3.2
Impact Factor
ISSN: 1837-9664
J Cancer 2022; 13(6):1725-1733. doi:10.7150/jca.69449 This issue Cite
Research Paper
Hinokitiol impedes tumor drug resistance by suppressing protein kinase B/mammalian targets of rapamycin axis
1. Division of Gastroenterology, Department of Internal Medicine, Kaohsiung Armed Forces General Hospital, Kaohsiung, Taiwan.
2. Department of Biological Sciences, National Sun Yat-sen University, Kaohsiung 80424, Taiwan.
3. Department of Medical Imaging and Radiological Sciences, Kaohsiung Medical University, Kaohsiung, Taiwan.
4. Department of Medical Education and Research, Kaohsiung Veterans General Hospital, Kaohsiung, Taiwan.
5. Department of Medical Research, China Medical University Hospital, China Medical University, Taichung 404, Taiwan.
6. Department of Medical Laboratory Science and Biotechnology, Kaohsiung Medical University, Kaohsiung 80708, Taiwan.
7. International PhD Program for Science, National Sun Yat-sen University, Kaohsiung 80424, Taiwan.
8. Aerosol Science Research Center, National Sun Yat-sen University, Kaohsiung, Taiwan, 80424, Taiwan.
Abstract
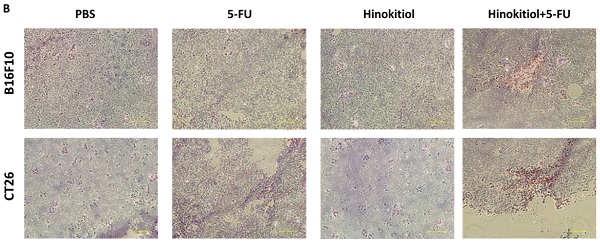
Chemotherapy is a treatment method commonly used for cancer and that patients showing low to no response to the treatment often developed drug resistance via multiple mechanisms. Natural products have been shown to reduce tumor drug resistance. Hinokitiol, a natural tropolone derivative, has potential as an antitumor agent. To improve the efficacy and safety of hinokitiol, a further understanding of hinokitiol interactions with the tumor microenvironment is necessary. The presence of plasma membrane multidrug resistance protein P-glycoprotein (P-gp) is favorable for tumor cells to elicit chemotherapeutic resistance. Here, we showed that hinokitiol dose-dependently decreased P-gp expression and suppressed the P-gp-driven efflux activity based on Rhodamine 123 assay. The protein expression levels of phosph-protein kinase B (P-AKT), phosph-mammalian targets of rapamycin (P-mTOR), and phosph-p70 ribosomal s6 kinase (P-p70s6K) in tumor cells were likewise reduced after hinokitiol treatment. The transfection of cells with active P-AKT rescued hinokitiol-induced downregulation of P-gp, suggesting the involvement of Akt/mTOR/p70s6K signaling in P-gp expression. Our results showed that hinokitiol can chemosensitize cancer cells. These findings indicate that hinokitiol could enhance 5-Fluorouracil therapeutic effects in murine B16F10 and CT26 tumor cells via downregulation of the AKT/mTOR pathway.
Keywords: Hinokitiol, P-glycoprotein, 5-Fluorouracil, tumor, combination therapy

 Global reach, higher impact
Global reach, higher impact