Impact Factor ISSN: 1837-9664
J Cancer 2022; 13(9):2933-2944. doi:10.7150/jca.71401 This issue Cite
Review
Role and mechanism of miR-211 in human cancer
1. Department of Gynecology, Zhangjiagang First People's Hospital, Zhangjiagang Affiliated Hospital of Soochow University, Zhangjiagang, Jiangsu, China; Department of Oncology, The Affiliated Jiangning Hospital with Nanjing Medical University, Nanjing, Jiangsu, China.
2. Department of Oncology, The Affiliated Jiangning Hospital with Nanjing Medical University, Nanjing, Jiangsu, China.
Received 2022-1-25; Accepted 2022-6-26; Published 2022-7-18
Abstract
MircoRNA (miRNA), which are a group of small, and highly conserved non-coding RNA consisting of 18-25 nucleotides, can modulate gene expression at post-transcriptional level, through complementary binding to the 3ʹ-untranslated region (3ʹ-UTR) of numerous target genes. Emerging evidence indicates that miRNAs play critical roles in tumorigenesis and progression of cancer. Among them, miR-211 has been extensively studied in multiple cancers. The expression of miR-211 significantly varies with cancer types and may be used as a potential prognostic marker for cancer. MiR-211 can regulate multiple biological processes in cancer, including proliferation, apoptosis, metastasis and drug resistance. Additionally, several factors may contribute to the dysregulation of miR-211 in cancer. Consequently, this review aims to discuss the novel findings that highlight latent value of miR-211 in the prognosis assessment and treatment of cancer.
Introduction
In recent years, research has identified microRNAs (miRNAs) as important endogenous regulators of gene expression in all tissues [1]. MiRNAs are small, highly conserved non-coding RNAs which typically consists of 18-25 nucleotides, and they regulate posttranscriptional gene expression. MiRNAs do not entail complete complementarity for target recognition, hence, a miRNA can bind to several target mRNAs by recognizing complementary sites in 3ʹ-untranslated regions (3ʹ-UTR) or in some situations at their 5ʹ-UTR [2, 3]. However, the “seed sequence” of the miRNA must perfectly match its target, the “seed sequence,” is the most important sequence for target recognition, is nucleotides 2-8 of the miRNA [1]. Dysregulation of one miRNA may regulate various genes expression through binding to numerous sites which have same seed matches [4]. By this means, they are involved in various biological processes, for instance, metabolic processes, cell proliferation, apoptosis, metastasis and differentiation [5-7].
A large number of researches identify that miRNAs are related to the pathogenesis of many diseases, particularly cancer [8, 9]. Depending on the nature of their target genes, miRNAs can function as tumor suppressors or oncogenes (also known as oncomiR) [10]. OncomiRs can inhibit the expression of tumor suppressive target mRNAs, and are frequently upregulated in the disease. Conversely, tumor suppressive miRNAs negatively regulating oncogenic targets, and are downregulated in cancer [11]. Furthermore, one miRNA can act as a tumor suppressor in one cancer and as an oncogene in another [12, 13], indicating that conducting research on the same miRNA in different type of cancers is very significant.
Among various miRNAs that have been reported to be dysregulated in cancer, miR-211 has been recognized as one of the miRNAs that play a crucial role in cancer pathogenesis. Regarding its structure, miR-211 is localized in intron 6 of Trpm1 gene at 15q13-q14 [14]. The miR-211 hairpin is further cleaved into the “guide strand” miR-211-5p and the sister “passenger” strand miR-211-3p. Here, we will describe the different roles, even opposing that miR-211 can play, depending on the tissue context or the cancer type. Most of studies have focused on miR-211-5p and only a few on miR-223-3p; however, in most cases, the strand is not specified. Furthermore, the role of miR-211 in cancer is closely related to its impact on various tumor biological processes, such as cell proliferation [15], apoptosis [16], epithelial-mesenchymal transition (EMT) [17], drug resistance [18], and metastasis [19].
In this article, we will mainly focus on summarizing the mechanisms and functions of miR-211 in tumors and aim to improve our understanding of its significance in the prognosis and treatment of tumors.
Expression and prognostic value of miR-211 in cancer
The miR-211 expression in cancer
A great number of researches investigate the expression pattern of miR-211 in the tumor and non-tumor tissues of various cancers (Table 1). The expression of miR-211 is found to be dysregulated in multiple cancers, although upregulation or downregulation is differentially observed, depending on the particular cancer type. In detail, miR-211 is significantly decreased in melanomas compared to melanocytes [20-24]. Downregulated miR-211 is observed in papillary thyroid cancer [25, 26]. The expression of miR-211 is also greatly downregulated in hepatocellular carcinoma (HCC) [15, 27-30], and gastric cancer [31, 32]. Various researches indicate that miR-211 is reduced in renal cell carcinoma (RCC) [33, 34], bladder cancer (BC) [35], ovarian cancer (OC) [36, 37] and cervical cancer [38].
Expression patterns and prognostic value of miR-211 in various types of cancer
| System | Type | Expression | Prognostic value | Reference |
|---|---|---|---|---|
| Respiratory system | NSCLC | Downregulation | Higher lymph node metastasis | [45] |
| Upregulation | / | [43,44] | ||
| Skeletal system | Osteosarcoma | Upregulation | / | [39] |
| Chondrosarcoma | Upregulation | Higher Histological stage, poorer overall survival (OS) | [40] | |
| Digestive system | OSCC | Upregulation | The most advanced nodal metastasis, vascular invasion, and poor prognosis | [14] |
| Gastric cancer | Downregulation | Distant metastasis, lymph node metastasis and poor outcome | [31,32] | |
| HCC | Downregulation | Poor OS | [15,27-30] | |
| Pancreatic cancer | Downregulation | Shorter median OS and higher metastatic ability | [48,49] | |
| CRC | Downregulation | Lymph node metastasis, distant metastasis, and poor cancer stage | [42] | |
| Urinary system | RCC | Downregulation | / | [33,34] |
| Bladder cancer | Downregulation | Poor TNM stage, lymph nodes metastasis and poor OS | [35] | |
| Reproductive system | Ovarian cancer | Downregulation | Shorter OS | [18, 36,37] |
| Cervical cancer | Downregulation | / | [38] | |
| Others | Breast cancer | Downregulation | Poor TNM stage, lymph nodes metastasis and poor OS | [17,47] |
| Upregulation | brain metastasis and poor survival | [46] | ||
| Thyroid cancer | Downregulation | / | [25,26] | |
| Melanoma | Downregulation | / | [20-24] | |
| Upregulation | / | [41] |
NSCLC: non-small cell lung cancer; OSCC: oral squamous cell carcinoma; CRC: colorectal cancer; HCC: hepatocellular carcinoma; RCC: renal cell carcinoma.
However, the expression of miR-211 is upregulated in osteosarcoma and chondrosarcoma which are tumors Originating in the skeletal system [39, 40]. MiR-211 expression is increased in vemurafenib-resistant melanoma cells [41]. In oral squamous cell carcinoma (OSCC), enforced miR-211 expression is observed [14]. Similarly, Upregulated miR-211 is also observe in colorectal cancer (CRC) [42].
In addition, there is still controversy over the expression of miR-211 in non-small cell lung cancer (NSCLC) [43-45]. One study suggests that miR-211 significantly increased in triple-negative breast cancer cells (TNBC) brain metastatic tumors in vitro and in vivo [46]. However, miR-211 is observes decreased in breast cancer in the other two studies [17, 47]. Therefore, the expression level of miR-211 varies with cancer type and may be strongly related to tumor heterogeneity and the different stages of tumor progression.
The prognostic value of miR-211 in cancer
It has been reported that the abnormal expression of mir-211 can be found in a variety of cancers and can be used to predict the prognosis of cancer (Table 1). Higher miR-211 expression is associated with the most advanced nodal metastasis, vascular invasion, and poor prognosis of oral carcinoma [14]. In pancreatic ductal adenocarcinoma (PDAC), the result illustrates that low miR-211 is related to higher metastatic ability and patients with decreased miR-211 have shorter median overall survival (OS) [48, 49]. Lower miR-211 expression is positively associated with distant metastasis, lymph node metastasis, and poor prognosis in gastric cancer patients [32]. The expression of miR-211 is reversely correlated with lymph node metastasis, distant recurrence, as well as clinical stage in patients with CRC [42]. In HCC, patients with downregulated miR-211 have poor OS [15]. Survival analysis indicated that low miR-211 expression is associated with shorter survival time in patients with OC [18, 37]. In BC, low miR-211 expression is associated with poor TNM stage, lymph nodes metastasis and poor OS and miR211 serve as an independent biomarker for predicting overall survival of BC patients [35]. High miR-211 level is associated with advancement of histological grade and MSTS stage in chondrosarcoma, and is identified as a predictor for poorer OS of chondrosarcoma patients [40]. Besides, in TNBC, high plasma miR-211 levels are associated with brain metastasis and poor survival of patients. The research demonstrates that high plasma levels of miR-211 can be used as a predictor of brain metastasis and prognosis in TNBC [46].
