Impact Factor ISSN: 1837-9664
J Cancer 2024; 15(8):2206-2213. doi:10.7150/jca.92656 This issue Cite
Research Paper
Prognostic Significance of KIF-12 Functioning as a Tumour Suppressor in Papillary Thyroid Carcinoma
1. Department of Ultrasound, State Key Laboratory of Complex Severe and Rare Diseases, Peking Union Medical College Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, China.
2. Department of General Surgery, State Key Laboratory of Complex Severe and Rare Diseases, Peking Union Medical College Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, China.
3. Department of Pathology, State Key Laboratory of Complex Severe and Rare Diseases, Peking Union Medical College Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, China.
#These authors contributed equally to this work.
Received 2023-11-27; Accepted 2024-2-11; Published 2024-2-25
Abstract

Objective: To explore the potential value of a novel marker, KIF-12, in the progression and prognosis of papillary thyroid carcinoma (PTC) through integrative bioinformatics analysis, and clinical sample validation of the prognostic value of KIF-12.
Materials and Methods: We extracted the clinicopathological data of 502 PTC patients from The Cancer Genome Atlas-Thyroid Cancer (TCGA-THCA) dataset to identify reliable differentially expressed genes (DEGs) between high and low KIF12 expression groups. Functional enrichment analysis was performed on upregulated DEGs. Gene set enrichment analysis (GESA) was performed to identify the biological pathways. We further applied Cox analysis to determine independent risk factors associated with the PTC progression-free interval (PFI), and a nomogram was established to predict disease outcome. Finally, the prognostic value of KIF12 was validated by means of clinical samples from PTC patients with and without lateral lymph node metastasis.
Results: On the basis of the TCGA-THCA database, we found that low KIF-12 expression was significantly related to a higher TNM stage (p<0.05), BRAF mutation status (p = 0.019), and extrathyroidal extension (p<0.001). KIF-12 was an independent prognostic factor of PTC (OR=0.319, 95% CI=0.130-0.784, P=0.013). The prognostic value of KIF12 was also successfully validated in clinical samples from twenty-nine PTC patients with lateral lymph node metastasis by comparison with twenty-two PTC patients without lymph node metastasis (P = 0.004).
Conclusions: We report that KIF-12 has a tumor suppressive function in PTC and may be a useful prognostic tool to predict patient outcomes.
Keywords: KIF-12, Prognosis, Differentiated thyroid carcinoma, Thyroid
Introduction
Thyroid cancer (THCA) is the most common endocrine malignancy and ranks eleventh among the leading types of cancer worldwide [1]. The incidence of THCA has been rising globally over recent decades, and the causes are multifactorial [2]. Differentiated thyroid cancer (DTC), including papillary thyroid carcinoma (PTC) and follicular thyroid carcinoma (FTC), are the two main types of THCA, and they have relatively good prognoses. Approximately 50% of the rise in THCA is attributed to the greater detection of small PTC, especially papillary thyroid microcarcinoma (PTMC) with a maximum diameter ≤ 1 cm. However, with the increasing incidence of THCA, thyroid cancer-related mortality rates have not surged simultaneously, and this has motivated discussions regarding the overdiagnosis of THCA [3].
This is because current diagnostic techniques (ultrasound, computed tomography, and magnetic resonance imaging) are accessed on a massive scale, resulting in more detection of THCA at the early stage. Given the indolent behaviour and > 99% 10-year disease-specific survival rate of patients with low-risk PTMC [4], active surveillance (AS) instead of immediate surgery has been proposed as an alternative treatment in recent years [5]. AS benefits patients in several ways; for example, postoperative complications and lifelong medication are avoided by AS [6]. Nonetheless, the application of AS is based on the accurate assessment of low-risk and high-risk PTMC preoperatively, thus ensuring that high-risk patients receive surgery and additional therapeutics in a timely manner while low-risk patients are provided with more options to avoid overtreatment. Several popular guidelines, namely, the 2015 American Thyroid Association (ATA) guidelines and the Thyroid Imaging Reporting and Data System (TI-RADS), proposed by KWAK and ACR, have recommended a staging system to assess the malignancy risk of thyroid nodules preoperatively using neck ultrasound examination. Surgery and additional therapeutics are recommended for DTC with growth or aggressive behaviour. But, none of them specifically mentions how to evaluate the growth rate or aggressive behaviour of thyroid carcinoma prior to surgery [7]. Therefore, identifying reliable biomarkers for predicting the biological behaviour of THCA is clinically crucial.
