Impact Factor ISSN: 1837-9664
J Cancer 2020; 11(12):3502-3511. doi:10.7150/jca.42645 This issue Cite
Review
Aberrant expression and regulatory network of splicing factor-SRSF3 in tumors
Institute of Chronic Disease, Qingdao Municipal Hospital, Qingdao University, Qingdao 266000, China.
Received 2019-12-2; Accepted 2020-2-26; Published 2020-3-15
Abstract
Alternative splicing facilitates the splicing of precursor RNA into different isoforms. Alternatively spliced transcripts often exhibit antagonistic functions or differential temporal or spatial expression patterns. There is increasing evidence that alternative splicing, especially by the serine-arginine rich (SR) protein family, leads to abnormal expression patterns and is closely related to the development of cancer. SRSF3, also known as SRp20, is a splicing factor. Through alternative splicing, it plays important roles in regulating various biological functions, such as cell cycle, cell proliferation, migration and invasion, under pathological and physiological conditions. Deregulation of SRSF3 is an essential feature of cancers. SRSF3 is also considered a candidate therapeutic target. Therefore, the involvement of abnormal splicing in tumorigenesis and the regulation of splicing factors deserve further analysis and discussion. Here, we summarize the function of SRSF3-regulated alternative transcripts in cancer cell biology at different stages of tumor development and the regulation of SRSF3 in tumorigenesis.
Keywords: SRSF3, tumor, alternative splicing, biological functions
Introduction
Posttranscriptional modification is an essential component of the genetic code of eukaryotic genomes. Alternative splicing is an important process involved in posttranscriptional modification [1]. It is known that ~95% of human genes can be spliced [2]. Through this process, the noncoding introns are removed, and coding exons containing the correct translational information are left to maintain the stability of eukaryotic organisms. Alternative splicing plays important roles in cell proliferation and differentiation, and thus participates in multiple diseases, including cancer [3, 4].
Alternative splicing refers to the process by which different splicing isomers are produced from a pre-mRNA by different splicing methods, leading to final protein products with different phenotypes, different functional characteristics or antagonistic effects on tumors [5]. Alternative splicing is accomplished via the spliceosome. The spliceosome is mainly composed of five kinds of small nuclear ribonucleic proteins (snRNPs) and three kinds of non-snRNPs [6-8]. Among them, non-snRNPs include the hnRNP protein family, serine/arginine-rich splicing factor (SR) protein family and SR- related protein family. SR protein can bind to pre-mRNA and influence the interaction between snRNP splicing factors and pre-mRNA, thus promoting or inhibiting the recognition and utilization of splicing sites [9].
At present, SRSF1-12 (also named SC35, SRp20, SRp75, SRp40, SRp55, 9G8, SRp46, SRp30c, SRp38, NET2, SRrp35) have been extensively studied [10]. These SR proteins are involved in many intracellular physiological functions, such as mRNA translocation, mRNA stability, genomic stability and mRNA translation [11-14]. Abnormal expression of splicing factors has been known to interfere with normal splicing of pre-mRNA, resulting in abnormal diseases and cancer [15, 16].
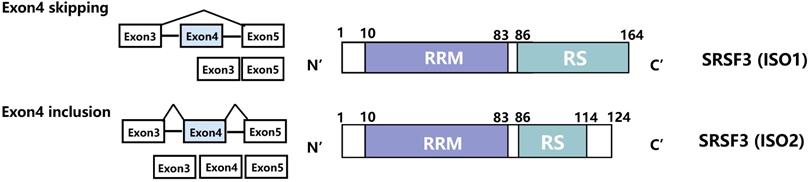
Serine/arginine-rich splicing factor 3 (SRSF3) is the smallest member of the SR protein family. SRSF3 is relatively conserved and binds to specific pre-mRNAs. It contains one RRM (RNA receptor binding domain) and one RS domain (Figure 1). The RRM domain, located at the N-terminus, can specifically recognize and bind to pre-mRNA. Thus, it is the key factor by which SR proteins determine the splicing site [17]. The RRM domain can interact with pre-mRNA cis-elements in a concentration-dependent manner and then regulate the alternative splicing of these genes [18, 19]. For example, exogenous noncoding SRSF3 (exon 4 inclusion) can increase the expression of endogenous SRSF3 and p53 in a dose-dependent way [20]. In addition, the high concentration of SR proteins can even replace U1 snRNP and restore splicing activity to pre-mRNA [21, 22]. The RS domain, located at the C-terminus, is composed of alternately arranged arginine and serine, which mainly mediate the interaction between protein and mRNA or protein and protein. Through interaction, the SR protein can bind to other splicing factors and aggregate near splicing sites of pre-mRNA. Therefore, the RS domain determines the splicing activity of the SR protein and is also a necessary region for splicing pre-mRNA [23].
Schematic represents the isoforms and domains of the SRSF3. The full-length SRSF3 contains an RRM (10-83bp) domain and an RS domain (86-164bp). The RRM domain is used to recognize and bind pre-mRNA, while the RS domain plays a splicing role. The SRSF3 exon 4 skipping promotes the production of full-length SRSF3 (ISO1). The SRSF3 exon 4 inclusion leads to the generation of truncated SRSF3 (ISO2). This splicing is regulated by SRSF1, HnRNP L, SLU7, PTBPs, RBM4 and circSMARCA5.
SRSF3-regulated alternative splicing in tumors
Overexpression of SRSF3 in cancer cell lines has been found through database analysis and tissue sample detection. SRSF3 is highly expressed in many tumor tissues, such as the thyroid, ovary, lung, colon, breast, stomach, skin and bladder [15, 24-26]. Thus, the expression of SRSF3 might be a useful biomarker for diagnosis and prognosis.
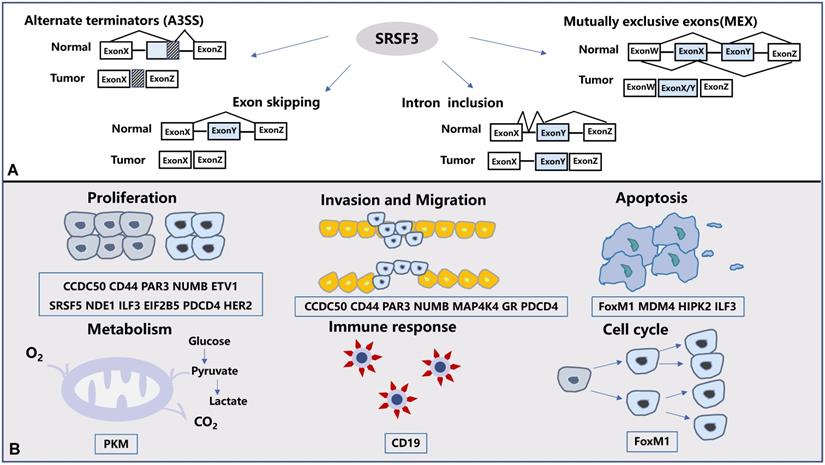
At present, there are five main types of alternative splicing that have been discovered: exon skip (ES), retained intron (RI), alternative promoters (A5SS), alternative terminators (A3SS), and mutually exclusive exons (MEX) [27]. Among them, SRSF3 is involved in four types of splicing (Figure 2A).
The alternative splicing and biological functions regulated by SRSF3. (A). SRSF3 is involved in four kinds of splicing ways in human tumors: exon skipping, intron retention, alternate terminators(A3SS) and mutually exclusive exons (MEX). (B). SRSF3-mediated alternative splicing results in different protein isoforms that regulate a variety of biological processes in tumors. The biological functions regulated by SRSF3 includes tumor proliferation, metastasis, apoptosis, cell metabolism, immune response and cell cycle.