Biological role of miR-211 in cancer
Proliferation and apoptosis
MiR-211 has been confirmed in a variety of cancers to regulate cell proliferation and apoptosis by targeting downstream targets (Table 2). Previous studies have shown that IL-10 promotes the cell proliferation and metastasis in melanoma [50]. IL-10 promotes downstream signaling pathways through binding to the IL-10 receptor (IL-10R), which consists of two different chains (IL-10Rα and IL-10Rβ). In melanoma, miR-211 inhibits cell proliferation by targeting IL-10Rα [51]. Upregulated miR-211 suppresses anchorage-independent colony formation in melanoma [24]. MiR-211 is confirmed to inhibit cells proliferation in thyroid cancer [26] and cervical cancer [38] by regulating secreted protein acidic and rich in cysteine (SPARC), which is an extracellular matrix glycoprotein. SRY-related HMG box transcription factor 11 (SOX11), a member of the SOX transcription factors, promotes thyroid cancer cells proliferation and is downregulated by miR-211 [25]. It is reported that miR-211 inhibits the proliferation of triple-negative breast cancer cells (TNBC) [47]. Sirtuin 1(SIRT1), which is a conserved NAD-dependent deacetylase, mediates deacetylation of p53 and regulates cell survival and apoptosis by this mechanism and thus potentially affects tumorigenesis [52]. By targeting SIRT1, miR-211 reduces breast cancer cell viability and induces apoptosis [16]. SRC Kinase Signaling Inhibitor 1 (SRCIN1) is identified to be regulator for affecting cell proliferation and migration in lung cancer [53]. MiR-211 is indicated to regulate the expression of SRCIN1 in NSCLC, thus attenuating the proliferation of cancer cells [54]. Upregulated miR-211-3p inhibits the proliferation of NSCLC cells by suppressing Zinc-figure protein 217 (ZNF217) [45]. ZNF217 is known as a member of the Kruppel-like family of transcriptional factors and works as an important effector stimulating oncogenicity during multiple cancer processes [55]. Ezrin is a membrane cytoskeleton cross-linker protein, and Ezrin phosphorylation regulates cell migration, mechanical properties, and cytoskeletal organization [56]. Overexpressed miR-211 significantly decreases cell proliferation in tongue cancer via targeting Ezrin/Fak/Src signaling [57]. MiR-211 inhibits HCC cells proliferation by downregulating special AT-rich sequence-binding protein-2 (SATB2), which is upregulated in HCC [27]. ZEB2 acts as an oncogene to promote cells proliferation and inhibit cells apoptosis in HCC, and is regulated by miR-211[28]. Moreover, miR-211 suppresses HCC cell proliferation by targeting Acyl‑CoA Synthetase Long Chain Family Member 4 (ACSL4), which can promote the malignant phenotype in HCC [15]. Overexpressed miR-211 suppresses HCC cell proliferation and angiogenesis by targeting Fork head box D1 (FOXD1) [29]. FOXD1 dysfunction is linked to different pathologies and is known as one of the mediators for angiogenesis in various types of tumors [58]. Some studies have indicated that the single-nucleotide polymorphism (SNP) is related to colon cancer occurrence and development [59]. SNP of rs187960998 in miR-211 inhibits colon cancer cell proliferation by upregulating chromodomain-helicase-DNA binding protein 5 (CHD5), which acts as a tumor suppressive gene [60]. SRY-related HMG box 4 (SOX4) is a member of the C subgroup of the SOX transcription factor family, and is a key transcription factor involved in a number of development procedures, including tumorigenesis [61]. MiR-211 inhibits cells proliferation by reducing SOX4 in gastric cancer [31] and cervical cancer [30]. MiR-211 may be a tumor suppressor in RCC to attenuate the proliferation and induce cell apoptosis of cancer cells [34]. Upregulated miR-211 obviously suppresses cells proliferation and promotes cell apoptosis via downregulating histone deacetylase9 (HDAC9) in bladder cancer [35]. HDAC9 as a target of miRNAs is involved in the progression of carcinogenesis through posttranscriptional regulation [62]. MiR-211 is reported as a tumor suppressor to suppress proliferation and induce apoptosis via downregulating Cyclin D1 and CDK6 in OC [36]. Overexpressed miR-211 apparently inhibits proliferation, xenograft growth, and induces apoptosis by suppressing PHD finger protein 19 (PHF19) in OC [37]. PHF19 is a component of the polycomb group of proteins and negatively regulated by miR-211. Knockdown of miR-211 promotes the viability and proliferation of OC cells [63].
MiR-211 regulates cell proliferation and apoptosis in multiple cancers
| Cancer type | Downstream target | Effect | Reference |
|---|---|---|---|
| Thyroid cancer | SPARC | Proliferation inhibition | [26] |
| SOX11 | Proliferation inhibition | [25] | |
| NSCLC | SRCIN1 | Proliferation inhibition | [54] |
| ZNF217 | Proliferation inhibition | [45] | |
| EPHB6 | Promotes proliferation | [44] | |
| SRCIN1 | Promotes proliferation | [43] | |
| Tongue cancer | Ezrin | Proliferation inhibition | [57] |
| Gastric cancer | SOX4 | Proliferation inhibition | [30] |
| HCC | SATB2 | Proliferation inhibition | [27] |
| ZEB2 | Proliferation inhibition and Apoptosis induction. | [28] | |
| ACSL4 | Proliferation inhibition | [15] | |
| FOXD1 | Proliferation inhibition | [29] | |
| CRC | CHD5 | Proliferation inhibition | [60] |
| CDK6 | Promotes proliferation | [65] | |
| Bladder cancer | HDAC9 | Proliferation inhibition and Apoptosis induction | [35] |
| Ovarian cancer | Cyclin D1/CDK6 | Proliferation inhibition and Apoptosis induction | [36] |
| PHF19 | Proliferation inhibition and Apoptosis induction | [37] | |
| Cervical Cancer | SPARC | Proliferation inhibition | [38] |
| SOX4 | Proliferation inhibition | [31] | |
| Melanoma | IL-10Rα | Proliferation inhibition | [51] |
| Chondrosarcoma | VHL | Promotes proliferation | [40] |
| Breast cancer | SIRT1 | Proliferation inhibition and Apoptosis induction | [16] |
NSCLC: non-small cell lung cancer; HCC: hepatocellular carcinoma; RCC: renal cell carcinoma.
Contrary to the anti-proliferative effect reported above, emerging researches indicate miR-211 may promote cell proliferation various types of cancer. In details, MiR-211 facilitates the proliferation of osteosarcoma cells and inhibits cellular apoptosis [39]. By the downregulation of Von Hippel-Lindau (VHL), miR-211 may function as an oncogenic in chondrosarcoma to modulate cell proliferation [40]. MiR-211 may function as an oncogenic in NSCLC to enhance cell proliferation and colony formation by regulating SRCIN1 [43]. Furthermore, miR-211 promotes the cell proliferation through targeting EPH receptor B6 (EPHB6) in NSCLC [44]. EPHB6, belongs to the Eph receptors family, that is found reduced in gastric carcinoma and correlated with malignancy indicators for cancer [64]. In CRC, miR-211-3p also serves as an oncogene. The overexpressed miR-211-3p may facilitate CRC cell proliferation by targeting CDK6 [65]. CDK6 is recognized not only as a typical cyclin-dependent kinase but as a transcriptional regulator which regulates the transcription of a number of genes [66].
In OSCC, the roles for miR-211 as a tumor suppressor and as an oncomiR have been reported. In details, upregulated miR-211 increases the proliferation anchorage-independent colony formation of OSCC cells [14]. In contrast to the above study, Guo et al. found that miR-211 suppresses cells proliferation in OSCC [67]. These researches may suggest that the role of miR211 in cancers is related to tumor heterogeneity.
Migration and invasion
Metastasis as main lethal feature of cancer is a multistep process, including tumor cell dissemination from the initial tumor and colonizing distant organs. The improved capacity of cancer cells to undergo migration and invasion contributes to the first step of this process. MiR-211 has been confirmed to regulate cells migration and invasion in various cancers.