Previous studies have demonstrated gene signatures predicting thyroid cancer prognosis [8-10]. However, these studies lack further exploration of certain genes. Advanced analysis of public databases could reveal more succinct and practical gene models to indicate the progression risk of PTC. Moreover, correlations with clinical pathological parameters and immune-related interactions will be elucidated, thus guiding individualized management. In this study, we aimed to analyse thyroid cancer datasets from the public databases to identify reliable differentially expressed genes (DEGs) in PTCs and identify DEGs associated with the progression-free interval (PFI) of PTC. We selected a potential gene KIF-12 predicting progression behavior of THCA using gene expression and clinical data from the TCGA-THCA dataset, and validated in our clinical samples. KIF12 is an orphan kinesin-12 family motor protein, which consists of microtubule-associated motor proteins involved in organizing the cytoskeleton and intracellular transport [11]. However, the value of KIF12 in tumors has never been reported.
Materials and Methods
Data acquisition and fundamental processing
Harmonized gene expression data (HTSeq-FPKM and HTSeq counts) and associated clinicopathological information of thyroid carcinoma were downloaded from the TCGA database (https://portal.gdc.cancer.gov/). The inclusion criteria of subjects were as follows: (i) histologically confirmed PTC, (ii) age>18 years old. Cases (i) without clinical information were excluded. There were 502 samples with gene transcriptome data retained for further analysis (58 normal and 502 tumour). After transforming level 3 HTSeq-FPKM into the format of transcripts per million reads (TPM), the expression level of KIF12 between tumour and normal samples was compared. Furthermore, differentially expressed genes (DEGs) between the high and low KIF12 expression THCA groups were identified using the DESeq2 package in R. The cut-off criteria were |log2-fold change (FC)| >1 and P< 0.05 [12].
Enrichment and interaction analysis
Functional enrichment analysis comprising gene ontology (GO) analysis and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway analysis was performed on upregulated DEGs using the clusterProfiler package. Gene set enrichment analysis (GSEA) was achieved by the clusterProfiler package in R to determine whether an a priori defined set of genes differs significantly between high- and low- KIF12 expression subsets [13]. The enriched pathways based on the varied expression level of KIF12 were classified by the adjusted p value (<0.05), false discovery rate (FDR) q-value (<0.25) and normalized enrichment score (|NES|>1) [14]. The immune infiltration analysis of THCA was conducted by the single-sample gene set enrichment analysis (ssGSEA) method using the GSVA package. Twenty-four types of immunocytes with signature genes labelled in the literature were correlated with KIF12 expression in all THCA samples [15]. To exhibit the interaction among the DEGs, protein‒protein interaction (PPI) networks were constructed employing the Search Tool for the Retrieval of Interacting Genes (STRING) (http:string-db.org/) database [16]. An interaction combined score>0.7 was set to explore densely connected network elements using the Molecular Complex Detection (MCODE) algorithm [17]. This study satisfies the publication requirements stated by TCGA (http://cancergenome.nih.gov/publications/publicationguidelines).
Validation in clinical samples
We retrospectively reviewed the medical records of PTC patients who underwent thyroid surgery at our centre between August 2021 and October 2021. The inclusion criteria of subjects were as follows: (i) thyroidectomy performed together with central and, when necessary, therapeutic lateral neck dissection, (ii) histologically confirmed PTC, (iii) target lesion size of r ≤1 cm (iv) age>18 years old. The exclusion criteria: (i) previous thyroid surgery or related invasive operation for the treatment of target lesions. Thirty PTC patients with histologically confirmed lateral lymph node metastases were included in the experimental group. Thirty PTC patients without evidence of lymph node metastases were enrolled in the control group. Pathological tissues of PTC patients meeting the inclusion criteria were collected, and tissue sections and paraffin blocks were produced. RNA was extracted from tissues from patients using an RNeasy FFPE Kit (RNeasy FFPE Kit, QIAGEN, Hilden, Germany). cDNA was then synthesized using a reverse transcriptase system (High-Capacity cDNA Reverse Transcription Kit, Thermo Fisher Scientific, Waltham, USA). RT‒qPCR was used to validate gene expression in accordance with the manufacturer's instructions (SYBR™ Green PCR Master Mix, Thermo Fisher Scientific, Waltham, USA). Relative gene expression was calculated using the 2-ΔΔCT method. Primers for RT‒qPCR were purchased from Sangon Biotech (Shanghai, China). Finally, there were twenty-nine patients in the experimental group and twenty-two patients in the control group.