SRSF3 and exon skipping (ES)
SRSF3 promotes exon skipping
CCDC50 (long isoform including alternative exon 6, CCDC50L) has been recognized as a tyrosine- phosphorylated protein. Overexpression of SRSF3 can promote CCDC50 exon 6 skipping to form CCDC50S (short isoform lacking alternative exon 6, CCDC50S) (Table 1). Compared with CCDC50L, CCDC50S has a higher expression and tissue specificity in hepatocellular carcinoma (HCC). SRSF3 can also improve the stability of CCDC50S in the cytoplasm and further increase the activation of the Ras/Foxo4 signaling pathway. Further, upregulated expression of CCDC50S can increase the proliferation and metastasis of HCC [28]. CCDC50S can be used as a new therapeutic target and prognostic biomarker for HCC.
SRSF3 regulates splicing variants
| Classification | SRSF3 | Splicing proteins | Mechanisms | Isoform | Tumor types | Cell lines | Molecular functions | Reference |
|---|---|---|---|---|---|---|---|---|
| Exon-skip (ES) | Promote | CCDC50 | Exon 6 skipping | CCDC50S | HCC | HepG2 cells Bel-7402 cells Bel-7404 cells | Increase the proliferation and migration | [28] |
| CD44 | Exons12-14 skipping | CD44E | Gastric cancer | MKN-45 cells | Increase the gastric tumorigenesis | [32, 114] | ||
| PAR3 | Exon 12 skipping | Δ12PAR3 isoform | TNBC | MDA-B231 cells | Increase metastasis and proliferation | [37] | ||
| NUMB | Exon 12 skipping | Δ12NUMB isoform | TNBC | MDA-MB231 cells | Increase metastasis and proliferation | [37] | ||
| Git2 | Exon 15 skipping | — | HCC | HKO mice | Mediate EMT in mice | [111] | ||
| Slk | Exon 13 skipping | — | HCC | HKO mice | Mediate EMT in mice | [111] | ||
| Inhibit | ETV1 | Exon 7 skipping | ETV1-∆E7 | GBM | GSC83 cells | Impair the growth and sphere formation ability of GSCs | [40] | |
| MAP4K4 | Exon16 skipping | MAP4K4 Iso2 | CRC | HCT-8 cells | Promote the migration and invasion of CRC cell | [42] | ||
| GR | Exon 9 skipping | GRα | TNBC | MDA-MB-231 cells | Promote transcriptional regulation and cell migration | [45] | ||
| FoxM1 | Exon 9 skipping | — | Osteosarcoma | U2OS cells | Affects G2/M arrest, growth retardation and apoptosis | [15] | ||
| MDM4 | Exon 6 skipping | MDM4-S | Melanoma | A375 cells | Promote cell cycle arrest and melanoma cell apoptosis | [52] | ||
| HIPK2 | Exon 8 skipping | HIPK2 ΔE8 | Colon cancer | HCT116 cells | Decrease cell cycle progression from G1 to S phase | [48] | ||
| CD19 | Exon 2 skipping | CD19 ΔEx2 | B-ALL | 697 cells; NALM-6 cells | Increase the abundance of CD19 Δex2 variant | [114] | ||
| Myo1B | Exon23 skipping | — | HCC | HKO mice | Mediate EMT in mice | [111] | ||
| Ctnnd1 | Exon18 skipping | — | HCC | HKO mice | Mediate EMT in mice | [111] | ||
| Retained intron (RI) | Overexpression | eIF2Bϵ | Intron retention | Truncated eIF2Bε | HNC | Hypoxic SQ20B cells | Increase HNC cancer cells growth | [115] |
| Alternate terminators(A3SS) | Overexpression | SRSF5 | Exon 6 inclusion | SRSF5-S | OSCC | CAL 27 cells | Promote OSCC proliferation | [71] |
| Mutually exclusive exons (MEX) | Overexpression | NDE1 | Exon 9 inclusion Exon 9' inclusion | NDE1_KMLL NDE1_SSSC | GBM | GSC83 cells | Promote the growth of GBM cells Inhibit the growth of GBM cells | [40] |
| Knockdown Overexpression | PK | Exon 10 skipping Exon 9 skipping | PKM1 PKM2 | Colon cancer | DLD-1 cells | Inhibits the proliferation and alters metabolic patterns of colon cancer | [75] |
SRSF3 can also mediate some receptors. CD44, an important membrane receptor of hyaluronic acid (HA), is also involved in various processes, including tumor proliferation and metastasis [29-31]. Overexpression of SRSF3 promotes skipping of CD44 exons 12-14 to form the CD44E variant, which enhances tumorigenesis (Table 1) [32].
TAR-DNA binding protein (TARDBP, also named TDP43) belongs to the hnRNP family [33]. The expression of SRSF3 is positively correlated with TDP43 in Triple-negative breast cancer (TNBC). In particular, TDP43 interacts with SRSF3, and they can enhance exon12 skipping of PAR3 (Δ12PAR3 isoform) and NUMB (Δ12NUMB isoform). In contrast to PAR3 and NUMB, which inhibit the proliferation and metastasis of tumors, Δ12PAR3 and Δ12NUMB are known as oncogenes [34-36]. Therefore, overexpression of Δ12PAR3 and Δ12NUMB can rescue the inhibition caused by TDP43 or SRSF3 knockdown in TNBC (Table 1) [37].
SRSF3 inhibits exon skipping
In addition to exon skipping, SRSF3 can also promote the exon inclusion (Table 1). Transcription factor ETS variant 1 (ETV1) is an oncogene belonging to the E twenty-six (ETS) family that regulates cell proliferation, differentiation, migration and angiogenesis [38]. ETV1-FL regulates the transcription of four cancer-related genes: ADAM metallopeptidase domain 19 (ADAM19), integrin alpha 2 (ITGA2), and fermitin family member 1 (FERMT) [38, 39]. Moreover, four serine/threonine (S/T) residues of ETV1 can be phosphorylated by MAPK4 to reduce the ubiquitination degradation of full-length ETV1 (ETV1-FL) [38]. Xiao et al. found that the knockout of SRSF3 promotes the skipping of ETV1-FL exon 7 (ETV1-∆E7) in glioma stem-like cells (GSCs). ETV1-∆E7 cannot be phosphorylated by MAPK4 and thus exhibits lower stability. In this way, ETV1-∆E7 inhibits its target gene expression and impairs the growth and sphere formation ability of GSCs [40].
Some kinase activity is also related to SRSF3. Only Iso2 (exon 16 inclusion) of mitogen-activated protein 4 kinase 4 (MAP4K4) can interact and phosphorylate c-Jun N-terminal kinase (JNK) [41]. Overexpressed SRSF3 binds to the CU element in exon 16 and promotes the formation of MAP4K4 Iso2, which further activates the JNK signaling pathway [42]. JNK signaling pathways widely promote the migration and invasion of cancer cells [43]. SRSF3 increases the expression of the EMT-related genes N-cadherin and vimentin, thus promoting the invasion and migration of colorectal cancer (CRC) cells [42].
SRSF3 can also promote migration in TNBC. Cortisol-induced SRSF3 overexpression can promote exon 9 inclusion of GR and subsequently form GRα. In contrast to GR, GRα can respond to glucocorticoid actions [44]. Erica et al. found that GRα promotes the transcriptional regulation of receptor for activated C kinase 1 (RACK1), which can further increase the migration of MDA-MB-231 cell lines [45]. TNBC lacks effective treatment strategies, but approximately 25% of invasive TNBC is known to be glucocorticoid receptor (GR)-positive [46]. SRSF3 might be a novel therapy target for GR-positive TNBC.
