Impact Factor
ISSN: 1837-9664
J Cancer 2022; 13(4):1252-1260. doi:10.7150/jca.66501 This issue Cite
Research Paper
MAGEA1 and hTERT Peptide Treatment Improves the Potency of The Dendritic Cell- Cytotoxic T Lymphocytes (DC-CTL) Immunotherapy in DAC Treated Acute Myeloid Leukemia
1. Department of Hematology and Oncology, Shenzhen University General Hospital, Shenzhen University Clinical Medical Academy, Shenzhen University; Shenzhen Key Laboratory, Hematology Institution of Shenzhen University, Shenzhen 518055, China
2. International Cancer Center, Health Science Center, Shenzhen University, Shenzhen 518037, China
3. Shenzhen Haoshi Biotechnology Co, Ltd. Shenzhen 518000, China
Abstract
Background: Acute myeloid leukemia (AML) is a type of heterogenous malignant hematological disorder. Recently developed immunotherapies such as chimeric antigen receptor T cell (CAR-T) do not demonstrated promising therapeutic results due to the off-target effect. The Dendritic cell-cytotoxic T lymphocyte adoptive immunotherapy (DC-CTL) is one of the recently developed immunotherapies. One of the reasons that DC-CTL does not work well in AML is the lack of antigens with high binding affinity, high antigen presentation potency, and the specificity to AML cells.
Methods: DAC was used to treat AML cells to find overexpressed CTAs upon DAC treatment. The overexpression was confirmed at both mRNA and protein level by realtime PCR and western blotting. Peptides was designed by using the NetMHCpan database and EPIP based on the out-screened protein sequences. The peptides were then used to pulse DC-CTL coculture in vitro and tested the cytotoxicity of CTLs in vitro and their cancer inhibition potency in vivo.
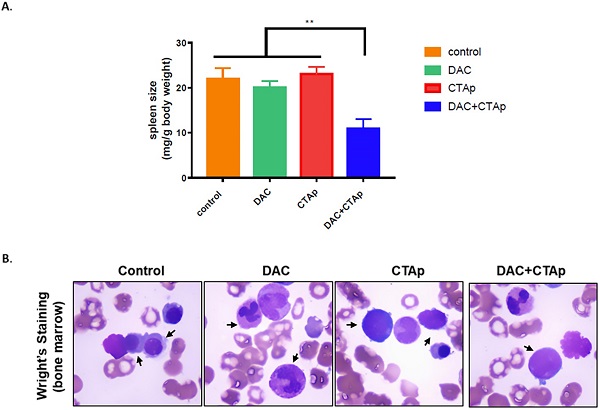
Results: Two cancer testis antigen (CTA) proteins, MAGEA1 and hTERT, was up-regulated in DAC treated AML cells. DC cells pulsed by the antigen peptides designed based on the sequence of these two proteins demonstrated increased potency to stimulate CTL cells in terms of cytokines secretion. These cytokines included IFN-γ, IL-6, and TNF-α. Moreover, enhanced in vitro cytotoxicity was found in CTL cells treated with peptide pulsed DC cells. AML progress was inhibited by CTA peptides pulsed DC-CTL in a mouse AML model.
Conclusions: MAGEA1 and hTERT could possibly serve as specific tumor antigens upon DAC treatment, providing potential targets for the development of immunotherapies for AML in the future.
Keywords: acute myeloid leukemia (AML), dendritic cell (DC), cytotoxic T lymphocyte(CTL)

 Global reach, higher impact
Global reach, higher impact