEN,P27 and BAG-1 in endometrial carcinoma. Chongqing Med. 2014;43:785-7
57. Liang YF, Ruan JB, Kang DP. et al. Expression of estrogen receptor and progesterone receptor on PTEN-deficient endometrial carcinoma. J Clin Exp Patho. 2015:31
58. Fiorillo M, Sanchez-Alvarez R, Sotgia F. et al. The ER-alpha mutation Y537S confers Tamoxifen-resistance via enhanced mitochondrial metabolism, glycolysis and Rho-GDI/PTEN signaling: Implicating TIGAR in somatic resistance to endocrine therapy. Aging. 2018;10:4000-23
59. Chiang KC, Chen HY, Hsu SY. et al. PTEN insufficiency modulates ER+ breast cancer cell cycle progression and increases cell growth in vitro and in vivo. Drug design, development and therapy. 2015;9:4631-8
60. Zhang W, Neo SP, Gunaratne J. et al. Feedback regulation on PTEN/AKT pathway by the ER stress kinase PERK mediated by interaction with the Vault complex. Cellular signalling. 2015;27:436-42
61. Suda T, Oba H, Takei H. et al. ER-activating ability of breast cancer stromal fibroblasts is regulated independently of alteration of TP53 and PTEN tumor suppressor genes. Biochemical and biophysical research communications. 2012;428:259-63
Author contact
Corresponding authors: Yanfang Liang, e-mail: lyfine84com; tel.:+86 0769 85010001; Department of Pathology, Dongguan Hospital Affiliated to Medical College of Jinan University, The Fifth People's Hospital of Dongguan, Dongguan 523905, China. Jincheng Zeng, e-mail:zengjcedu.cn; tel.:+86 0769 22896440; Dongguan Key Laboratory of Medical Bioactive Molecular Developmental and Translational Research, Guangdong Medical University, Dongguan, Guangdong 523808, China.

 Global reach, higher impact
Global reach, higher impact