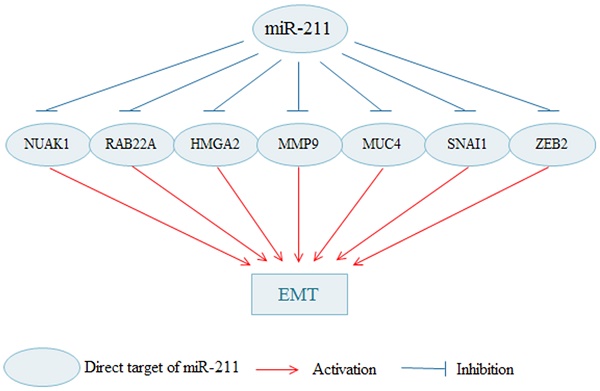
Cancer cells can acquire the migration and invasion capacity through the EMT progress. Various researches suggest that miR-211 is implicated to inhibit various genes that are involved in EMT (Fig. 1). MiR-211 contributes to cell adhesion and inhibits invasion by targeting NUAK Family SNF1-like Kinase 1 (NUAK1) in melanoma [68]. NUAK1 (also named ARK5) is a member of AMPK catalytic subunit family involved in invasion and metastasis of malignant tumor [69]. MiR-211 can inhibits EMT of melanoma cells via downregulating RAB22A, which is negatively regulated by MiR-211 [22]. HMGA2, a non-histone architectural transcription factor, influences a variety of biological processes, such as the cell cycle process, apoptosis, DNA damage repair process, senescence and EMT [70]. MiR-211 suppresses breast cancer cells migration, invasion and EMT phenotype and through the downregulation of HMGA2 [71]. Matrix metalloproteinase 9 (MMP9) has been widely found to link to the pathology of cancers, such as invasion, metastasis and angiogenesis [72]. MiR-211 suppresses cell invasion and cell EMT process by reducing MMP9 in gastric cancer [32]. MiR-211 suppresses invasion and EMT of cervical cancer cells by targeting MUCIN-4(MUC4) [73]. MUC4, a high molecular weight glycoprotein, is found to be associated with enhanced cancer cell invasion and EMT in several types of cancers [74]. In addition, MiR-211-5p attenuates cell migration in HCC through targeting ZEB2 [28]. Snai1 family transcriptional repressor 1 (SNAI1) is zinc-finger transcription factor and is proved to induce EMT in cancer [75]. Upregulated miR-211 inhibits cell migration and invasion by downregulating SNAI1 in RCC [33]. Knockdown of miR-211 promotes the migration, invasion and EMT of OC cells [63].
The miR-211 regulates EMT in cancer cells. By targeting various molecules, miR-211 inhabits the process of the EMT; thus, inhibiting the migration and invasion of cancer cells. EMT: epithelial-to-mesenchymal transition.
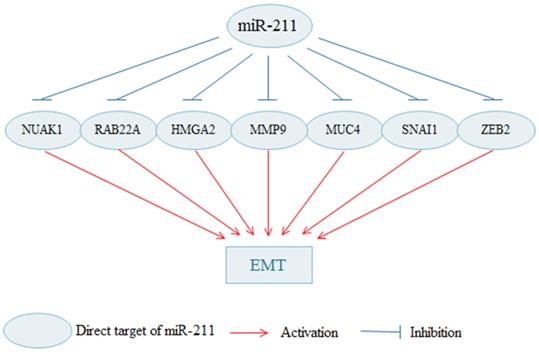
In addition to the EMT process, miR-211 can also regulate the metastasis of cancer cells by influencing other pathways (Fig. 2). MiR-211 is indicated to decreases cells migration and invasion in malignant melanomas [20]. Upregulated miR-211 decreases the invasive potential of melanoma cells through targeting BRN2, which is an important transcription factors [23, 76]. In melanoma, Bcl-2 overexpression increases cell migration, and overexpression of miR-211 in Bcl-2 overexpressing cells can rescue cell migration [19]. RAB22A is a member of the Rab family of small GTPases, and the oncogenic role of RAB22A is also observed in several types of cancer [77]. In a xenograft model, miR-211 exhibits a dual role in melanoma progression, facilitating cell proliferation while suppressing metastatic spread [78]. MiR-211 acts as a tumor suppressor in in thyroid cancer to modulate cells migration and invasion by decreasing SOX11 [25]. Besides, it is also reported that miR-211 can suppress cells invasion by regulating SPARC in thyroid cancer [26] and cervical cancer [38]. In gastric cancer [31] and cervical cancer [30], SOX4 is identified a target of miR-211, and promotes cells invasion. In HCC, miR-211 suppresses cells invasion by targeting SATB2, which is negatively regulated by miR-211 [27]. In addition, miR-211 can inhibit HCC cells migration and invasion via downregulating ACSL4 [15]. Upregulated miR-211 inhibits HCC cells migration and angiogenesis through regulating FOXD1 [29]. Enforced expression of miR-211 significantly decreases cell invasion and migration in pancreatic cancer [49]. MiR-211 may be a tumor suppressor in RCC to inhibit cells migration and invasion [34]. Through regulating HDAC9, MiR-211 markedly suppresses bladder cancer cells migration and invasion [35]. In OC, upregulated miR-211 apparently suppresses migration by inhibiting PHF19 [37].
Apart from the EMT, miR-211 regulates cell migration and invasion in cancers through other pathways. By targeting BRN2, SPARC, SOX11, among others, miR-211 suppresses cell migration and invasion in cancers. In contrary, miR-211 promotes cell migration and invasion in cancers by targeting SRCIN1, EPHB6 and VHL.
MiR-211 is confirmed as an oncogenic function in multiple cancers to regulate cells migration and invasion via targeting different genes. Upregulated miR-211 increases the migration of OSCC cells [14]. MiR-211 facilitates cell migration in osteosarcoma [39]. Furthermore, miR-211 may accelerate cell migration by the down-regulation of VHL in chondrosarcoma [40].
In some tumors, miR-211 can act as both a tumor suppressor and a tumor promoter. In detail, SNP of rs187960998 in miR-211 inhibits colon cancer cell invasion by upregulating CHD5 [60]. Another research shows that miR-211-3p acts as a tumor promoter to increase CRC cells invasion [65]. In NSCLC, overexpressed miR-211-3p inhibits cells migration by downregulating ZNF217 [45]. However, miR-211 may also function as an oncogenic to facilitate cell migration and invasion by regulating SRCIN1 [43] or EPHB6 [44] in NSCLC. These results suggest that the effects of miR-211 may be strongly related to tumor heterogeneity. Furthermore, miR-211 inhibits cells migration and invasion ability, and reverse the EMT phenotype by downregulating high mobility group A2 (HMGA2) in breast cancer [71]. MiR-211 suppresses cell invasion and migration in triple-negative breast cancer [47]. However, high miR-211 facilitates brain metastasis by increasing the cells trans-blood-brain barrier (BBB) migration ability, BBB adherence, and stem cell properties in TNBC. SOX11 is the downstream target of miR-211 and negatively regulated by miR-211 [46]. Interestingly, miR-211 studies on breast cancer shows that miR-211 targets different genes to inhibit breast cancer proliferation, invasion and migration, but promotes metastasis. These results may indicate that miR-211 may play a different role in different stages of breast cancer progression.
Chemoresistance
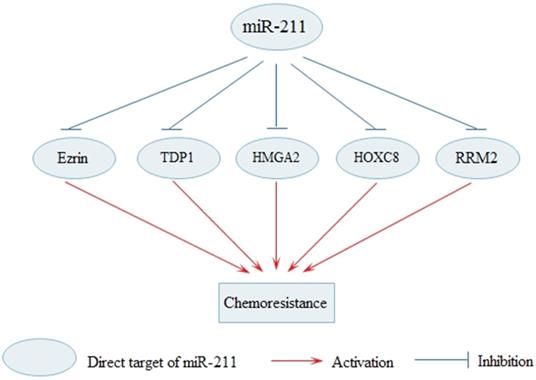
Systemic chemotherapy is important treatment for cancer but long-term administration of chemotherapy may lead to the development of chemoresistance. Effective therapies to reverse chemoresistance in cancer are still in exploration. At present, effective treatments to reverse cancer chemoresistance are still being explored. Various researches indicate that dysregulated miR-211 is involved in the molecular mechanisms of chemoresistance (Fig. 3). Overexpression miR-211 significantly decreases cell cisplatin resistance in tongue cancer via targeting Ezrin/Fak/Src signaling [57]. The upregulated miR-211 enhances chemosensitivity of breast cancer cells by downregulating HMGA2 [71]. In addition, upregulated miR-211 in PDAC increased the sensitivity to gemcitabine by reducing ribonucleotide reductase subunit 2(RRM2) [49]. Elevated ribonucleotide reductase (RR) activity and over expression of RRM2 pronouncedly increases the drug-resistant properties of human malignant cells [79]. MiR-211 enhances platinum chemosensitivity through inhibiting DNA damage response (DDR) in ovarian cancer, and TDP1, one of the DDR genes, is identified as a target of miR-211 [18]. Multiple studies have shown that the enforced DDR is one of the most pivotal mechanisms for resistance to platinum-based chemotherapy [80]. Moreover, miR-211 can promote cell sensitivity to paclitaxel (PTX) by targeting HOXC8 [81]. Besides, the finding also shows that miR-211 contributes to BRAF inhibitor resistance in melanoma [41]. The BRAF V600E mutation which is common in melanoma, is the focus of recently developed BRAF inhibitors (BRAFi), such as vemurafenib and dabrafenib [82].
