Impact Factor ISSN: 1837-9664
J Cancer 2022; 13(4):1282-1288. doi:10.7150/jca.67523 This issue Cite
Research Paper
A comparative study of efficacy and safety of transarterial chemoembolization with CalliSpheres and conventional transarterial chemoembolization in treating unresectable intrahepatic cholangiocarcinoma patients
1. Department of Radiology, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430022, China.
2. Hubei Province Key Laboratory of Molecular Imaging, Wuhan 430022, China.
3. Department of Radiology, Union Hospital, Tongji Medical College and Wuhan National Laboratory for Optoelectronics, Huazhong University of Science and Technology.
#These authors contributed equally to this work.
Abstract
Objective: Previous studies reported that drug-eluting beads transarterial chemoembolization (DEB-TACE) with CalliSpheres is effective and safe to treat hepatocellular carcinoma patients and metastatic liver cancer patients, however few studies reported its clinical application in intrahepatic cholangiocarcinoma (ICC) patients. Therefore, this study aimed to compare the efficacy between DEB-TACE versus conventional transarterial chemoembolization (cTACE) in unresectable ICC patients.
Methods: Between January 2016 and June 2020, 89 patients with unresectable ICC were retrospectively analyzed, and enrolled into DEB-TACE group (N=40) and cTACE group (N=49) based on the transarterial treatment. Treatment response was assessed according to modified Response Evaluation Criteria in Solid Tumors (mRECIST). Time to progression (TTP) and overall survival (OS) were analyzed by using the Kaplan-Meier curve. Factors affecting OS and TTP were determined by Cox's proportional hazards regression model.
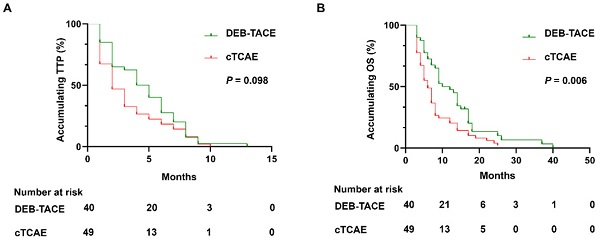
Results: DEB-TACE group showed higher DCR (87.5% vs. 65.3%, P=0.011), while similar ORR (67.5% vs. 57.1%, P=0.317) compared to cTACE group. Furthermore, DEB-TACE group had longer OS (median 10 months vs 6 months, P=0.006), while similar TTP compared to cTACE group (median 4 months vs 2 months, P=0.098). After adjustment by multivariant Cox's regression, DEB-TACE (versus cTACE) independently correlated with longer OS (P=0.031). Further subgroup analyses displayed that OS was prolonged in DEB-TACE group compared to cTACE group in patients with multiple tumors (P=0.032) and patients with no lymph node metastasis (P=0.023). Apart from abdominal pain, no difference of adverse events between the two groups was observed. There was no difference in liver function (Bilirubin, Albumin, Prothrombin time) before and after treatment (4 weeks) in both groups.
Conclusion: In patients with unresectable ICC, DEB-TACE significantly improved the OS when compared with cTACE and was well tolerated.
Keywords: Intrahepatic cholangiocarcinoma, drug-eluting beads transarterial chemoembolization, overall survival, time to progression, treatment response

 Global reach, higher impact
Global reach, higher impact