Forkhead box transcription factor M1 (FoxM1) is also a transcription factor related to tumor proliferation [47]. It has been reported that SRSF3 knockdown promotes FoxM1 exon 9 skipping and downregulates the expression of FoxM1 and its two transcriptional targets PLK1 and Cdc25B, both of which are cell cycle-related proteins. As a result, decreased expression of FoxM1, PLK1 and Cdc25B results in inhibition of the G2/M phase transition. Therefore, it hinders the progression of the cell cycle and promotes the apoptosis of U2OS cells [15].
In addition to the G2/M phase transition, SRSF3 plays an important role of in regulating the progression of the G1/S phase transition in colon cancer cells. Knockdown of SRSF3 can reduce the expression of cyclin D1, cyclin D3, cyclin E1, E2F1 and E2F7, which can impair the G1-to-S-phase progression. In this way, SRSF3 induces G1 cell cycle arrest and apoptosis [48].
Knockdown of SRSF3 causes apoptosis in ovarian cancer cells [49]. The oncogene MDM4 can effectively promote the occurrence and proliferation of melanoma [50, 51]. Overexpressed SRSF3 binds to MDM4 exon 6, promotes the inclusion of exon 6 and forms normal function of MDM4 (MDM4-L), thereby inhibiting p53-mediated target gene transcription and promoting cell cycle arrest and melanoma cell apoptosis [52]. Strikingly, the regulation of exon 6 inclusion is also a key determinant of MDM4 protein abundance in normal tissues and cancer cells. Exon 6 skipping can reduce MDM4 protein abundance [52].
Homeodomain-interacting protein kinase-2 (HIPK2) is also related to the apoptosis of cancer cells, and the deletion of HIPK2 increases the proliferation potential of cancer cells [53-55]. In addition, full-length HIPK2 can promote the E3 ligase activity of Siah-1 [56]. Knocking down SRSF3 leads to the skipping of exon 8 of HIPK2, thus promoting the formation of the Δe8 isoform. Because the Δe8 isoform lacks 27 amino acids that mediate its binding to Siah-1, it cannot participate in protein degradation [48]. In this way, depletion of SRSF3 promotes the apoptosis of colon cancer cells.
In addition to these solid tumors, significant progress has been made in the treatment of pediatric B-cell acute lymphoblastic leukemias (B-ALL), but the mortality and recurrence rates are still high in childhood cancer [57]. CD19 is a cell surface signaling protein. Because it can recruit intracellular kinases, CD19 is considered to play an indispensable role in promoting B cell proliferation and differentiation [58-61]. Elena et al. found that knockdown of SRSF3 resulted in enhanced abundance of the CD19 Δex2 isoform. Consistent with this, in relapsed leukemia, low expression of SRSF3 is correlated with high expression of the CD19Δex2 variant. At present, the effective treatment strategy combats CD19 through the application of T cells expressing chimeric antigen receptors (CAR-T) to improve the cure rate of B-ALL in pediatrics [62, 63]. However, because of the existence of alternative splicing, a sequence of CD19Δex2 is missing, and the CAR-T cannot recognize CD19Δex2, which reduces the survival rate of pediatric B-ALL [64]. It is worth mentioning that the role of the CD19 Δex2 variant in the differentiation and proliferation of normal B cells is unknown.
SRSF3 and retained intron (RI)
EIF2B5, a major regulator of translation initiation, has been shown to be a marker protein in various cancers [65, 66]. EIF2Bε is the largest subunit of EIF2B5, which has two isoforms, truncated eIF2Bε (65 kDa) and full-length eIF2Bε (80 kDa). Under hypoxic conditions, SRSF3 promotes the intron 12 retention of eIF2Bε and produces a premature termination codon (PTC) that inhibits the overall translation and forms truncated eIF2Bε in head and neck carcinoma (HNC) cells (Table 1). This truncated eIF2Bε lacks the binding region of eIF2Bα and an activating guanine exchange factor (GEF) domain [67]. Therefore, it cannot bind eIF2Bα (a subunit of EIF2B5) and guanosine triphosphate (GTP) to initiate the translation process. Finally, truncated eIF2Bε improves the survival rate of HNC cancer cells under prolonged or acute hypoxia [68].
SRSF3 and alternate terminators (A3SS)
Serine/arginine-rich splicing factor 5 (SRSF5) is another important member of the SR protein family that can promote the proliferation of lung cancer cells [69, 70]. SRSF5 has recently been discovered as a novel oncogene that promotes oral squamous cell carcinoma (OSCC) tumor formation and proliferation [71]. When alternative splicing occurs at the distal 3' splice site of exon 6, it results in a normal functional long-SRSF5 (SRSF5-L). However, when alternative splicing occurs at the proximal 3' splice site of exon 6, a short fragment (approximately 956 bp) is generated due to the presence of an in-frame stop codon (Table 1). Finally, the short fragment either causes RNA degradation induced by nonsense-mediated mRNA decay (NMD) or produces a SRSF5-L lacking the RS domain [72]. In general, SRSF3 is positively correlated with SRSF5 protein expression in OSCC. Moreover, overexpression of SRSF3 promotes the utilization of the distal 3' splice site of SRSF5 exon 6, resulting in an increase in SRSF5-L, thereby promoting OSCC proliferation [71].
SRSF3 and mutually exclusive exons (MEX)
NudE neurodevelopment protein 1 (NDE1) is a dynein-binding protein that regulates various microtubule-mediated processes, including mitosis [73]. Because NDE1 terminal exon 9 and exon 9' are mutually exclusive, there are two isoforms in GSCs: NDE1_KMLL (exon 9 inclusion) and NDE1_SSSC (exon 9' inclusion). NDE1_SSSC exists in normal brain tissue and participate in mitosis. However, NDE1- KMLL has the specific function of promoting the growth of GBM cells in the process of mitotic spindle formation. Knockdown of SRSF3 can promote a transition from NDE1_KMLL to NDE1_SSSC (Table 1) [40].
By alternative splicing, the pyruvate kinase muscle (PKM) gene can produce two isoforms: PKM1 (exon 10 skipping) and PKM2 (exon 9 skipping). In addition, exon 9 and exon 10 are mutually exclusive. Most cancer cells mainly express PKM2 to maintain glycolysis-dominant energy metabolism [74]. Three splicing factors, SRSF3, polypyrimidine tract binding protein 1 (PTBP1) and heterogeneous nuclear ribonucleoprotein A1 (hnRNPA1), can bind to exon 10 and promote the inclusion of exon 10, reducing the PKM1/PKM2 ratio (Table 1). Three splicing factors, PTBP1, SRSF3 and hnRNPA1, are highly expressed in colorectal cancer compared with adjacent tissue [75]. In addition, this splicing process inhibits the proliferation of colon cancer tumors and alters their metabolic patterns, from glycolysis to oxidative phosphorylation [75].
SRSF3 regulates various splicing variants
SRSF3 alone regulates various splicing variants
Interleukin enhancer binding factor 3 (ILF3), a double stranded RNA binding protein, is the main factor regulating the proliferation of cancer cells [76, 77]. SRSF3 binds to different motifs on ILF3 pre-mRNA and produces multiple ILF3 isoforms by exclusion/inclusion of ILF3 exon 18 or by selection of an alternative 3' splice site within exon 18. Among them, ILF3 isoform 1 and isoform 2 can promote cell proliferation, while isoform 5 and isoform 7 suppress tumor cell proliferation and the isoform 7 induces cell apoptosis. Rong et al. confirmed that knocking down SRSF3 attenuated the expression of isoform 1 and isoform 2, and thus inhibited in HeLa and U2OS cell proliferation [78]. Meanwhile, when SRSF3 is knocked down, increased ILF3 isoform 5 inhibits the proliferation of HeLa and U2OS cells, and increased ILF3 isoform 7 induces apoptosis (Table 2) [79].