MiR-211 regulates the sensitivity of cancer cells to the chemotherapy. MiR-211 enhances the sensitivity of cancer cells to the chemotherapy through the inhibition of Ezrin, TDP1, HMGA2, HOXC8 and RRM2.
Cell cycle
Numerous signaling pathways and checkpoints are involved in regulating the cell cycle. Studies on cell cycle regulation provide a new direction for cancer treatments. A large number of studies have shown that miR-211 regulates cell cycle by regulating multiple targets (Fig. 4). Various factors affect the G1-S phase transition, including cyclin D1, cyclin-dependent kinases (CDKs), and CDK inhibitors (CKIs). In OSCC, miR-211 can increase the number of cells in G1 phase, and p53, p21, c-PARP levels, and decrease cyclin D1 levels [67]. Overexpressed miR-211 induces cell cycle arrest and apoptosis by targeting SOX11 in thyroid tumor [25]. As a tumor suppressor, miR-211 inhibits cell proliferation and induces cell apoptosis by down-regulating cyclin D1 and CDK6 in OC to block cells in G0/G1 phase [36]. Apart from its inhibitory effect, miR-211 also acts as a facilitator in cell cycle process. MiR-211-3p promotes CRC cell proliferation and decreases cell cycle arrested at G1 phase by targeting CDK6 [65]. Therefore, research on miR-211 can better reveal the molecular mechanism of miR-211 in cell cycle regulation.
MiR-211 regulates cell cycle progression in cancers via various signaling pathways. MiR-211 suppresses cell cycle progression through regulating SOX11, CDK6, Cyclin D1, P53, P21 and c-PARP. In contrary, miR-211 promotes cell cycle progression by inhibiting CDK6.
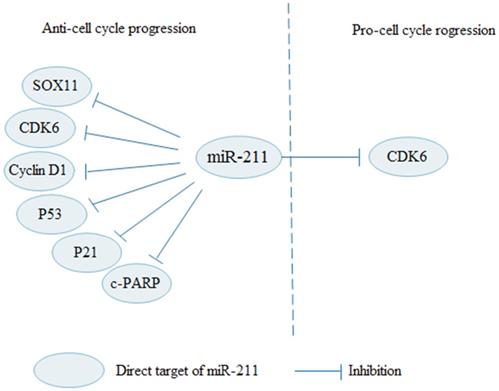
Cancer stem cell
Cancer stem cells (CSCs) are a small group of solid tumor cells with self-renewal, differentiation properties and tumorigenic potential [83]. CSCs are thought to be closely related to tumor formation, metastasis and recurrence [84]. Metastatic cancer stem cells (MCSCs) are a subpopulation of cancer cells with stem cell properties, invasion capabilities, and trafficking functions [85]. MiR-211 levels are found to be regulating the stemness properties of CSCs. High expression of miR-211 enhances stemness properties of cancer cells to promote brain metastasis in TNBC by regulating the SOX11/NGN2-dependent axis [46].
The targets of miR-211
SOX transcription factor family
SRY-related HMG box (SOX) transcription factors have been recognized to play a critical role in a variety of tumor biological processes, including tumorigenesis, proliferation and metastasis, and change in the tumor microenvironment [86]. Among the more than 20 members of the SOX family, SOX11 and SOX4 have been shown to play important roles as targets of mir211 in a variety of cancers.
SOX11 is a crucial member of the SOX transcription factors, which encodes a neural transcription factor, have functional role in multiple cancers [87]. SOX11 exerts tumor-stimulative effects in various tumors by promoting cell survival and proliferation, and initiating metastasis angiogenesis [88]. In a limited number of malignant tumors, SOX11 acts as a tumor suppressor by inhibiting cell proliferation and metastasis, as well as suppressing the maintenance of cancer-initiating cells [89]. MiR-211 acts as a tumor suppressor or tumor promoter in malignancies by targeting SOX11. In detail, miR-211 modulates cells proliferation, migration and invasion by decreasing SOX11 in thyroid cancer [25]. High miR-211 increases tumor cell adhesion, trans-blood-brain barrier (BBB) ability and cancer stem cell capability to promote brain metastasis in TNBC. SOX11 is the downstream targeting molecules of miR-211 and negatively regulated by miR-211 [46]. These results suggest that the role of miR-211 in tumors is closely related to the function of its targets.
SOX4 is thought to be involved in the development of many diseases, including malignancies [90]. Numerous findings highlight the explicit role of SOX4 in regulating cancer progression by affecting the ability of cancer cells to survive, migrate and invade [91]. Emerging researches show that miR-211 targets SOX4 to regulate cancer progression. MiR-211 serves as a tumor suppressor to inhibit cells proliferation and invasion by reducing SOX4 in gastric cancer and cervical cancer [30, 31].
SPARC
SPARC is an extracellular matrix glycoprotein secreted by kinds of cells and has been shown to be associated with the cancer progression [92]. Numerous studies have confirmed that SPARC affects cancer progression by modulating cell adhesion, invasion, metastasis, and angiogenesis [93]. However, there are still emerging studies highlighting the role of SPARC in cancer cells proliferation regulation, such as oral squamous cell carcinoma [94], liver cancer [95], and cervical cancer [96]. SPARC is involved in regulating multiple cancers progression as a target gene of miR-211. In thyroid cancer, SPARC acts as an oncogene to promote cells proliferation and invasion, and is regulated by miR-211 [26]. Besides, miR-211 inhibits cervical cancer cells proliferation, invasion and migration by inhibiting SPARC [38].
SNAI1
SNAI1 is a zinc finger transcription factor and functions as a driver of cancer progression [97]. Numerous studies have shown that Snail1 is a crucial inducer of EMT and plays a vital role in tumor metastasis [98]. Recently, the relationship between SNAI1 and miRNAs is reported in various cancers [99]. Moreover, Wang K et al. [33] suggests that miR-211 suppresses the cells migration and invasion by the downregulation of SNAI1 in RCC, emphasizing the significance of the miR211/SNAI1 axis in cancer.
ZEB2
ZEB2, also known as smad-interacting protein 1 (SIP1), belongs to the zinc-finger E-box binding protein (ZEB) family and has been shown to induce cancer progression [100]. Existing studies indicate that ZEB2 promotes cancer metastasis by inducing EMT [101]. ZEB2 is modulated by multiple miRNAs in cancers, such as miR-139-5P [102] and miR-338-3p [103]. Besides, miR-211 suppresses the cells survival and attenuates the cells metastasis through downregulating ZEB2 in HCC [28].
The other targets of miR-211
In addition to the above targets, miR-211 also acts on a range of other targets. The targets and the effects of miR-211 are explicitly shown in Table 3. Overall, miR-211 plays an important role in cancer progression in diverse types of cancer, through regulating different targets.
Targets of miR-211 known to be involved in cancers
| Target | Cancer type | Effect | Reference |
|---|---|---|---|
| SOX11 | Thyroid cancer | Proliferation, migration, and invasion | [25] |
| SOX11 | Breast cancer | Metastasis | [46] |
| HMGA2 | Breast cancer | Migration and invasion and EMT | [71] |
| SIRT1 | Breast cancer | Proliferation and apoptosis | [16] |
| SPARC | Thyroid cancer | Proliferation and invasion | [26] |
| SPARC | Cervical cancer | Proliferation, invasion and migration | [38] |
| MMP9 | Gastric cancer | Invasion and EMT | [32] |
| SOX4 | Gastric cancer | Proliferation and invasion | [30] |
| SOX4 | Cervical Cancer | Proliferation and invasion | [31] |
| MUC4 | cervical cancer | Invasion and EMT | [72] |
| SRCIN1 | NSCLC | Proliferation, migration and invasion | [43] |
| SRCIN1 | NSCLC | Proliferation | [54] |
| ZNF217 | NSCLC | Proliferation and migration | [45] |
| EPHB6 | NSCLC | Proliferation, migration and invasion | [44] |
| Ezrin | Tongue cancer | Proliferation | [57] |
| SATB2 | HCC | Proliferation and invasion | [27] |
| ZEB2 | HCC | Proliferation, apoptosis and metastasis | [28] |
| ACSL4 | HCC | Proliferation, migration and invasion | [15] |
| FOXD1 | HCC | Proliferation and migration | [29] |
| SNAI1 | RCC | Migration and invasion | [33] |
| CHD5 | CRC | Proliferation inhibition invasion | [60] |
| CDK6 | CRC | Promotes proliferation | [65] |
| HDAC9 | Bladder cancer | Proliferation, apoptosis, migration and invasion | [35] |
| Cyclin D1/CDK6 | Ovarian cancer | Proliferation and apoptosis | [36] |
| PHF19 | Ovarian cancer | Proliferation, apoptosis and migration | [37] |
| RAB22A | Melanoma | EMT | [22] |
| IL-10Rα | Melanoma | Proliferation | [51] |
| NUAK1 | Melanoma | Adhesion and invasion | [68] |
| BRN2 | Melanoma | Migration and invasion | [23] |
| Bcl-2 | Melanoma | Migration and invasion | [19] |
| VHL | Chondrosarcoma | Proliferation and migration | [40] |
NSCLC: non-small cell lung cancer; CRC: colorectal cancer; HCC: hepatocellular carcinoma.
