3.2
Impact Factor
ISSN: 1837-9664
J Cancer 2022; 13(4):1370-1384. doi:10.7150/jca.66978 This issue Cite
Research Paper
Highly Potent Immunotoxins Targeting the Membrane-distal N-lobe of GPC3 for Immunotherapy of Hepatocellular Carcinoma
1. College of Life Science and Technology, Huazhong Agricultural University, Wuhan, Hubei 430070, China.
2. Beijing Advanced Innovation Center for Structural Biology, School of Life Sciences, Tsinghua University, No. 30 Shuangqing Road, Beijing 100084, China.
3. Department of Internal Medicine-Oncology, Hubei Cancer Hospital, Wuhan, Hubei 430070, China.
4. CAS Key Laboratory of Quantitative Engineering Biology, Shenzhen Institute of Synthetic Biology, Shenzhen Institutes of Advanced Technology, Chinese Academy of Sciences, Shenzhen, Guangdong 518055, China.
5. College of Biomedicine and Health, Huazhong Agricultural University, Wuhan, Hubei 430070, China.
Abstract
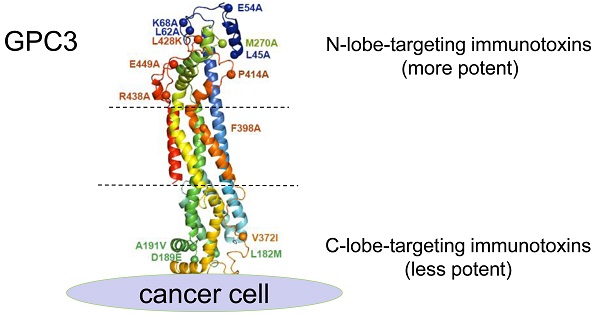
Glypican-3 (GPC3) has become a compelling target for immunotherapy of hepatocellular carcinoma, including antibody-drug conjugate (ADC), and ADC-like immunotoxin. To investigate the impact of epitopes on the potency of ADCs, current study generated a large panel of chicken monoclonal antibodies (mAbs) that targeted 12 different and over-lapping epitopes on GPC3. These mAbs demonstrated a very high affinity with Kd values in the range of 10-9-10-14 M, and the highest affinity (Kd value of 0.0214 pM) was 40-fold higher than the previously generated high-affinity mAb YP7 (Kd value of 0.876 nM). Additionally, these mAbs exhibited excellent thermostability with Tm values in the range of 45-82 °C. As a proof-of-concept study for ADC, we made immunotoxins (scFv fused with PE24, the 24-kDa cytotoxic domain of Pseudomonas exotoxin A) based on these mAbs, and we found that immunotoxins targeting the N-lobe of GPC3 were overall much more potent than those targeting the C-lobe and other locations. One representative N-lobe-targeting immunotoxin J80A-PE24 demonstrated 3 to 13-fold more potency than the hitherto best immunotoxin HN3-PE24 that was previously developed. J80A-PE24 could suppress tumor growth much greater than HN3-PE24 in a xenograft mouse model. Combination of J80A-PE24 with an angiogenesis inhibitor FGF401 showed additive effect, which dramatically shrank tumor growth. Our work demonstrated that, due to high affinity, excellent thermostability and potency, chicken mAbs targeting the N-lobe of GPC3 are appealing candidates to develop potent ADCs for immunotherapy of liver cancer.
Keywords: GPC3, immunotoxin, monoclonal antibody, epitope mapping, hepatocellular carcinoma

 Global reach, higher impact
Global reach, higher impact