Statistical analysis
We used R (v.3.5.1) and SPSS software version 19.0 (IBM, Armonk, NY, USA) to complete the statistical analysis. The Wilcoxon rank-sum test was used for comparisons of KIF12 expression levels between THCA and normal samples. Clinically, the normality of variables was verified by the Shapiro‒Wilk test. For parametric data, an unpaired t test was used to evaluate differences between the two groups. For nonparametric data, differences between groups were analysed using a Mann‒Whitney U test. The progression-free interval (PFI) was defined as the period from the date of diagnosis until the date of the newly discovered tumour event, mainly including locoregional recurrence, distant metastasis or progression of the disease in our selected THCA samples. The chi-square test was applied to analyse categorical variables, and the Kruskal‒Wallis test was used for comparisons of continuous variables. Multivariate Cox regression analyses and the Kaplan‒Meier method were adopted to identify independent prognostic factors. Moreover, a nomogram was developed according to the results of the multivariate analysis for visualizing the PFI probability at 5 years individually. The calibration curves were also created by plotting predicted probabilities against the observed rates. A 2-sided P<0.05 was viewed as the threshold for a statistically significant difference between the low- and high-expression KIF12 groups.
Results
Association of KIF-12 expression with clinical characteristics
To investigate whether KIF-12 expression is related to the clinical parameters of PTC, variables such as sex, age, race, neoplasm location, focus number, thyroid gland disorder history, extrathyroidal extension, BRAF status, RAS status, TNM stage, pathologic stage, residual tumour and histologic type were analysed. As shown in Table 1, the results of the analyses showed that there were significant differences in T stage (p = 0.001), N stage (p = 0.03), pathologic stage (p = 0.003), residual tumour (p = 0.023), histologic type (p = 0.003), race (p = 0.025), extrathyroidal extension (p<0.001), and BRAF status (p = 0.019) in the TCGA cohort Table 1.
Association of KIF-12 expression with clinical characteristics
| Characteristics | Level | Low expression of KIF12 | High expression of KIF12 | p | test |
|---|---|---|---|---|---|
| n | 251 | 251 | |||
| T stage (%) | T1 | 57(22.8%) | 86(34.4%) | 0.001 | exact |
| T2 | 77(30.8%) | 87(34.8%) | |||
| T3 | 99(39.6%) | 71(28.4%) | |||
| T4 | 17(6.8%) | 6(2.4%) | |||
| N stage (%) | N0 | 105(45.5%) | 124(56.1%) | 0.030 | |
| N1 | 126(54.5%) | 97(43.9%) | |||
| M stage (%) | M0 | 128(97.7%) | 154(96.2%) | 0.521 | exact |
| M1 | 3(2.3%) | 6(3.8%) | |||
| Pathologic stage (%) | Stage I | 124(49.4%) | 157(63.1%) | 0.003 | |