Factors regulating the expression of miR-211 in cancer
LncRNA and CircRNA
Emerging researches indicate that miRNAs, long non-coding RNAs (lncRNAs), and circular RNAs (circRNAs), which are major members of non-coding RNAs (ncRNAs), are involved in cancer development. Competing endogenous RNAs (ceRNAs) networks describe competitive binding between sponge RNAs and miRNA targets as well as the regulation of miRNA targets activity [104]. Various lncRNAs and circRNAs are indicated to participate in the regulation of miR-211 in different cancers (Table 4).
Regulatory factors regulate the expression of miR-211 in various cancers
| Type of Factors | Factor | Cancer type | Reference |
|---|---|---|---|
| LncRNA | lncRNA KCNQ1OT1 | Tongue cancer | [57] |
| LncRNA MCM3AP-AS1 | Thyroid cancer | [26] | |
| lncRNA NEAT1 | Breast cancer | [71] | |
| LncRNA SNHG15 | Breast cancer | [17] | |
| LncRNA TTN-AS1 | Breast cancer | [47] | |
| LncSNHG15 | NSCLC | [45] | |
| LncRNA DGCR5 | NSCLC | [44] | |
| LncRNA-uc002kmd.1 | CRC | [107] | |
| LncRNA TUSC7 | CRC | [75] | |
| LncRNA TUSC7 | Osteosarcoma | [39] | |
| LncRNA NORAD | HCC | [29] | |
| LncRNA MALAT1 | Ovarian cancer | [37] | |
| CircRNA | CircCCDC66 | NSCLC | [54] |
| Circ_0008285 | Cervical Cancer | [30] | |
| CircNRIP1 | Ovarian cancer | [81] | |
| Others | Ligustrazine | Ovarian cancer | [63] |
| Alpinetin | OSCC | [67] | |
| Adipocytes | Melanoma | [78] | |
| PERK | Burkitt's lymphoma | [110] |
NSCLC: non-small cell lung cancer; OSCC: oral squamous cell carcinoma; CRC: colorectal cancer; HCC: hepatocellular carcinoma.
LncRNA KCNQ1OT1 facilitates proliferation and chemo-resistance in tongue cancer by acting as a sponge of miR-211 through Ezrin/Fak/Src signaling [57]. LncRNA MCM3AP-AS1 promotes the papillary thyroid cancer cell proliferation and invasion by regulating miR-211/SPARC axis [26]. In breast cancer, lncRNA NEAT1 is upregulated. NEAT1 negatively regulates miR-211 expression via acting as an endogenous sponge [71]. In breast cancer, LncRNA SNHG15 facilitates cells proliferation and migration through sponging miR-211-3p [17]. In addition, LncRNA TTN-AS1 is significantly upregulated and regulates the proliferation and invasive and migratory abilities of triple-negative breast cancer cells by inhibiting miR-211 [47]. Upregulated LncSNHG15 accelerates the NSCLC cells proliferation and migration by sponging miR-211-3p [45]. Moreover, LncRNA DGCR5 suppresses cell proliferation, migration, and invasion through modulating miR-211/EPHB6 axis in NSCLC [44]. Studies show that decreased microphthalmia-associated transcription factor (MITF) are associated with increased proliferation. MITF/miR-211 axis inhibits the invasive program in melanoma by blocking adhesion [105]. CD44, a non-kinase transmembrane glycoprotein, is frequently shows alternative spliced variants that are considered to play a significant role in cancer development and progression [106]. LncRNA-uc002kmd.1 expression is highly expressed in CRC. The results indicate that lncRNA-uc002kmd.1 may upregulate CD44 by competing for miR-211-3p, subsequently regulating cell migration and proliferation in CRC [107]. LncRNA TUSC7 inhibits proliferation and invasion of CRC cells and induces cell cycle arrest through completely sponging miR-211-3p. TUSC7 is negatively correlated with miR-211-3p and CDK6 is a downstream target of miR-211-3p [75]. Likewise, another research also indicates that LncRNA TUSC7 suppresses the proliferation, migration of osteosarcoma cells and induces cellular apoptosis, which is mediated by miR-211. miR-211 is inhibited remarkably by TUSC7 and the reciprocal negative regulation exists between TUSC7 and miR-211[39]. In HCC, LncRNA NORAD facilitates cells proliferation, migration and vessel formation abilities by sponging miR-211 [29]. LncRNA MALAT1 can sponge miR-211 as a competing endogenous RNA thus facilitating the OC progression [37].
CircCCDC66 acts as a sponge of miR-211 and promotes NSCLC cell proliferation by regulating miR-211/SRCIN1 axis [54]. Besides, irc_0008285 is remarkably elevated in cervical cancer and facilitates cervical cancer cells proliferation and invasion through regulating miR-211-5p/SOX4 Axis [30]. Homeobox C8 (HOXC8) is remarkably overexpressed in cancer and identified as a regulator in resistance of hepatocellular carcinoma to oxaliplatin [108]. Furthermore, circNRIP1 promotes the resistance of OC cells to paclitaxel through affecting the miR-211-5p/HOXC8 axis [81].
Other factors
Ligustrazine (LSZ) can significantly increase the expression of miR-211 in OC. Ligustrazine inhibits the OC cells proliferation and migration by regulating miR-211 [63]. Alpinetin suppresses OSCC cells proliferation by upregulating miR-211 and Notch Pathway deactivation [67]. Adipocytes secretes the cytokines IL-6 and TNF-α, which induces a proliferative-to-invasive phenotypic switch in melanoma by decreasing the expression of miR-211 [78]. Protein kinase RNA (PKR)-like ER kinase (PERK), which is an ER transmembrane protein kinase, catalyzes serine 51 phosphorylation on eIF2α, thus, resulting in decreased protein synthesis [109]. A PERK-miR-211 axis inhibits circadian regulators and protein synthesis to facilitate cancer cell survival in Burkitt's lymphoma, and miR-211 diametrically modulates Bmal1through distinct mechanisms [110].
Conclusion
Emerging experimental evidence reveal that miR-211 play a crucial role in cancer, because it is involved in various biological processes, including cell proliferation, apoptosis, invasion, and metastasis. The complicated regulatory network targeting miR-211 and its target genes remain incompletely understood. Nevertheless, miR-211 is a potential prognostic biomarker, even if its extensive functions and dysregulation are associated with multiple types of cancer.
More attractive is miR-211-based therapy that replaces miR-211 in cancer cells and regains control of numerous signaling pathways regulated by endogenous miR-211. Also, overexpressed miR-211 can enhance the sensitivity of cancer cells to chemotherapy, indicating a new approach for overwhelming drug resistance. The implementation of specific miRNA mimics has been proved to have strong tumor inhibitory ability in experimental tumor models, such as miR-34 [111].
However, miRNA-based therapy still has some limitations, for instance, the lack of dosage requirements and its administration. The premature termination of the two registered clinical trials using the miR-Rx34 were due to serious immune-related adverse events that may be caused by off-target effects due to high doses of miR-34 mimic [112]. For all that, work is under way to address these limitations, and recent advances in drug-delivered systems provide a technical solution for the administration and delivery of miRNA mimics. These discoveries will usher in a new era of RNA-based therapy, especially in the field of cancer treatment. Thence, there are two main goals that must be realized in order to enter the RNA therapeutic era: further improve the drug delivery strategy and determine the possible adverse reactions of these molecules.
In summary, further researches should be implemented before miR-211 can be widely used as a prognostic biomarker and therapeutic target, but its dysregulation in numerous cancers, and participation in a variety of biological functions, makes it a promising molecule target for cancer therapy.
Acknowledgements
The authors would like to thank colleagues from the department of oncology, the Affiliated Jiangning Hospital with Nanjing Medical University and colleagues from Department of Gynecology, Zhangjiagang First People's Hospital.
