3.2
Impact Factor
ISSN: 1837-9664
J Cancer 2022; 13(6):1945-1957. doi:10.7150/jca.65017 This issue Cite
Research Paper
LncRNA T-UCR Uc.339/miR-339/SLC7A11 Axis Regulates the Metastasis of Ferroptosis-Induced Lung Adenocarcinoma
1. Division of Gastroenterology, Ganzhou People's Hospital, the Affiliated Ganzhou Hospital of Nanchang University, Ganzhou, Jiangxi 341000, China
2. Department of Hematology, Wenzhou Central Hospital, Theorem Clinical College, Wenzhou Medical University, Wenzhou, Zhejiang 325000, China.
3. Department of Oncology, Ganzhou People's Hospital, the Affiliated Ganzhou Hospital of Nanchang University, Ganzhou, Jiangxi 341000, China.
Abstract
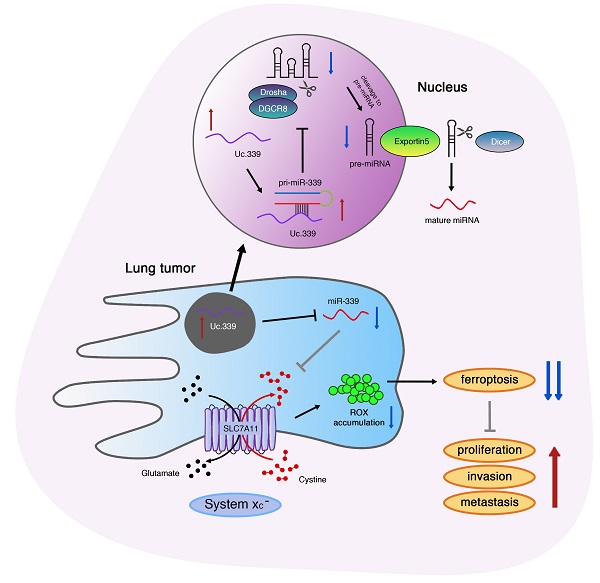
Lung adenocarcinoma progression is closely linked to ferroptosis suppression. Emerging studies have found that the expression of its related gene SLC7A11 may be regulated by LncRNA. However, the mechanism of LncRNA in affecting the development of SLC7A11-mediated lung adenocarcinoma remains unclear. Here, we identified a Uc.339/miR-339/SLC7A11 axis that involves LncRNA T-UCR Uc.339-mediated repression of miR-339 and affects the expression of SLC7A11 to participate in tumor metastasis and development. In this study, we identified Uc.339 as upregulated in patients with lung adenocarcinoma. RAP-qPCR proved that LncRNA Uc.339 competitively binds to pri-miR-339 and inhibits the production of mature miR-339. The interaction between miR-339 and SCL7A11 was confirmed by luciferase reporter assay. The Uc.339/miR-339/SLC7A11 axis regulated the proliferation, migration and invasion of A549 and H1299cells in vitro by affecting ferroptosis. Finally, in mouse xenograft models, knocking down Uc.339 in LLC cells was able to inhibits tumor growth by blocking the axis of Uc.339/miR-339/SLC7A11 in vivo, but miR-339 inhibitors could reverse this inhibition. Taken together, our results uncovered a Uc.339/miR-339/SLC7A11 axis that leads to defects in the ferroptosis in lung cancer, and constitutes a potential mechanism that drives the metastasis of lung adenocarcinoma.
Keywords: Uc.339, LncRNA T-UCR, miR-339, lung adenocarcinoma, ferroptosis

 Global reach, higher impact
Global reach, higher impact