| Stage II | 26(10.4%) | 26(10.4%) | |||
| Stage III | 63(25.1%) | 49(19.7%) | |||
| Stage IV | 38(15.1%) | 17(6.8%) | |||
| Residual tumour (%) | R0 | 186(83.8%) | 198(90.8%) | 0.023 | exact |
| R1 | 32(14.4%) | 20(9.2%) | |||
| R2 | 4(1.8%) | 0(0.0%) | |||
| Histological type (%) | Classical | 175(69.7%) | 181(72.1%) | 0.003 | exact |
| Follicular | 45(17.9%) | 56(22.3%) | |||
| Other | 3(1.2%) | 6(2.4%) | |||
| Tall Cell | 28(11.2%) | 8(3.2%) | |||
| Sex (%) | Female | 185(73.7%) | 182(72.5%) | 0.840 | |
| Male | 66(26.3%) | 69(27.5%) | |||
| Race (%) | Asian | 16(8.1%) | 35(16.4%) | 0.025 | |
| Black or African American | 16(8.1%) | 11(5.2%) | |||
| White | 165(83.8%) | 167(78.4%) | |||
| Neoplasm location (%) | Bilateral | 36(14.5%) | 50(20.2%) | 0.117 | |
| Isthmus | 15(6.0%) | 7(2.8%) | |||
| Left lobe | 85(34.3%) | 90(36.3%) | |||
| Right lobe | 112(45.2%) | 101(40.7%) | |||
| Primary neoplasm focus type (%) | Multifocal | 113(45.6%) | 113(46.3%) | 0.940 | |
| Unifocal | 135(54.4%) | 131(53.7%) | |||
| Thyroid gland disorder history (%) | Lymphocytic Thyroiditis | 40(18.0%) | 31(14.0%) | 0.260 | |
| Nodular Hyperplasia | 34(15.3%) | 34(15.3%) | |||
| Normal | 132(59.5%) | 148(66.7%) | |||
| Other, specify | 16(7.2%) | 9(4.1%) | |||
| Extrathyroidal extension (%) | No | 145(59.4%) | 186(77.5%) | <0.001 | |
| Yes | 99(40.6%) | 54(22.5%) | |||
| BRAF status (%) | Mut | 156(64.2%) | 129(53.3%) | 0.019 | |
| WT | 87(35.8%) | 113(46.7%) | |||
| NRAS status (%) | Mut | 22(9.1%) | 17(7.0%) | 0.513 | |
| WT | 221(90.9%) | 225(93.0%) | |||
| HRAS status (%) | Mut | 10(4.1%) | 7(2.9%) | 0.623 | exact |
| WT | 233(95.9%) | 235(97.1%) | |||
| Age (median [IQR]) | 49.00[35.00,61.00] | 44.00[35.00,55.50] | 0.060 | nonnorm |
Prognostic value of KIF-12 in d-THCA
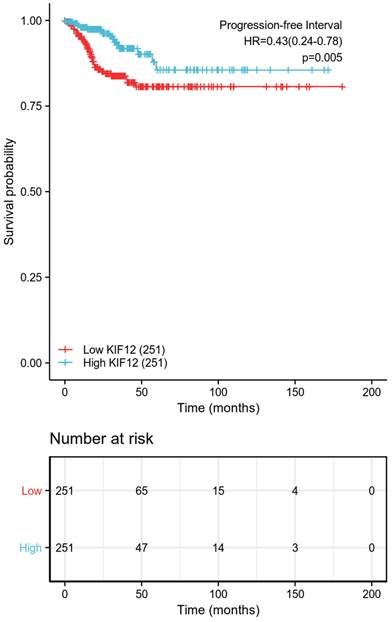
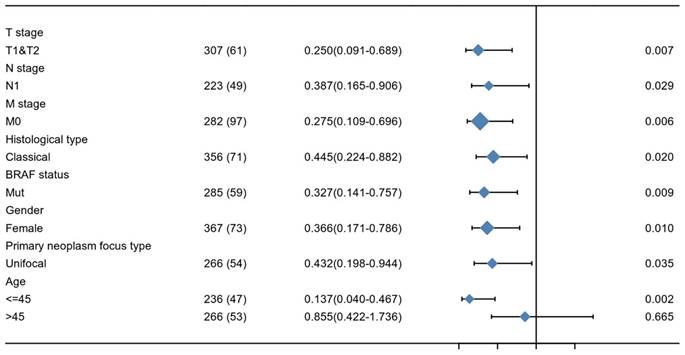
The relationship between KIF-12 expression as well as clinicopathological factors and progression-free interval was further explored by COX regression analysis. In the univariate Cox regression analysis, T stage, M stage, pathologic stage, extrathyroidal extension, and KIF-12 expression satisfied the threshold of p<0.1 and were included in the multivariate study. Multivariate Cox analysis revealed that M stage (OR=6.818, 95% CI=2.103-22.106, p=0.001) and KIF-12 expression (OR=0.319, 95% CI=0.130-0.784, P=0.013) were independent prognostic factors for PFI [Table 2], and the results were demonstrated in the form of a nomogram for predicting the individual probability of PFI [Figure 1]. In terms of subgroup analysis, KIF-12 showed significant expression differences in the T1 and T2 tumour stage subsets, M0 and N1 stage subsets, classical PTC, female population, BRAF mutation group, patients with unifocal neoplasm and patients aged ≤ 45 [Figure 2].