Funding
This research was funded by the National Nature Science Foundation of China, grant number 82103032, Medical Research Grant of Jiangsu Commission of Health, grant number M2020010, Health Science and Technology Development Fund of Nanjing, grant number YKK21224, the Science Foundation of Jiangsu Health vocational college, grant number JKC201948, the Science and Technology Development Fund of Nanjing Medical University, grant number NMUB2019235 and the Research and development fund of Kangda College of Nanjing Medical University, grant number KD2020KYJJZD006, KD2021KYJJZD026.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Bartel DP. MicroRNAs: target recognition and regulatory functions. Cell. 2009;136:215-33
2. Chen X, Mangala LS, Rodriguez-Aguayo C, Kong X, Lopez-Berestein G, Sood AK. RNA interference-based therapy and its delivery systems. Cancer metastasis reviews. 2018;37:107-24
3. Broughton JP, Lovci MT, Huang JL, Yeo GW, Pasquinelli AE. Pairing beyond the Seed Supports MicroRNA Targeting Specificity. Molecular cell. 2016;64:320-33
4. Peng B, Theng PY, Le MTN. Essential functions of miR-125b in cancer. Cell proliferation. 2021;54:1-17
5. Montoya V, Fan H, Bryar PJ, Weinstein JL, Mets MB, Feng G. et al. Novel miRNA-31 and miRNA-200a-Mediated Regulation of Retinoblastoma Proliferation. PloS one. 2015;10:1-21
6. Ong SG, Lee WH, Kodo K, Wu JC. MicroRNA-mediated regulation of differentiation and trans-differentiation in stem cells. Advanced drug delivery reviews. 2015;88:3-15
7. Pinti MV, Hathaway QA, Hollander JM. Role of microRNA in metabolic shift during heart failure. American journal of physiology Heart and circulatory physiology. 2017;312:H33-h45
8. Si W, Shen J, Zheng H, Fan W. The role and mechanisms of action of microRNAs in cancer drug resistance. Clinical epigenetics. 2019;11:25-49
9. Paladini L, Fabris L, Bottai G, Raschioni C, Calin GA, Santarpia L. Targeting microRNAs as key modulators of tumor immune response. Journal of experimental & clinical cancer research: CR. 2016;35:103-122
10. Labbaye C, Testa U. The emerging role of MIR-146A in the control of hematopoiesis, immune function and cancer. Journal of hematology & oncology. 2012;5:13-23
11. Esquela-Kerscher A, Slack FJ. Oncomirs - microRNAs with a role in cancer. Nature reviews Cancer. 2006;6:259-69
12. Shen K, Cao Z, Zhu R, You L, Zhang T. The dual functional role of MicroRNA-18a (miR-18a) in cancer development. Clinical and translational medicine. 2019;8:32-45
13. Hansen TB, Kjems J, Damgaard CK. Circular RNA and miR-7 in cancer. Cancer research. 2013;73:5609-12
14. Chang KW, Liu CJ, Chu TH, Cheng HW, Hung PS, Hu WY. et al. Association between high miR-211 microRNA expression and the poor prognosis of oral carcinoma. Journal of dental research. 2008;87:1063-8
15. Qin X, Zhang J, Lin Y, Sun XM, Zhang JN, Cheng ZQ. Identification of MiR-211-5p as a tumor suppressor by targeting ACSL4 in Hepatocellular Carcinoma. Journal of translational medicine. 2020;18:326-339
16. Yarahmadi S, Abdolvahabi Z, Hesari Z, Tavakoli-Yaraki M, Yousefi Z, Seiri P. et al. Inhibition of sirtuin 1 deacetylase by miR-211-5p provides a mechanism for the induction of cell death in breast cancer cells. Gene. 2019;711:1-40
17. Kong Q, Qiu M. Long noncoding RNA SNHG15 promotes human breast cancer proliferation, migration and invasion by sponging miR-211-3p. Biochemical and biophysical research communications. 2018;495:1594-600
18. Wang T, Hao D, Yang S, Ma J, Yang W, Zhu Y. et al. miR-211 facilitates platinum chemosensitivity by blocking the DNA damage response (DDR) in ovarian cancer. Cell Death Dis. 2019;10:495-509
19. De Luca T, Pelosi A, Trisciuoglio D, D'Aguanno S, Desideri M, Farini V. et al. miR-211 and MITF modulation by Bcl-2 protein in melanoma cells. Molecular carcinogenesis. 2016;55:2304-12
20. Levy C, Khaled M, Iliopoulos D, Janas MM, Schubert S, Pinner S. et al. Intronic miR-211 assumes the tumor suppressive function of its host gene in melanoma. Molecular cell. 2010;40:841-9
21. Babapoor S, Horwich M, Wu R, Levinson S, Gandhi M, Makkar H. et al. microRNA in situ hybridization for miR-211 detection as an ancillary test in melanoma diagnosis. Modern pathology: an official journal of the United States and Canadian Academy of Pathology, Inc. 2016;29:461-75
22. Yu H, Yang W. MiR-211 is epigenetically regulated by DNMT1 mediated methylation and inhibits EMT of melanoma cells by targeting RAB22A. Biochemical and biophysical research communications. 2016;476:400-5
23. Boyle GM, Woods SL, Bonazzi VF, Stark MS, Hacker E, Aoude LG. et al. Melanoma cell invasiveness is regulated by miR-211 suppression of the BRN2 transcription factor. Pigment cell & melanoma research. 2011;24:525-37
24. Xu Y, Brenn T, Brown ER, Doherty V, Melton DW. Differential expression of microRNAs during melanoma progression: miR-200c, miR-205 and miR-211 are downregulated in melanoma and act as tumour suppressors. British journal of cancer. 2012;106:553-61
25. Wang L, Shen YF, Shi ZM, Shang XJ, Jin DL, Xi F. Overexpression miR-211-5p hinders the proliferation, migration, and invasion of thyroid tumor cells by downregulating SOX11. Journal of clinical laboratory analysis. 2018;3:1-13
26. Liang M, Jia J, Chen L, Wei B, Guan Q, Ding Z. et al. LncRNA MCM3AP-AS1 promotes proliferation and invasion through regulating miR-211-5p/SPARC axis in papillary thyroid cancer. Endocrine. 2019;65:318-26
27. Jiang G, Cui Y, Yu X, Wu Z, Ding G, Cao L. miR-211 suppresses hepatocellular carcinoma by downregulating SATB2. Oncotarget. 2015;6:9457-66
28. Jiang G, Wen L, Deng W, Jian Z, Zheng H. Regulatory role of miR-211-5p in hepatocellular carcinoma metastasis by targeting ZEB2. Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie. 2017;90:806-12
29. Sun DS, Guan CH, Wang WN, Hu ZT, Zhao YQ, Jiang XM. LncRNA NORAD promotes proliferation, migration and angiogenesis of hepatocellular carcinoma cells through targeting miR-211-5p/FOXD1/VEGF-A axis. Microvascular research. 2020;134:1-16
30. Bai Y, Li X. hsa_circ_0008285 Facilitates the Progression of Cervical Cancer by Targeting miR-211-5p/SOX4 Axis. Cancer management and research. 2020;12:3927-36
31. Wang CY, Hua L, Sun J, Yao KH, Chen JT, Zhang JJ. et al. MiR-211 inhibits cell proliferation and invasion of gastric cancer by down-regulating SOX4. International journal of clinical and experimental pathology. 2015;8:14013-20
32. Wang XD, Wen FX, Liu BC, Song Y. MiR-211 inhibits cell epithelial-mesenchymal transition by targeting MMP9 in gastric cancer. International journal of clinical and experimental pathology. 2017;10:7551-8
33. Wang K, Jin W, Jin P, Fei X, Wang X, Chen X. miR-211-5p Suppresses Metastatic Behavior by Targeting SNAI1 in Renal Cancer. Molecular cancer research: MCR. 2017;15:448-56
34. Quan J, Pan X, He T, Lin C, Lai Y, Chen P. et al. Tumor suppressor miR-211-5p is associated with cellular migration, proliferation and apoptosis in renal cell carcinoma. Experimental and therapeutic medicine. 2018;15:4019-28
35. Wang W, Liu Z, Zhang X, Liu J, Gui J, Cui M. et al. miR-211-5p is down-regulated and a prognostic marker in bladder cancer. The journal of gene medicine. 2020;22:1-10
36. Xia B, Yang S, Liu T, Lou G. miR-211 suppresses epithelial ovarian cancer proliferation and cell-cycle progression by targeting Cyclin D1 and CDK6. Molecular cancer. 2015;14:57-69
37. Tao F, Tian X, Ruan S, Shen M, Zhang Z. miR-211 sponges lncRNA MALAT1 to suppress tumor growth and progression through inhibiting PHF19 in ovarian carcinoma. FASEB journal: official publication of the Federation of American Societies for Experimental Biology. 2018;32:1-14
