Impact Factor
ISSN: 1837-9664
J Cancer 2023; 14(10):1848-1858. doi:10.7150/jca.84535 This issue Cite
Research Paper
The prognostic value of including S-1 regimens in stage II and III gastric cancer patients: A propensity score matching and subgroup analyses
1. Second Department of General Surgery, Shanxi Province Cancer Hospital, Shanxi Hospital Affiliated to Cancer Hospital, Chinese Academy of Medical Sciences, Cancer Hospital Affiliated to Shanxi Medical University, Taiyuan, Shanxi, 030013, Peoples R China.
2. Department of pharmacy, Shanxi Province Cancer Hospital, Shanxi Hospital Affiliated to Cancer Hospital, Chinese Academy of Medical Sciences, Cancer Hospital Affiliated to Shanxi Medical University, Taiyuan, Shanxi, 030013, Peoples R China.
Abstract
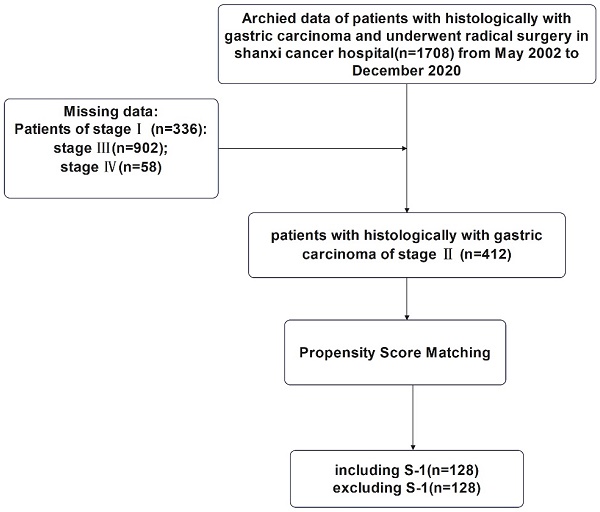
Background: Although numerous studies have indicated the increased efficacy of including S-1 in chemotherapy regimens, the effect of S-1 in the treatment of gastric cancer remains to be determined. In this study, we analyzed the prognostic value of chemotherapy regimens of including S-1 for stage II and III gastric cancer patients.
Methods: A total of 412 patients with stage II gastric cancer and 902 patients with stage III gastric cancer who received D2 gastrectomy plus adjuvant chemotherapy or neoadjuvant chemotherapy were included in this study. Analysis and comparison at a ratio of 1:1 was performed to reduce the baseline differences. Progression-free survival, overall survival, and recurrence were the main outcome indicators.
Results: After propensity score matching (PSM), we found that including S-1 in the chemotherapy regimen was only better than without S-1 in 5-year overall survival (OS) (83.6% VS 68.8%, Log-Rank P=0.005) and 5-year progression free survival (PFS) (71.6% VS 61.5%, Log-Rank P=0.005) for stage II gastric cancer patients. The difference in the recurrence (P=0.102), local-regional recurrence (P=0.062), and distant metastases (P=0.328) between the two groups were not significant. As for the stage III gastric cancer patients, Kaplan-Meier survival curves revealed that including S-1 was inferior to excluding S-1 in OS (P=0.023), but not in PFS(P=0.740). However, the difference in recurrence (P<0.001), local-regional recurrence (P=0.002), and distant metastases (P=0.011) between the two groups were significant. Furthermore, including S-1 increased mortality hazard by 27.2% compared to without S-1 (P=0.023) in the subgroup analyses of OS, but not in the subgroup analyses of PFS (P=0.268).
Conclusions: Including S-1 did not exhibit superior effect over excluding S-1 in the prognosis of stage II and III gastric cancer patients, but significantly increased the risk of mortality in stage III gastric cancer patients. Moreover, for patients with stage III gastric cancer, including S-1 significantly increased the recurrence of the disease.
Keywords: gastric cancer, propensity score matching, chemotherapy regimen, S-1

 Global reach, higher impact
Global reach, higher impact