SRSF3 regulates various splicing variants
| Coordination | Splicing proteins | Mechanisms | Isoform | Tumor types | Cell lines | Molecular functions | Reference |
|---|---|---|---|---|---|---|---|
| — | ILF3 | Exon 18 skipping Exon 18 inclusion | ILF3 isoform -1 ILF3 isoform -2 | Osteosarcoma | U2OS cells; MEF3T3 cells | Promote cell proliferation and transformation | [79] |
| — | PDCD4 | Cassette exon skipping Cassette exon inclusion | PDCD4 AS-1 PDCD4 AS-2 | CRC | SW480 cells | Reduce PDCD4 protein expression | [81] |
| hnRNP H1 | HER2 | Exon 16 skipping Intron 15 retention Exon 15a inclusion | Δ16 HER2 p100 X5 | Breast cancer | SKBR3 cells | Inhibit the proliferation of breast cancer | [85] |
As a tumor suppressor, programmed cell death 4 (PDCD4) is downregulated in most tumors [80]. The PDCD4 gene consists of 12 exons, and the cassette exon involved in alternative splicing is located between exons 2 and 3. Under normal conditions, total PDCD4 exists in major isoform of PDCD4 (PDCD4 AS-1), but there is little PDCD4 AS-2 (cassette exon inclusion) expression in the cytoplasm. Knockdown of SRSF3 increases the expression level of PDCD4 AS-2 mRNA in the cytoplasm and its nuclear export in colorectal cancer cells (Table 2) [81]. Interestingly, SRSF3 can bind directly to PDCD4 mRNA and mediate translational repression of PDCD4 AS-1 [15, 27, 82]. Taken together, SRSF3 represses PDCD4 AS-1 mRNA at the translational level, and PDCD4 AS-2 mRNA at the alternative splicing and mRNA export levels, finally promoting the proliferation and migration of colorectal tumors.
Interestingly, SRSF3 can interact with the cellular RNA-binding protein, PCBP2, a protein that binds to internal ribosome entry site (IRES) sequences within the genomic RNAs of certain picornaviruses and is required for viral translation. SRSF3 regulates IRES-induced translation initiation [83].
The regulation of SRSF3 on PDCD4 AS-1 mRNA translation proves that SRSF3 can also play a role in the translation process.
SRSF3 collaboratively regulates various splicing variants
HER2 is overexpressed in 20-30% of breast cancer cases. The increase in HER2 expression is related to invasive tumors and adverse prognosis [84]. At present, three splicing variants (Δ16 HER2/p100/X5) of full-length HER2 have been identified, and these isomers have different functions and roles in cancer development. Knockdown of HnRNP H1 can increase the expression of the X5 variant (exon 15a inclusion) and oncogenic Δ16 HER2 (exon 16 skipping), thus increasing the trend of breast tumor proliferation. Notably, knockdown of SRSF3 results in the transition from the Δ16 HER2 to p100 variant (intron 15 retention), which inhibits the proliferation of breast cancer cells [85]. SRSF3 and HnRNP H1 are the first splicing factors shown to produce HER2 splicing variants with different regulatory functions (Table 2).
The regulation of SRSF3
SRSF3 is frequently upregulated in many human tumors and therefore plays a role as an oncogene in various cancer processes. Cells maintain stable expression of SRSF3 through the splicing mechanism of SRSF3. However, because of its carcinogenic effect, SRSF3 must be strictly monitored (Table 3).
The regulation of SRSF3 in tumor
| Regulation | Splicing proteins | Mechanisms | Isoform | Tumor types | Cell lines | Molecular functions | Reference |
|---|---|---|---|---|---|---|---|
| Splicing regulation | SRSF1 | Exon 4 skipping | SRSF3(Iso1) | Osteosarcoma | U2OS cells HeLa cells | Inhibit the expression in cancer cells | [86] |
| HnRNP L | Exon 4 skipping | SRSF3(Iso1) | OSCC | CAL 27 cells | Inhibit the proliferation of OSCC cells | [71] | |
| SLU7 | Exon 4 inclusion | SRSF3(Iso2) | Hepatocytes | AAV-SLU7 cells | Maintain liver homeostasis | [91] | |
| PTBPs | Exon 4 skipping | SRSF3(Iso1) | CRC | HepG2 cells | Decrease the proliferation and migration | [42] | |
| RBM4 | Exon 4 inclusion | SRSF3(Iso2) | CRC | HepG2 cells | Decrease the proliferation and migration | [42] | |
| circSMARCA5 | Exon 4 inclusion | SRSF3(Iso2) | GBM | U87MG cells | Inhibit GBM migration | [98] | |
| Other regulation | Ubiquitination | DARPP-32 | SRSF3 ↑ | Gastric cancer | AGS cells | Increase the gastric tumorigenesis | [103] |
| Neddylation | NEDD8 | SRSF3 ↓ | HCC | HepG2 cells | Increase the risk of liver disease | [104] | |
| Phosphorylation | SRPK2 | SRSF3 ↑ | GBM | HEK293cells | Promote the generation of tumors | [109] |
Splicing regulation of SRSF3
SRSF3 has two splicing forms: the full-length normal form (lacking exon 4, Iso1) and the splicing isoform (containing exon 4, Iso2). Iso2 contains a preterminated codon, which is degraded by NMD to avoid cell toxicity, or it encodes a nonfunctional truncated SRSF3 protein that lacks the RS domain at the C-terminus [86] (Figure 1). Hassan et al. found that SRSF3 can regulate alternative splicing of its own mRNA. Overexpression of SRSF3 attenuates the production of the exon 4-deleted transcript and activates the production of exon 4-containing transcripts [86].
SRSF1
Serine/arginine-rich splicing factor 1 (SRSF1, ASF/SF2) was the first member of the SR protein family to be found. As a cancer-promoting factor, SRSF1 can promote the proliferation and EMT of breast epithelial cells [68]. SRSF1 has been proven to antagonize the self-splicing effect of SRSF3 exon 4. Overexpressed SRSF3 promotes the recognition of exon 4, whereas SRSF1 inhibits the recognition and antagonizes the inclusion of SRSF3 exon 4, thus producing full-length SRSF3 (Iso1) [17, 86].
HnRNP L
Nuclear heterogeneous ribonucleoprotein L (HnRNP L) is a selective pre-RNA splicing factor that maintains translation and RNA stability [36, 87-89]. There is a positive correlation between HnRNP L and SRSF3 in OSCC tissues [71, 90]. It has been reported that HnRNP L can promote splicing of SRSF3 exon 4 and then promote the proliferation of OSCC cells [90]. This suggests that HnRNP L may be a potential carcinogenic target. Therefore, in OSCC, SRSF3 can be spliced by HnRNP L or used as a splicing factor to act on SRSF5 [71].
SLU7
SLU7, as a key splicing factor of RNA precursors, has an effect on inhibiting the development of HCC and maintaining the homeostasis of normal hepatocytes [91]. Moreover, the downregulation of SLU7 can damage liver glucometabolic and lipid metabolism disorders, and increase the risk of cancer [91]. Jiménez M et al. also found that deletion of SLU7 can promote the production of truncated SRSF3-Iso2 in a variety of different cancer cell lines. SLU7 knockdown not only increases the abnormal expression of truncated SRSF3 but also leads to the altered proliferation and abnormal metabolism of tumor cells [92].
