3.2
Impact Factor
ISSN: 1837-9664
J Cancer 2024; 15(7):2024-2032. doi:10.7150/jca.92624 This issue Cite
Research Paper
Efficacy and Safety Assessment of Intrathoracic Perfusion Chemotherapy Combined with immunological factor Interleukin-2 in the Treatment of Advanced Non-Small Cell Lung Cancer: A Retrospective Cohort Study
1. Department of Pharmacy, Nanjing Drum Tower Hospital, Affiliated Hospital of Medical School, Nanjing University, Nanjing, China.
2. School of Medicine, Macau University of Science and Technology, 999078, Macau, Macao.
3. Department of Earth Sciences, Kunming University of Science and Technology, 650093, Kunming, China.
4. Affiliated Hospital of Weifang Medical University, School of Clinical Medicine, Weifang Medical University, Weifang, China.
5. College of Veterinary Medicine, Sichuan Agricultural University, 610000, Chengdu, China.
6. Undergraduate Department, University of Toronto, M2J4A6, Toronto, Canada.
7. Institute of Medical Information/Library, Chinese Academy of Medical Sciences, 100020 Beijing, China.
8. Division of Sports Science & Physical Education, Tsinghua University, Beijing 100084, China.
9. The Third Affiliated Hospital of Shandong First Medical University, Jinan, 250000, Shandong, China.
10. Faculty of Dentistry, University of Debrecen, Debrecen, Hungary.
11. College of Agriculture, Henan University of Science and Technology, 471023, Luoyang, China.
12. Department of Cardiothoracic Surgery, the Affiliated Hospital of Youjiang Medical University for Nationalities, 18 zhongshan 2nd Road, Baise, Guangxi Province, China.
†These authors contributed equally to this work.
Abstract
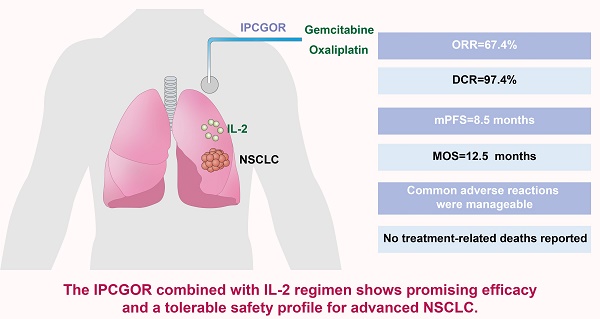
Objective: This study evaluated the efficacy and safety of the gemcitabine and oxaliplatin intrathoracic perfusion chemotherapy (IPCGOR) regimen combined with interleukin-2 (IL-2) for advanced non-small cell lung cancer (NSCLC).
Methods: We conducted a retrospective analysis of 460 advanced NSCLC patients from the Yunnan Province Early Cancer Diagnosis and Treatment Project (June 2020-October 2022), assessing the IPCGOR and IL-2 combination. Outcomes were measured based on RECIST 1.1 criteria, focusing on objective response rate (ORR), disease control rate (DCR), median progression-free survival (mPFS), median overall survival (MOS), and treatment safety.
Results: The treatment demonstrated an ORR of 67.4%, a DCR of 97.4%, an mPFS of 8.5 months, and an MOS of 12.5 months. 14 patients underwent successful surgery post-treatment. Common adverse reactions were manageable, with no treatment-related deaths reported.
Conclusion: The IPCGOR combined with IL-2 regimen shows promising efficacy and a tolerable safety profile for advanced NSCLC. These findings suggest its potential as a reference for treating advanced NSCLC. However, the study's retrospective nature and single-center design pose limitations. Future research should focus on prospective studies, randomized controlled trials, and long-term outcome assessments, particularly in diverse patient subgroups, to further validate and refine the clinical application of this regimen.
Keywords: Interleukin-2 Treatment, immunotherapy, Advanced Non-Small Cell Lung Cancer, Intrathoracic Perfusion Chemotherapy, Efficacy and Safety Evaluation, Retrospective Cohort Study

 Global reach, higher impact
Global reach, higher impact