38. Qu X, Gao D, Ren Q, Jiang X, Bai J, Sheng L. miR-211 inhibits proliferation, invasion and migration of cervical cancer via targeting SPARC. Oncology letters. 2018;16:853-60
39. Cong M, Jing R. Long non-coding RNA TUSC7 suppresses osteosarcoma by targeting miR-211. Bioscience reports. 2019;39:1-18
40. Chen C, Tian A, Zhou H, Zhang X, Liu Z, Ma X. Upregulation of miR-211 Promotes Chondrosarcoma Development via Targeting Tumor Suppressor VHL. OncoTargets and therapy. 2020;13:2935-43
41. Díaz-Martínez M, Benito-Jardón L, Alonso L, Koetz-Ploch L, Hernando E, Teixidó J. miR-204-5p and miR-211-5p Contribute to BRAF Inhibitor Resistance in Melanoma. Cancer research. 2018;78:1017-30
42. Chang CC, Lin CC, Wang CH, Huang CC, Ke TW, Wei PL. et al. miR-211 regulates the expression of RRM2 in tumoral metastasis and recurrence in colorectal cancer patients with a k-ras gene mutation. Oncology letters. 2018;15:8107-17
43. Ye L, Wang H, Liu B. miR-211 promotes non-small cell lung cancer proliferation by targeting SRCIN1. Tumour biology: the journal of the International Society for Oncodevelopmental Biology and Medicine. 2016;37:1151-7
44. Kang M, Shi J, Li B, Luo M, Xu S, Liu X. LncRNA DGCR5 regulates the non-small cell lung cancer cell growth, migration, and invasion through regulating miR-211-5p/EPHB6 axis. BioFactors (Oxford, England). 2019;45:788-94
45. Ma XR, Xu YL, Qian J, Wang Y. Long non-coding RNA SNHG15 accelerates the progression of non-small cell lung cancer by absorbing miR-211-3p. European review for medical and pharmacological sciences. 2019;23:1536-44
46. Pan JK, Lin CH, Kuo YL, Ger LP, Cheng HC, Yao YC. et al. MiR-211 determines brain metastasis specificity through SOX11/NGN2 axis in triple-negative breast cancer. Oncogene. 2021;40:1737-51
47. Sun E, Liu X, Lu C, Liu K. Long noncoding RNA TTNAS1 regulates the proliferation, invasion and migration of triplenegative breast cancer by targeting miR2115p. Molecular medicine reports. 2021;23:35-49
48. Giovannetti E, van der Velde A, Funel N, Vasile E, Perrone V, Leon LG. et al. High-throughput microRNA (miRNAs) arrays unravel the prognostic role of MiR-211 in pancreatic cancer. PloS one. 2012;7:1-19
49. Maftouh M, Avan A, Funel N, Frampton AE, Fiuji H, Pelliccioni S. et al. miR-211 modulates gemcitabine activity through downregulation of ribonucleotide reductase and inhibits the invasive behavior of pancreatic cancer cells. Nucleosides, nucleotides & nucleic acids. 2014;33:384-93
50. Itakura E, Huang RR, Wen DR, Paul E, Wünsch PH, Cochran AJ. IL-10 expression by primary tumor cells correlates with melanoma progression from radial to vertical growth phase and development of metastatic competence. Modern pathology: an official journal of the United States and Canadian Academy of Pathology, Inc. 2011;24:801-9
51. Venza I, Visalli M, Beninati C, Benfatto S, Teti D, Venza M. IL-10Rα expression is post-transcriptionally regulated by miR-15a, miR-185, and miR-211 in melanoma. BMC medical genomics. 2015;8:81-97
52. Yeung F, Hoberg JE, Ramsey CS, Keller MD, Jones DR, Frye RA. et al. Modulation of NF-kappaB-dependent transcription and cell survival by the SIRT1 deacetylase. The EMBO journal. 2004;23:2369-80
53. Dong J, Li B, Lin D, Lu D, Liu C, Lu X. et al. LincRNA00494 Suppresses Non-small Cell Lung Cancer Cell Proliferation by Regulating SRCIN1 Expression as a ceRNA. Frontiers in oncology. 2020;10:79-96
54. Hong W, Yu S, Zhuang Y, Zhang Q, Wang J, Gao X. SRCIN1 Regulated by circCCDC66/miR-211 Is Upregulated and Promotes Cell Proliferation in Non-Small-Cell Lung Cancer. BioMed research international. 2020;2020:530-41
55. Si W, Zhao Y, Zhou J, Zhang Q, Zhang Y. The coordination between ZNF217 and LSD1 contributes to hepatocellular carcinoma progress and is negatively regulated by miR-101. Experimental cell research. 2019;379:1-10
56. Zhang X, Flores LR, Keeling MC, Sliogeryte K, Gavara N. Ezrin Phosphorylation at T567 Modulates Cell Migration, Mechanical Properties, and Cytoskeletal Organization. International journal of molecular sciences. 2020;21:23-41
57. Zhang S, Ma H, Zhang D, Xie S, Wang W, Li Q. et al. LncRNA KCNQ1OT1 regulates proliferation and cisplatin resistance in tongue cancer via miR-211-5p mediated Ezrin/Fak/Src signaling. Cell Death Dis. 2018;9:742-56
58. Chen C, Xu ZQ, Zong YP, Ou BC, Shen XH, Feng H. et al. CXCL5 induces tumor angiogenesis via enhancing the expression of FOXD1 mediated by the AKT/NF-κB pathway in colorectal cancer. Cell Death Dis. 2019;10:178-95
59. Koelzer VH, Zlobec I, Lugli A. Tumor budding in colorectal cancer-ready for diagnostic practice? Human pathology. 2016;47:4-19
60. Zhu L, Wang R, Zhang L, Zuo C, Zhang R, Zhao S. rs187960998 polymorphism in miR-211 prevents development of human colon cancer by deregulation of 3'UTR in CHD5. OncoTargets and therapy. 2019;12:405-12
61. Bilir B, Osunkoya AO, Wiles WGt, Sannigrahi S, Lefebvre V, Metzger D. et al. SOX4 Is Essential for Prostate Tumorigenesis Initiated by PTEN Ablation. Cancer research. 2016;76:1112-21
62. Salgado E, Bian X, Feng A, Shim H, Liang Z. HDAC9 overexpression confers invasive and angiogenic potential to triple negative breast cancer cells via modulating microRNA-206. Biochemical and biophysical research communications. 2018;503:1087-91
63. Zhang H, Ding S, Xia L. Ligustrazine inhibits the proliferation and migration of ovarian cancer cells via regulating miR-211. Bioscience reports. 2020;11:1-18
64. Liu J, Xu B, Xu G, Zhang X, Yang X, Wang J. Reduced EphB6 protein in gastric carcinoma and associated lymph nodes suggests EphB6 as a gastric tumor and metastasis inhibitor. Cancer biomarkers: section A of Disease markers. 2017;19:241-8
65. Xu J, Zhang R, Zhao J. The Novel Long Noncoding RNA TUSC7 Inhibits Proliferation by Sponging MiR-211 in Colorectal Cancer. Cellular physiology and biochemistry: international journal of experimental cellular physiology, biochemistry, and pharmacology. 2017;41:635-44
66. Handschick K, Beuerlein K, Jurida L, Bartkuhn M, Müller H, Soelch J. et al. Cyclin-dependent kinase 6 is a chromatin-bound cofactor for NF-κB-dependent gene expression. Molecular cell. 2014;53:193-208
67. Guo Y, Chen Y, Liu H, Yan W. Alpinetin Inhibits Oral Squamous Cell Carcinoma Proliferation via miR-211-5p Upregulation and Notch Pathway Deactivation. Nutrition and cancer. 2020;72:757-67
68. Bell RE, Khaled M, Netanely D, Schubert S, Golan T, Buxbaum A. et al. Transcription factor/microRNA axis blocks melanoma invasion program by miR-211 targeting NUAK1. The Journal of investigative dermatology. 2014;134:441-51
69. Suzuki A, Lu J, Kusakai G, Kishimoto A, Ogura T, Esumi H. ARK5 is a tumor invasion-associated factor downstream of Akt signaling. Molecular and cellular biology. 2004;24:3526-35
70. Zhang S, Mo Q, Wang X. Oncological role of HMGA2 (Review). International journal of oncology. 2019;55:775-88
71. Li X, Wang S, Li Z, Long X, Guo Z, Zhang G. et al. The lncRNA NEAT1 facilitates cell growth and invasion via the miR-211/HMGA2 axis in breast cancer. International journal of biological macromolecules. 2017;105:346-53
72. Huang H. Matrix Metalloproteinase-9 (MMP-9) as a Cancer Biomarker and MMP-9 Biosensors: Recent Advances. Sensors (Basel, Switzerland). 2018 18
73. Xu D, Liu S, Zhang L, Song L. MiR-211 inhibits invasion and epithelial-to-mesenchymal transition (EMT) of cervical cancer cells via targeting MUC4. Biochemical and biophysical research communications. 2017;485:556-62