PTBPs and RBM4
PTBPs (PTBP1 and PTBP2) can bind to the CU element of SRSF3 exon 4, impairing the automatic splicing regulation of SRSF3 and promoting SRSF3 exon 4 skipping [93]. Intriguingly, RNA-binding motif protein 4 (RBM4) is an antagonist of PTBP that can bind to intronic CU elements and promote the inclusion of SRSF3 exon 4, decrease full-length SRSF3 expression and inhibit the proliferation of CRC cells [94]. Overall, PTBP1 can promote the skipping of SRSF3 exon 4, and increased SRSF3 enhances the production of MAP4K4 Iso2 and Iso5 in HCT-8 and HCT-116 cells. When the expression of RBM4 is higher than that of PTBP1, RBM4 promotes the inclusion of SRSF3 exon 4 and the production of the MAP4K4 Iso1 variant in Colo205 cells [42].
circSMARCA5
Imbalances in the expression of circular RNAs (circRNAs) have been emphasized in several types of cancers [95-97]. Without splicing, circSMARCA5 is a novel tumor suppressor that inhibits the migration of glioblastoma multiforme (GBM) cells [98]. Barbagallo et al. have found that the overexpression of circSMARCA5 increased the inclusion of SRSF3 exon 4 (Iso2) and that the downregulation of circSMARCA5 significantly increased the skipping of SRSF3 exon 4 (Iso1) in the process of splicing SRSF3 by SRSF1. In addition, the ratio of SRSF3 Iso2/ Iso1 is positively correlated with circSMARCA5 expression [98].
Ubiquitination
DARPP-32, also named dopamine and cAMP-regulated phosphoprotein (Mr 32000) [99]. DARPP-32 is overexpressed in 2/3 early stage gastric cancer patients and can increase the survival rate, drug resistance and invasive activity of gastric cancer cells [100-102]. DARPP-32 can enhance the SRSF3 protein expression. The reason is that DARPP-32 can significantly reduce the ubiquitination of SRSF3 in digoxin-induced AGS cells. Conversely, the elimination of endogenous DARPP-32 in MKN-45 cells significantly increases the ubiquitination of SRSF3. DARPP-32 can enhance the stability of SRSF3 and regulate SRSF3-induced CD44 splicing, thus promoting CD44E expression and gastric growth [103]. However, the detailed ubiquitination mechanism is still elusive.
Neddylation
Deepak et al. found that the expression of SRSF3 decreased in early human liver diseases, including nonalcoholic fatty liver disease (NAFLD), nonalcoholic steatohepatitis (NASH), or cirrhosis [104]. Neddylation is a posttranslational modification that is similar to ubiquitination [105]. NEDD8 is a member of the ubiquitin-like protein family and covalently binds to Lys residues of substrates [106]. Under oxidative stress induced by palmitic acid, NEDD8 promotes the neddylation of SRSF3 at 11 lysine sites, resulting in SRSF3 degradation in liver diseases. When lysine 11 mutation (K11R) occurs, SRSF3 neddylation is reduced, protecting SRSF3 from degradation and reducing the risk of liver disease and further progression to HCC [104]. SRSF3 is regulated in a neddylation-dependent manner in the liver, which provides an effective therapeutic strategy for suppressing liver diseases and hepatocellular carcinogenesis.
Phosphorylation
Similar to SR proteins, SRSF3 is mainly located in the nucleus. In the absence of splicing, the SRSF3 protein mainly aggregates into irregular granular nuclei and becomes a typical structure in the nucleus, which is called a "speckle" [107].
SRSF3 is also a nuclear shuttle protein. Its shuttling property is related to the phosphorylation of the RS domain [108]. When serine (residue 105 relative to the C-terminus) in the RS domain is phosphorylated by serine-arginine protein kinase 2 (SRPK2), the SR protein can be released from the speckles in the nucleus. Then, the N-terminal RRM domain specifically binds to the pre-mRNA to complete splicing with other splicing factors [109]. Therefore, the phosphorylation of SR protein is very important to the shuttling property of SR protein. SRSF3 is hypophosphorylated in cell homeostasis. Enhanced phosphorylation of SRSF3 promotes the occurrence of splicing events mediated by carcinogenic SRSF3 and the generation of GBM tumors [40]. In addition, Wang et al. found that hypothalamic gonadotropin-releasing hormone (GnRH) has normal reproductive function and can induce phosphorylation of SRSF3 in a time-dependent way [110].
Conclusions
SRSF3 is upregulated in many kinds of tumors and functions as a proto-oncogene. However, SRSF3 expression is decreased in >50% of human HCCs [111]. Specific loss of SRSF3 in hepatocytes impairs the differentiation and metabolism of hepatocytes in early adulthood [112]. Hepatocyte-specific SRSF3 knockout mice are at risk of liver fibrosis, while SRSF3 can protect mice from CCL4-induced fibrosis and carcinogenesis by inhibiting the inclusion of EDA fibronectin 1 exon [111]. In addition, deletion of SRSF3 promotes the skipping of Git2 exon 15/Slk exon 13, the inclusion of Myo1B exon 23/Ctnnd1 exon 18, and the development of HCC metastasis with aging [113].
In alternative splicing, most of the isomers produced by SRSF3 have different phenotypes, which are manifested in promoting and suppressing cancer. SRSF3 can regulate the cell cycle, apoptosis, cell metastasis and proliferation according to different splicing variations (Figure 2B).
At present, although it is known that SRSF3 can splice related proteins, the mechanism by which SRSF3 splices related proteins are still unclear. In addition, the splicing role of SRSF3 in tumorigenesis and its clear molecular mechanism deserve further investigation. Elucidation of the molecular mechanism of abnormal splicing of SRSF3 caused by tumorigenesis could be of great significance in clarifying the occurrence and development of tumors.
Abbreviations
SRSF3: Serine/arginine-rich splicing factor 3; HCC: Hepatocellular Carcinoma; TNBC: Triple- negative breast cancer; GSCs: Glioma stem-like cells; OSCC: Oral squamous cell carcinoma; B-ALL: B-cell acute lymphoblastic leukemias; CRC: Colorectal cancer; HNC: Head and neck carcinoma; GBM: Glioblastoma multiforme; ETV1: Transcription factor ETS variant 1; MAP4K4: Mitogen-activated protein 4 kinase 4; GR: Glucocorticoid receptor; FoxM1: Forkhead box transcription factor M1; HIPK2: Homeodomain-interacting protein kinase-2; SRSF5: Serine/arginine-rich splicing factor 5; NDE1: NudE neurodevelopment protein 1; PKM: Pyruvate kinase muscle; ILF3: Interleukin enhancer binding factor 3; PDCD4: Programmed cell death 4; SRSF1: Serine/arginine-rich splicing factor 1; HnRNP L: Nuclear heterogeneous ribonucleoprotein L; PTBPs: Polypyrimidine tract binding proteins; RBM4: RNA-binding motif protein 4; SRPK2: Serine-arginine protein kinase 2.
Acknowledgements
This project is supported by grants from the National Natural Science Foundation of China [81702743] and the China Postdoctoral Science Foundation [2019T120568, 2018M640612].
