3.2
Impact Factor
ISSN: 1837-9664
J Cancer 2023; 14(18):3404-3415. doi:10.7150/jca.87692 This issue Cite
Research Paper
miR-134-3p driven by anisomycin impairs ovarian cancer stem cell activity through inhibiting GPR137 expression
1. Department of Acupuncture and Moxibustion, Shanghai General Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai 200086, China.
2. Shanghai Geriatric Institute of Chinese Medicine, Shanghai University of Traditional Chinese Medicine, Shanghai 200031, China.
3. Department of Obstetrics and Gynecology, Xinhua Hospital Affiliated to Shanghai Jiao Tong University School of Medicine, Shanghai 200092, China.
4. Department of Dermatology, Yueyang Hospital of Integrated Traditional Chinese and Western Medicine, Shanghai University of Traditional Chinese Medicine, Shanghai 200437, China.
5. Gongli Hospital Affiliated to the Second Military Medicical University in Pudong New Area of Shanghai City, Shanghai 200135, China.
* These authors contributed equally to this work and shared the first authorship.
Abstract
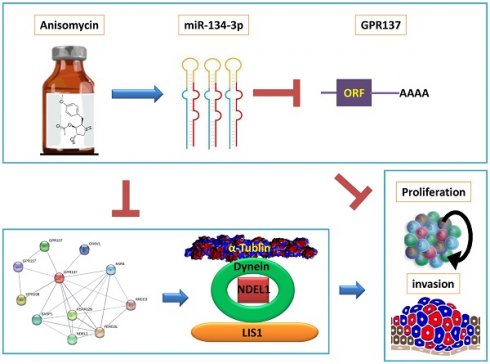
Background: Ovarian cancer recurrence and metastasis are predominantly attributed to ovarian cancer stem cells; however, the mechanism by which anisomycin regulates human ovarian cancer stem cells (HuOCSCs) remains unclear.
Methods: cDNA microArray was used to screen microRNAs (miRNAs) targeted by anisomycin, and RT-qPCR validated the miRNA targets. TargetScan database, GO enrichment analysis, and RT-qPCR, accompanied by a fluorescent reporter system, were employed to verify the miRNA target genes. In vitro experimental cell proliferation inhibition assay, flow cytometry, Transwell, angiogenesis assay, and in vivo transplantation tumor assay were implemented to assess the ability of the overexpressed miRNAs to hinder HuOCSC activity. Western blot, RT-qPCR, and immunofluorescence were applied to measure the transcriptional and protein-level expression of the miRNA target genes and their related genes. Bioinformatic analysis predicted and deciphered the role of the miRNA target genes and related genes in the development and prognosis of ovarian cancer.
Results: The expression levels of multiple DLK1-DIO3 imprinted microRNA cluster members were altered by anisomycin, among which miR-134-3p expression was most significantly elevated. miR-134-3p overexpression significantly suppressed HuOCSC activity. The screening and validation of target genes uncovered that miR-134-3p was able to markedly suppress GPR137 expression. Additionally, miR-134-3p regulated the cytoskeleton, migration-related protein in the NDEL1/DYNEIN/TUBA1A axis through targeting GPR137. Bioinformatics prediction unveiled a close association of GPR137, NDEL1, DYNC1H1, and TUBA1A with ovarian cancer development and prognosis.
Conclusions: The activity of HuOCSCs may be compromised by anisomycin through the regulation of miR-134-3p, which inhibits the GPR137/NDEL1/DYNEIN/TUBA1A axis.
Keywords: Human ovarian cancer stem cells, DLK1-DIO3 imprinted microRNA cluster, miR-134-3p, Anisomycin, GPR137

 Global reach, higher impact
Global reach, higher impact