74. Ponnusamy MP, Lakshmanan I, Jain M, Das S, Chakraborty S, Dey P. et al. MUC4 mucin-induced epithelial to mesenchymal transition: a novel mechanism for metastasis of human ovarian cancer cells. Oncogene. 2010;29:5741-54
75. Zhao F, Wang M, Li S, Bai X, Bi H, Liu Y. et al. DACH1 inhibits SNAI1-mediated epithelial-mesenchymal transition and represses breast carcinoma metastasis. Oncogenesis. 2015;4:143-57
76. Pierce CJ, Simmons JL, Broit N, Karunarathne D, Ng MF, Boyle GM. BRN2 expression increases anoikis resistance in melanoma. Oncogenesis. 2020;9:64-79
77. Zhang Y, Zhao FJ, Chen LL, Wang LQ, Nephew KP, Wu YL. et al. MiR-373 targeting of the Rab22a oncogene suppresses tumor invasion and metastasis in ovarian cancer. Oncotarget. 2014;5:12291-303
78. Golan T, Parikh R, Jacob E, Vaknine H, Zemser-Werner V, Hershkovitz D. et al. Adipocytes sensitize melanoma cells to environmental TGF-β cues by repressing the expression of miR-211. Science signaling. 2019;12:1-17
79. Chen P, Wu JN, Shu Y, Jiang HG, Zhao XH, Qian H. et al. Gemcitabine resistance mediated by ribonucleotide reductase M2 in lung squamous cell carcinoma is reversed by GW8510 through autophagy induction. Clinical science (London, England: 1979). 2018;132:1417-33
80. Stefanou DT, Bamias A, Episkopou H, Kyrtopoulos SA, Likka M, Kalampokas T. et al. Aberrant DNA damage response pathways may predict the outcome of platinum chemotherapy in ovarian cancer. PloS one. 2015;10:1-19
81. Li M, Cai J, Han X, Ren Y. Downregulation of circNRIP1 Suppresses the Paclitaxel Resistance of Ovarian Cancer via Regulating the miR-211-5p/HOXC8 Axis. Cancer management and research. 2020;12:9159-71
82. Chapman PB, Hauschild A, Robert C, Haanen JB, Ascierto P, Larkin J. et al. Improved survival with vemurafenib in melanoma with BRAF V600E mutation. The New England journal of medicine. 2011;364:2507-16
83. Babaei G, Aziz SG, Jaghi NZZ. EMT, cancer stem cells and autophagy; The three main axes of metastasis. Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie. 2021;133:1109-19
84. Clarke MF. Clinical and Therapeutic Implications of Cancer Stem Cells. The New England journal of medicine. 2019;380:2237-45
85. Oskarsson T, Batlle E, Massagué J. Metastatic stem cells: sources, niches, and vital pathways. Cell stem cell. 2014;14:306-21
86. Grimm D, Bauer J, Wise P, Krüger M, Simonsen U, Wehland M. et al. The role of SOX family members in solid tumours and metastasis. Seminars in cancer biology. 2020;67:122-53
87. Tsang SM, Oliemuller E, Howard BA. Regulatory roles for SOX11 in development, stem cells and cancer. Seminars in cancer biology. 2020;67:3-11
88. Dey A, Kundu M, Das S, Jena BC, Mandal M. Understanding the function and regulation of Sox2 for its therapeutic potential in breast cancer. Biochimica et biophysica acta Reviews on cancer. 2022;1877:1886-92
89. Yang Z, Jiang S, Lu C, Ji T, Yang W, Li T. et al. SOX11: friend or foe in tumor prevention and carcinogenesis? Therapeutic advances in medical oncology. 2019;11:175-88
90. Hanieh H, Ahmed EA, Vishnubalaji R, Alajez NM. SOX4: Epigenetic regulation and role in tumorigenesis. Seminars in cancer biology. 2020;67:91-104
91. Vervoort SJ, van Boxtel R, Coffer PJ. The role of SRY-related HMG box transcription factor 4 (SOX4) in tumorigenesis and metastasis: friend or foe? Oncogene. 2013;32:3397-409
92. Camacho D, Jesus JP, Palma AM, Martins SA, Afonso A, Peixoto ML. et al. SPARC-p53: The double agents of cancer. Advances in cancer research. 2020;148:171-99
93. Tai IT, Tang MJ. SPARC in cancer biology: its role in cancer progression and potential for therapy. Drug resistance updates: reviews and commentaries in antimicrobial and anticancer chemotherapy. 2008;11:231-46
94. Jing Y, Jin Y, Wang Y, Chen S, Zhang X, Song Y. et al. SPARC promotes the proliferation and metastasis of oral squamous cell carcinoma by PI3K/AKT/PDGFB/PDGFRβ axis. Journal of cellular physiology. 2019;1:1-13
95. Gao ZW, Liu C, Yang L, He T, Wu XN, Zhang HZ. et al. SPARC Overexpression Promotes Liver Cancer Cell Proliferation and Tumor Growth. Frontiers in molecular biosciences. 2021;8:775-743
96. Zou T, Gao Y, Qie M. MiR-29c-3p inhibits epithelial-mesenchymal transition to inhibit the proliferation, invasion and metastasis of cervical cancer cells by targeting SPARC. Annals of translational medicine. 2021;9:125-136
97. Tang X, Sui X, Weng L, Liu Y. SNAIL1: Linking Tumor Metastasis to Immune Evasion. Frontiers in immunology. 2021;12:1-11
98. Cho ES, Kang HE, Kim NH, Yook JI. Therapeutic implications of cancer epithelial-mesenchymal transition (EMT). Archives of pharmacal research. 2019;42:14-24
99. Zhu Y, Wang C, Becker SA, Hurst K, Nogueira LM, Findlay VJ. et al. miR-145 Antagonizes SNAI1-Mediated Stemness and Radiation Resistance in Colorectal Cancer. Molecular therapy: the journal of the American Society of Gene Therapy. 2018;26:744-54
100. Díaz-López A, Moreno-Bueno G, Cano A. Role of microRNA in epithelial to mesenchymal transition and metastasis and clinical perspectives. Cancer management and research. 2014;6:205-16
101. Vandamme N, Denecker G, Bruneel K, Blancke G, Akay Ö, Taminau J. et al. The EMT Transcription Factor ZEB2 Promotes Proliferation of Primary and Metastatic Melanoma While Suppressing an Invasive, Mesenchymal-Like Phenotype. Cancer research. 2020;80:2983-95
102. Qiu G, Lin Y, Zhang H, Wu D. miR-139-5p inhibits epithelial-mesenchymal transition, migration and invasion of hepatocellular carcinoma cells by targeting ZEB1 and ZEB2. Biochemical and biophysical research communications. 2015;463:315-21
103. He J, Wang J, Li S, Li T, Chen K, Zhang S. Hypoxia-inhibited miR-338-3p suppresses breast cancer progression by directly targeting ZEB2. Cancer science. 2020;111:3550-63
104. Salmena L, Poliseno L, Tay Y, Kats L, Pandolfi PP. A ceRNA hypothesis: the Rosetta Stone of a hidden RNA language? Cell. 2011;146:353-8
105. Hoek KS, Eichhoff OM, Schlegel NC, Döbbeling U, Kobert N, Schaerer L. et al. In vivo switching of human melanoma cells between proliferative and invasive states. Cancer research. 2008;68:650-6
106. Chen C, Zhao S, Karnad A, Freeman JW. The biology and role of CD44 in cancer progression: therapeutic implications. Journal of hematology & oncology. 2018;11:64-75
107. Wu X, He X, Li S, Xu X, Chen X, Zhu H. Long Non-Coding RNA ucoo2kmd.1 Regulates CD44-Dependent Cell Growth by Competing for miR-211-3p in Colorectal Cancer. PloS one. 2016;11:1-16
108. Xu P, Zhang X, Ni W, Fan H, Xu J, Chen Y. et al. Upregulated HOXC8 Expression Is Associated with Poor Prognosis and Oxaliplatin Resistance in Hepatocellular Carcinoma. Digestive diseases and sciences. 2015;60:3351-63
109. Pytel D, Majsterek I, Diehl JA. Tumor progression and the different faces of the PERK kinase. Oncogene. 2016;35:1207-15
110. Bu Y, Yoshida A, Chitnis N, Altman BJ, Tameire F, Oran A. et al. A PERK-miR-211 axis suppresses circadian regulators and protein synthesis to promote cancer cell survival. Nature cell biology. 2018;20:104-15
111. Bader AG. miR-34 - a microRNA replacement therapy is headed to the clinic. Frontiers in genetics. 2012;3:120-35
112. Catela Ivkovic T, Voss G, Cornella H, Ceder Y. microRNAs as cancer therapeutics: A step closer to clinical application. Cancer letters. 2017;407:113-22
Author contact
Corresponding author: Department of Gynecology, Zhangjiagang First People's Hospital, Zhangjiagang Affiliated Hospital of Soochow University, Zhangjiagang 215600, Jiangsu, Peoples Republic of China. E-mail: huanhaiweidoccom.

 Global reach, higher impact
Global reach, higher impact