Author Contributions
Y.C. writes manuscripts and figures; L.F. edits manuscripts and provides ideas.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Yang X, Coulombe-Huntington J, Kang S. et al. Wide spread expansion of protein interaction capabilities by alternative splicing. Cell. 2016;164:805-17
2. Pan Q, Shai O, Lee LJ. et al. Deep surveying of alternative splicing complexity in the human transcriptome by high-throughput sequencing. Nature genetics. 2008;40:1413-5
3. Norris AD, Calarco JA. Emerging roles of alternative pre-mRNA splicing regulation in neuronal development and function. Front Neurosc. 2012;6:122
4. Chabot B, Shkreta L. Defective control of pre-messenger RNA splicing in human disease. J Cell Biol. 2016;212:13-27
5. Wang P, Yan B, Guo JT. et al. Structural genomics analysis of alternative splicing and application to isoform structure modeling. Proc Natl Acad Sci USA. 2005;102:18920-5
6. Jurica MS, Moore MJ. Pre-mRNA splicing: awash in a sea of proteins. Mol Cell. 2003;12:5-14
7. Zhou Z, Licklider LJ, Gygi SP. et al. Comprehensive proteomic analysis of the human spliceosome. Nature. 2002;419:182-5
8. Will CL, Luhrmann R. Spliceosomal UsnRNP biogenesis, structure and function. Current opinion in cell biology. 2001;13:290-301
9. Kornblihtt AR, Schor IE, Allo M. et al. Alternative splicing: a pivotal step between eukaryotic transcription and translation. Nature reviews Molecular cell biology. 2013;14:153-65
10. AR MJaK. A rational nomenclature for serine/arginine-rich protein splicing factors (SR proteins). Genes & development. 2010 Dev; 24: 1073-4
11. Huang Y GR, Stevenin J. SR splicing factors serve as adapter proteins for TAP-dependent mRNA export. Mol Cell. 2003;11:837-43
12. AR ZZaK. Involvement of SR proteins in mRNA surveillance. Mol Cell. 2004;16(4):597-607
13. JL LXaM. Inactivation of the SR protein splicing factor ASF/SF2 results in genomic instability. Cell. 2005;122:365-78
14. Sanford JR EJ, Cazalla D. Reversible phos- phorylation differentially affects nuclear and cytoplasmic functions of splicing factor 2/alternative splicing factor. Proc Natl Acad Sci U S A. 2005;102:15042-7
15. Jia R, Li C, McCoy JP. et al. SRp20 is a proto-oncogene critical for cell proliferation and tumor induction and maintenance. Int J Biol Sci. 2010;6:806-26
16. Grosso AR, Martins S, Carmo-Fonseca M. The emerging role of splicing factors in cancer. EMBO reports. 2008;9:1087-93
17. Jumaa H, Nielsen PJ. Regulation of SRp20 exon 4 splicing. Biochim Biophys Acta. 2000;1494:137-43
18. Walsh CM, Suchanek AL, Cyphert TJ. et al. Serine arginine splicing factor 3 is involved in enhanced splicing of glucose-6-phosphate dehydrogenase RNA in response to nutrients and hormones in liver. J Biol Chem. 2013;288:2816-28
19. Sen S, Talukdar I, Webster NJ. SRp20 and CUG-BP1 modulate insulin receptor exon 11 alternative splicing. Mol Cell Biol. 2009;29:871-80
20. Lu GY, Liu ST, Huang SM. et al. Multiple effects of digoxin on subsets of cancer-associated genes through the alternative splicing pathway. Biochimie. 2014;106:131-9
21. Crispino JD, Blencowe BJ, Sharp PA. Complementation by SR proteins of pre-mRNA splicing reactions depleted of U1 snRNP. Science. 1994;265:1866-9
22. Tarn WY, Steitz JA. SR proteins can compensate for the loss of U1 snRNP functions in vitro. Genes Dev. 1994;8:2704-17
23. Xiao SH, Manley JL. Phosphorylation of the ASF/SF2 RS domain affects both protein-protein and protein-RNA interactions and is necessary for splicing. Genes Dev. 1997;11:334-44
24. He X EP, Coon JS, Beck WT. Alternative splicing of the multidrug resistance protein 1/ATP binding cassette transporter subfamily gene in ovarian cancer creates functional splice variants and is associated with increased expression of the splicing factors PTB and SRp20. Clin Cancer Res. 2004;10:4652-60
25. Jia R LC, McCoy JP, Deng CX. et al. SRp20 is a proto-oncogene critical for cell proliferation and tumor induction and maintenance. Int J Biol Sci. 2010;6:806-26
26. Iborra S HM, Jaeger M, Zur Hausen A. et al. Alterations in expression pattern of splicing factors in epithelial ovarian cancer and its clinical impact. Int J Gynecol Cancer. 2013;23:990-6
27. Graveley BR. Alternative splicing: increasing diversity in the proteomic world. Trends in genetics: TIG. 2001;17:100-7
28. Wang H, Zhang CZ, Lu SX. et al. A Coiled-Coil Domain Containing 50 Splice Variant Is Modulated by Serine/Arginine-Rich Splicing Factor 3 and Promotes Hepatocellular Carcinoma in Mice by the Ras Signaling Pathway. Hepatology. 2019;69:179-95
29. Nagano O SH. Mechanism and biological significance of CD44 cleavage. Cancer Sci. 2004;95:930-5
30. Cheng C SP. Regulation of CD44 alternative splicing by SRm160 and its potential role in tumor cell invasion. Mol Cell Biol. 2006;26:362-70
31. Naor D SR, Ish-Shalom D. CD44: structure, function, and association with the malignant process. Adv Cancer Res. 1997;71:241-319
32. Bourguignon LY, Zhu D, Zhu H. CD44 isoform-cytoskeleton interaction in oncogenic signaling and tumor progression. Frontiers in bioscience: a journal and virtual library. 1998;3:637-49
33. D'Ambrogio A, Buratti E, Stuani C. et al. Functional mapping of the interaction between TDP-43 and hnRNP A2 in vivo. Nucleic Acids Res. 2009;37:4116-26
34. Bechara EG, Sebestyen E, Bernardis I. et al. RBM5, 6, and 10 differentially regulate NUMB alternative splicing to control cancer cell proliferation. Mol Cell. 2013;52:720-33
35. Xue B, Krishnamurthy K, Allred DC. et al. Loss of Par3 promotes breast cancer metastasis by compromising cell-cell cohesion. Nature cell biology. 2013;15:189-200
36. McCaffrey LM, Montalbano J, Mihai C. et al. Loss of the Par3 Polarity Protein Promotes Breast Tumorigenesis and Metastasis. Cancer cell. 2016;30:351-2
37. Ke H, Zhao L, Zhang H. et al. Loss of TDP43 inhibits progression of triple-negative breast cancer in coordination with SRSF3. Proceedings of the National Academy of Sciences. 2018;115:E3426-E35
38. Oh S, Shin S, Janknecht R. ETV1, 4 and 5: an oncogenic subfamily of ETS transcription factors. Biochim Biophys Acta. 2012;1826:1-12
39. Britton E, Rogerson C, Mehta S. et al. Open chromatin profiling identifies AP1 as a transcriptional regulator in oesophageal adenocarcinoma. PLoS genetics. 2017;13:1068-79
40. Song X, Wan X, Huang T. et al. SRSF3-Regulated RNA Alternative Splicing Promotes Glioblastoma Tumorigenicity by Affecting Multiple Cellular Processes. Cancer Res. 2019Oct15;79:5288-301
41. Wright JH, Wang X, Manning G. et al. The STE20 kinase HGK is broadly expressed in human tumor cells and can modulate cellular transformation, invasion, and adhesion. Mol Cell Biol. 2003Mar;23:2068-82
