Impact Factor
ISSN: 1837-9664
J Cancer 2022; 13(4):1203-1213. doi:10.7150/jca.66674 This issue Cite
Research Paper
3BDO inhibits the proliferation, epithelial-mesenchymal transition (EMT), and stemness via suppressing survivin in human glioblastoma cells
Institute of Neuroscience, Department of Neurosurgery, the Second Affiliated Hospital of Guangzhou Medical University, Guangzhou 510260 China
*contribute equally
Abstract
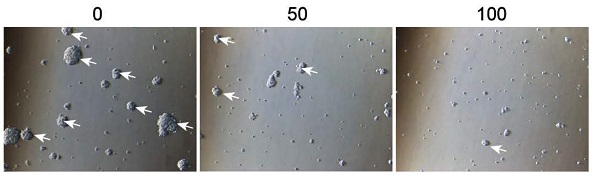
Background: Glioblastoma (GBM) is a tumor of the central nervous system with an extremely poor prognosis. Stemness and EMT play important roles in GBM progression. 3-benzyl-5-((2-nitrophenoxy) methyl) dihydrofuran-2(3H)-one (3BDO), an autophagy inhibitor, has been reported to exert anti-cancer activities on lung carcinoma. However, the effects of 3BDO on GBM remain unknown. Therefore, the purpose of this study was to explore the effects of 3BDO on GBM and to investigate the underlying molecular mechanisms.
Method: CCK-8 experiments and clone formation assays were conducted to determine the level of cell proliferation. Transwell assay was conducted to examine cell migration and invasion abilities. Western blotting and immunofluorescence staining were used to analyze protein expression levels. A xenograft mouse model was used to evaluate the effect of 3BDO in vivo.
Results: We found that 3BDO inhibited U87 and U251 cell proliferation in a dose-dependent manner. Additionally, 3BDO decreased the degree of sphere formation and levels of stemness markers (sox2, nestin, and CD133) in GSCs. 3BDO also inhibited migration and invasion abilities and suppressed EMT markers (N-cadherin, vimentin, and snail) in GBM cells. Moreover, we found that 3BDO downregulated the expression of survivin in both GBM cells (U87, U251) and GSCs. Furthermore, overexpression of survivin decreased the therapeutic effect of 3BDO on EMT, invasion, migration, and proliferation of GBM cells, as well as decreased the stemness of GSCs. Finally, we demonstrated that 3BDO could inhibit tumor growth in a tumor xenograft mouse model constructed using U87 cells. Similar to the in vitro findings, 3BDO decreased the expression of survivin, EMT makers, and the degree of stemness in vivo.
Conclusions: Our results demonstrate that 3BDO can repress GBM both in vitro and in vivo via downregulating survivin-mediated stemness and EMT.
Keywords: 3BDO, glioblastoma, EMT, GSC, survivin

 Global reach, higher impact
Global reach, higher impact