Impact Factor
ISSN: 1837-9664
J Cancer 2022; 13(6):2029-2039. doi:10.7150/jca.63533 This issue Cite
Research Paper
KLF9 regulates miR-338-3p/NRCAM axis to block the progression of osteosarcoma cells
1. Department of Orthopedics, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430022, China.
2. Department of Orthopedics, Clinical Medical College of Yangzhou University, Northern Jiangsu People's Hospital, Yangzhou, Jiangsu, 225001, China.
*Yunlu Liu and Kuijing Han contributed equally to this work.
#These authors also contributed equally to this work.
Abstract
Background: MiR-338-3p is revealed to serve as a tumor suppressor in several carcinomas. Whereas, the effect of miR-338-3p in the progression of osteosarcoma has not been explored. The aim of this paper was to analyze the functional influences of miR-338-3p on osteosarcoma progression and the potential mechanism.
Methods: The expression of genes and miRNAs in osteosarcoma cells was assessed via western blotting or quantitative reverse transcription-polymerase chain reaction (qRT-PCR). Osteosarcoma cellular proliferation was explored by MTT and EdU incorporation assay. Osteosarcoma cellular migratory and invasive capacity was explored by wound-healing and transwell assay. Bioinformatics approaches were adopted to predict target genes. The relationships between miR-338-3p and neuron‑glial‑related cell adhesion (NRCAM), between kruppel-like factor 9 (KLF9) and miR-338-3p were verified by dual-luciferase reporter assay.
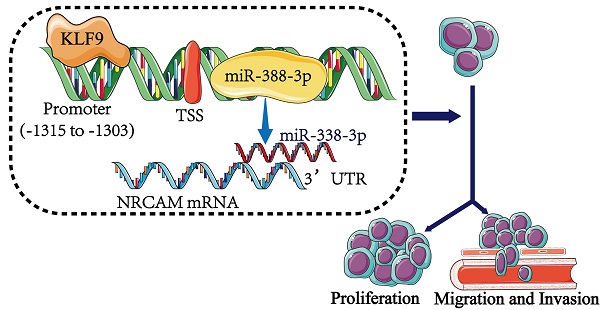
Results: We found that miR-338-3p was reduced in osteosarcoma and that higher expression of miR-338-3p suppressed proliferative, invasive and migratory ability of osteosarcoma cells. Furthermore, the result showed that overexpression of NRCAM could reduce the anti-tumor role of miR-338-3p in osteosarcoma cells. In addition, we found that overexpression of KLF9 could enhance the expression level of miR-338-3p in osteosarcoma cells.
Conclusion: The KLF9/miR-338-3p/NRCAM axis played a significant role in regulating osteosarcoma progression, which may become a promising therapeutic method for osteosarcoma.
Keywords: MiR-338-3p, KLF9, NRCAM, osteosarcoma, GEO, TCGA.

 Global reach, higher impact
Global reach, higher impact