42. Lin JC, Lee YC, Tan TH. et al. RBM4-SRSF3-MAP4K4 splicing cascade modulates the metastatic signature of colorectal cancer cell. Biochim Biophys Acta Mol Cell Res. 2018;1865:259-72
43. Ma X, Wang H, Ji J. et al. Hippo signaling promotes JNK-dependent cell migration. Proc Natl Acad Sci U S A. 2017;114:1934-9
44. Buoso E, Galasso M, Ronfani M. et al. Role of spliceosome proteins in the regulation of glucocorticoid receptor isoforms by cortisol and dehydroepiandrosterone. Pharmacol Res. 2017;120:180-7
45. Buoso E, Ronfani M, Galasso M. et al. Cortisol-induced SRSF3 expression promotes GR splicing, RACK1 expression and breast cancer cells migration. Pharmacol Res. 2019;143:17-26
46. Abduljabbar R, Negm OH, Lai CF. et al. Clinical and biological significance of glucocorticoid receptor (GR) expression in breast cancer. Breast cancer research and treatment. 2015;150:335-46
47. Zhang S, Zhao BS, Zhou A. et al. m(6)A Demethylase ALKBH5 Maintains Tumorigenicity of Glioblastoma Stem-like Cells by Sustaining FOXM1 Expression and Cell Proliferation Program. Cancer cell. 2017;31:591-606
48. Kurokawa K AY, Masuda K, Kuwano Y. et al. Downregulation of serine/arginine-rich splicing factor 3 induces G1 cell cycle arrest and apoptosis in colon cancer cells. Oncogene. 2014Mar13;33:1407-17
49. He X, Arslan AD, Pool MD. et al. Knockdown of splicing factor SRp20 causes apoptosis in ovarian cancer cells and its expression is associated with malignancy of epithelial ovarian cancer. Oncogene. 2011;30:356-65
50. De Clercq S. Widespread overexpression of epitope-tagged Mdm4 does not accelerate tumor formation in vivo. Mol Cell Biol. 2010;30:5394-405
51. Carrillo AM BA, Arrate MP, Eischen CM. Mdmx promotes genomic instability independent of p53 and Mdm2. Oncogene. 2015;34:846-56
52. Dewaele M, Tabaglio T, Willekens K. et al. Antisense oligonucleotide-mediated MDM4 exon 6 skipping impairs tumor growth. J Clin Invest. 2016;126:68-84
53. Hofmann TG, Moller A, Sirma H. et al. Regulation of p53 activity by its interaction with homeodomain-interacting protein kinase-2. Nature cell biology. 2002;4:1-10
54. D'Orazi G, Cecchinelli B, Bruno T. et al. Homeodomain-interacting protein kinase-2 phosphorylates p53 at Ser 46 and mediates apoptosis. Nature cell biology. 2002;4:11-9
55. Wei G, Ku S, Ma GK. et al. HIPK2 represses β-catenin-mediated transcription, epidermal stem cell expansion, and skin tumorigenesis. Proc Natl Acad Sci USA. 2007;104:13040-5
56. Winter M, Sombroek D, Dauth I. et al. Control of HIPK2 stability by ubiquitin ligase Siah-1 and checkpoint kinases ATM and ATR. Nature cell biology. 2008;10:812-24
57. Roberts KG MC. Genomics in acute lymphoblastic leukaemia: insights and treatment implications. Nat Rev Clin Oncol. 2015;12:344-57
58. Del Nagro CJ OD, Anzelon AN, Omori SA. et al. CD19 function in central and peripheral B-cell development. Immunol Res. 2005;31:119-31
59. Chung EY PJ, Yu D, Li Y. et al. CD19 is a major B cell receptor-independent activator of MYC-driven B-lymphomagenesis. J Clin Invest. 2012;122:2257-66
60. Rickert RC RK, Roes J. Impairment of T-cell-dependent B-cell responses and B-1 cell development in CD19-deficient mice. Nature. 1995;376:352-5
61. Poe JC M-CV, Kountikov EI, Haas KM. et al. A c-Myc and surface CD19 signaling amplification loop promotes B cell lymphoma development and progression in mice. J Immunol. 2012;189:2318-25
62. David L, Porter MD, Bruce L. et al. Chimeric Antigen Receptor-Modified T Cells in Chronic Lymphoid Leukemia; Chimeric Antigen Receptor-Modified T Cells for Acute Lymphoid Leukemia; Chimeric Antigen Receptor T Cells for Sustained Remissions in Leukemia. The New England journal of medicine. 2016;374:998
63. Kalos M, Levine BL, Porter DL. et al. T cells with chimeric antigen receptors have potent antitumor effects and can establish memory in patients with advanced leukemia. Sci Transl Med. 2011;3(95):95ra73
64. Sotillo E BD, Black KL, Bagashev A. et al. Convergence of Acquired Mutations and Alternative Splicing of CD19 Enables Resistance to CART-19 Immunotherapy. Cancer Discov. 2015Dec;5:1282-95
65. Bugiani M BI, Powers JM, Scheper GC. et al. Leukoencephalopathy with vanishing white matter: a review. J Neuropathol Exp Neurol. 2010;69:987-96
66. Palaniappan A RK, Ramalingam S. Computational identification of novel stage-specific biomarkers in colorectal cancer progression. PLoS One. 2016;11:155-65
67. Gomez E, Pavitt GD. Identification of domains and residues within the epsilon subunit of eukaryotic translation initiation factor 2B (eIF2Bepsilon) required for guanine nucleotide exchange reveals a novel activation function promoted by eIF2B complex formation. Mol Cell Biol. 2000;20:3965-76
68. K BL. Transcriptome-Wide Studies of Hypoxic Cancer Cells Identify Alternative Splicing as a Mechanism to Inhibit Translation. ProQuest. 2017;21:1091-96
69. Long JC, Caceres JF. The SR protein family of splicing factors: master regulators of gene expression. The Biochemical journal. 2009;417:15-27
70. Chen Y, Huang Q, Liu W. et al. Mutually exclusive acetylation and ubiquitylation of the splicing factor SRSF5 control tumor growth. Nat Commun. 2018;9:24-64
71. Yang S, Jia R, Bian Z. SRSF5 functions as a novel oncogenic splicing factor and is upregulated by oncogene SRSF3 in oral squamous cell carcinoma. Biochim Biophys Acta Mol Cell Res. 2018;1865:1161-72
72. Lareau LF, Inada M, Green RE. et al. Unproductive splicing of SR genes associated with highly conserved and ultraconserved DNA elements. Nature. 2007;446:926-9
73. Wynne CL, Vallee RB. Cdk1 phosphorylation of the dynein adapter Nde1 controls cargo binding from G2 to anaphase. The Journal of cell biology. 2018;217:3019-29
74. Wang Z, Chatterjee D, Jeon HY. et al. Exon-centric regulation of pyruvate kinase M alternative splicing via mutually exclusive exons. J Mol Cell Biol. 2012;4:79-87
75. Kuranaga Y, Sugito N, Shinohara H. et al. SRSF3, a Splicer of the PKM Gene, Regulates Cell Growth and Maintenance of Cancer-Specific Energy Metabolism in Colon Cancer Cells. Int J Mol Sci. 2018;19:3012
76. Castella S, Bernard R, Corno M. et al. Ilf3 and NF90 functions in RNA biology. Wiley interdisciplinary reviews RNA. 2015;6:243-56
77. Roberts KG, Mullighan CG. Genomics in acute lymphoblastic leukaemia: insights and treatment implications. Nat Rev Clin Oncol. 2015;12:344-57
78. Duchange N, Pidoux J, Camus E. et al. Alternative splicing in the human interleukin enhancer binding factor 3 (ILF3) gene. Gene. 2000;261:345-53
79. Jia R, Ajiro M, Yu L. et al. Oncogenic splicing factor SRSF3 regulates ILF3 alternative splicing to promote cancer cell proliferation and transformation. RNA. 2019;25:630-44
80. Chen Y, Knosel T, Kristiansen G. et al. Loss of PDCD4 expression in human lung cancer correlates with tumour progression and prognosis. The Journal of pathology. 2003;200:640-6
81. Park SK, Jeong S. SRSF3 represses the expression of PDCD4 protein by coordinated regulation of alternative splicing, export and translation. Biochem Biophys Res Commun. 2016;470:431-8
82. Xiaolong He PLRE, John S. Coon and William T. Beck. Alternative Splicing of the Multidrug Resistance Protein 1/ATP Binding Cassette Transporter Subfamily Gene in Ovarian Cancer Creates Functional Splice Variants and Is Associated with Increased Expression of the Splicing Factors PTB and SRp20. Clinical Cancer Research. July. 2004;10:4652-60
83. Bedard KM, Daijogo S, Semler BL. A nucleo-cytoplasmic SR protein functions in viral IRES-mediated translation initiation. The EMBO journal. 2007;26:459-67
84. Slamon DJ GW, Jones LA, Holt JA. et al. Studies of the HER-2/neu proto-oncogene in human breast and ovarian cancer. Science. 1989;244:707-12
85. Hannah Gautrey CJ, Anna-Lena Dittrich, David Browell. et al. SRSF3 and hnRNP H1 regulate a splicing hotspot of HER2 in breast cancer cells. RNA Biology. 2015 October; 12: 1139-51
86. Jumaa H, Nielsen PJ. The splicing factor SRp20 modifies splicing of its own mRNA and ASF/SF2 antagonizes this regulation. Embo j. 1997;16:5077-85
87. Heiner M HJ, Schreiner S, Hung LH & Bindereif A. HnRNP L-mediated regulation of mammalian alternative splicing by interference with splice site recognition. RNA Biol. 2010;7:56-64
88. Majumder M, Yaman I, Gaccioli F. et al. The hnRNA-binding proteins hnRNP L and PTB are required for efficient translation of the Cat-1 arginine/lysine transporter mRNA during amino acid starvation. Mol Cell Biol. 2009;29:2899-912
89. Shih SC, Claffey KP. Regulation of human vascular endothelial growth factor mRNA stability in hypoxia by heterogeneous nuclear ribonucleoprotein L. J Biol Chem. 1999;274:1359-65
90. Jia R ZS, Liu M, Zhang Y. et al. HnRNP L is important for the expression of oncogene SRSF3 and oncogenic potential of oral squamous cell carcinoma cells. Sci Rep. 2016Nov3;6:35976
91. Elizalde M UR, Azkona M, Latasa MU. et al. Splicing regulator SLU7 is essential for maintaining liver homeostasis. J Clin Invest. 2014;124:2909-20
92. Maddalen Jiménez RU, María Elizalde, María Azkona. et al. Splicing events in the control of genome integrity: role of SLU7 and truncated SRSF3 proteins. Nucleic Acids Research. 2019;47:3450-66
93. Guo J, Jia J, Jia R. PTBP1 and PTBP2 impaired autoregulation of SRSF3 in cancer cells. Sci Rep. 2015;5:14548
94. Liang YC, Lin WC, Lin YJ. et al. The impact of RNA binding motif protein 4-regulated splicing cascade on the progression and metabolism of colorectal cancer cells. Oncotarget. 2015;6:38046-60
95. Kristensen LS, Hansen TB, Veno MT. et al. Circular RNAs in cancer: opportunities and challenges in the field. Oncogene. 2018;37:555-65
96. Barbagallo D, Condorelli A, Ragusa M. et al. Dysregulated miR-671-5p / CDR1-AS / CDR1 / VSNL1 axis is involved in glioblastoma multiforme. Oncotarget. 2016;7:4746-59
97. Ragusa M, Barbagallo C, Statello L. et al. Non-coding landscapes of colorectal cancer. World journal of gastroenterology. 2015;21:11709-39
98. Barbagallo D, Caponnetto A, Cirnigliaro M. et al. CircSMARCA5 Inhibits Migration of Glioblastoma Multiforme Cells by Regulating a Molecular Axis Involving Splicing Factors SRSF1/SRSF3/PTB. Int J Mol Sci. 2018;19:480
99. Belkhiri A, Zaika A, Pidkovka N. et al. Darpp32: a novel antiapoptotic gene in upper gastrointestinal carcinomas. Cancer Res. 2005;65:6583-92
100. Mukherjee K, Peng D, Brifkani Z. et al. Dopamine and cAMP regulated phosphoprotein MW 32 kDa is overexpressed in early stages of gastric tumorigenesis. Surgery. 2010;148:354-63
101. Zhu S, Belkhiri A, El-Rifai W. DARPP-32 increases interactions between epidermal growth factor receptor and ERBB3 to promote tumor resistance to gefitinib. Gastroenterology. 2011;141:1738-48
102. Zhu S, Hong J, Tripathi MK. et al. Regulation of CXCR4-mediated invasion by DARPP-32 in gastric cancer cells. Molecular cancer research: MCR. 2013;11:86-94
103. Zhu S, Chen Z, Katsha A. et al. Regulation of CD44E by DARPP-32-dependent activation of SRp20 splicing factor in gastric tumorigenesis. Oncogene. 2016;35:1847-56
104. Kumar D, Das M, Sauceda C. et al. Degradation of splicing factor SRSF3 contributes to progressive liver disease. J Clin Invest. 2019;130:4477-91
105. Rabut G, Peter M. Function and regulation of protein neddylation. 'Protein modifications: beyond the usual suspects' review series. EMBO reports. 2008;9:969-76
106. Xirodimas DP. Novel substrates and functions for the ubiquitin-like molecule NEDD8. Biochemical Society transactions. 2008;36:802-6
107. Tillemans V, Dispa L, Remacle C. et al. Functional distribution and dynamics of Arabidopsis SR splicing factors in living plant cells. The Plant journal: for cell and molecular biology. 2005;41:567-82
108. Caceres JF, Screaton GR, Krainer AR. A specific subset of SR proteins shuttles continuously between the nucleus and the cytoplasm. Genes Dev. 1998;12:55-66
109. Long Y, Sou WH, Yung KWY. et al. Distinct mechanisms govern the phosphorylation of different SR protein splicing factors. J Biol Chem. 2019;294:1312-27
110. Wang Q, Chikina MD, Pincas H. et al. Homer1 alternative splicing is regulated by gonadotropin-releasing hormone and modulates gonadotropin gene expression. Mol Cell Biol. 2014;34:1747-56
111. Sen S, Langiewicz M, Jumaa H. et al. Deletion of serine/arginine-rich splicing factor 3 in hepatocytes predisposes to hepatocellular carcinoma in mice. Hepatology. 2015;61:171-83
112. Sen S, Jumaa H, Webster NJ. Splicing factor SRSF3 is crucial for hepatocyte differentiation and metabolic function. Nature communications. 2013;4:1336
113. Kensler TW, Qian GS, Chen JG. et al. Translational strategies for cancer prevention in liver. Nature reviews Cancer. 2003;3:321-9
114. Elena Sotillo DMB, Kathryn LB, Asen Bagashev. et al. Convergence of Acquired Mutations and Alternative Splicing of CD19 Enables Resistance to CART-19 Immunotherapy. Cancer Discov. 2015 December 5; 12: 1282-95
115. Brady LK, Wang H, Radens CM. et al. Transcriptome analysis of hypoxic cancer cells uncovers intron retention in EIF2B5 as a mechanism to inhibit translation. PLoS Biol. 2017;15:1544-54
Author contact
Corresponding author: Dr. Lin Fu, E-mail: smilefulincom or fulinedu.cn

 Global reach, higher impact
Global reach, higher impact