Prognostic value of KIF-12 in d-THCA
| Characteristics | Total (N) | HR (95% CI) Univariate analysis | P value Univariate analysis | HR (95% CI) Multivariate analysis | P value Multivariate analysis |
|---|---|---|---|---|---|
| T stage (T3&T4 vs. T1&T2) | 500 | 2.507(1.434-4.385) | 0.001 | 0.752(0.181-3.115) | 0.694 |
| N stage (N1 vs. N0) | 452 | 1.578(0.885-2.814) | 0.122 | ||
| M stage (M1 vs. M0) | 291 | 7.542(2.860-19.891) | <0.001 | 6.818(2.103-22.106) | 0.001 |
| Pathologic stage (Stage III & Stage IV vs. Stage I & Stage II) | 500 | 2.615(1.515-4.515) | <0.001 | 2.712(0.811-9.068) | 0.105 |
| Residual tumour (R1&R2 vs. R0) | 440 | 1.773(0.856-3.674) | 0.123 | ||
| Histological type (Classical vs. Follicular & Other & Tall Cell) | 502 | 1.022(0.553-1.888) | 0.944 | ||
| Age (>45 vs. <=45) | 502 | 1.618(0.924-2.834) | 0.092 | 1.109(0.370-3.323) | 0.853 |
| Sex (Male vs. Female) | 502 | 1.529(0.863-2.709) | 0.146 | ||
| Race (White vs. Asian & Black or African American) | 410 | 1.183(0.529-2.646) | 0.682 | ||
| Neoplasm location (Bilateral vs. Isthmus & Left lobe & Right lobe) | 496 | 1.150(0.558-2.370) | 0.704 | ||
| Primary neoplasm focus type (Multifocal vs. Unifocal) | 492 | 1.036(0.595-1.802) | 0.901 | ||
| Thyroid gland disorder history (Lymphocytic Thyroiditis & Nodular Hyperplasia & Other, specify vs. Normal) | 444 | 0.966(0.520-1.795) | 0.913 | ||
| Extrathyroidal extension (Yes vs. No) | 484 | 1.995(1.151-3.458) | 0.014 | 1.054(0.303-3.663) | 0.935 |
| BRAF status (Mut vs. WT) | 485 | 1.266(0.703-2.280) | 0.432 | ||
| NRAS status (Mut vs. WT) | 485 | 1.958(0.830-4.615) | 0.125 | ||
| HRAS status (Mut vs. WT) | 485 | 1.033(0.251-4.258) | 0.964 | ||
| KIF12 (High vs. Low) | 502 | 0.431(0.239-0.777) | 0.005 | 0.319(0.130-0.784) | 0.013 |
Progression-free survival between the high-low expression groups of KIF-12.
Subgroup analysis of KIF-12 expression.
Identification of DEGs in differentiated thyroid carcinoma
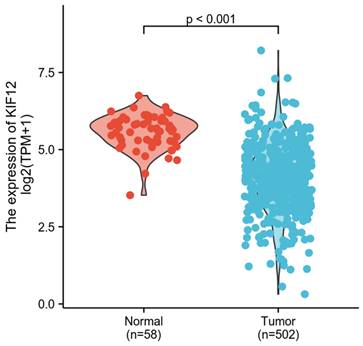
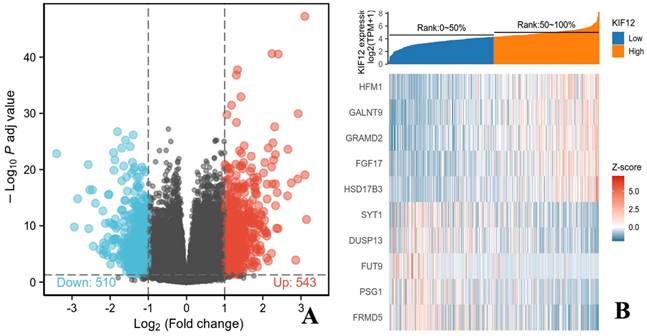
In total, 502 tumour samples and 58 adjacent noncancerous samples were obtained from PTC patients in the TCGA cohort with matched RNA-sequencing data and clinical information. As shown in Figure 3, KIF-12 expression was significantly lower in tumour specimens than in normal control specimens. This suggests the potential role of KIF-12 as a tumour-suppressing gene in PTC. The comparison of the high- and low-KIF-12 expression groups generated 1054 DEGs in total (543 upregulated and 510 downregulated) for further analysis using the cut-off criteria of padj < 0.05 and |logFC| > 1 [Figures 4].
Comparison of KIF-12 expression in tumour specimens and normal specimens.
Genes related to KIF-12 in thyroid cancer. (A). Volcano plot showing KIF-12 and associated genes' expression. Dark red dots represent genes positively correlated with KIF-12 and dark green dots represent genes negatively correlated with KIF-12. (B). Heat map showing top genes correlated with KIF-12.
Predicted KIF-12 functions and pathways based on DEGs
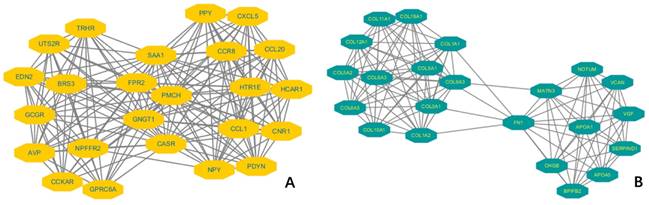
To understand the molecular mechanism of KIF-12 in d-THCA, gene ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) functional enrichment analysis were performed on the DEGS. The GO analysis categorized the genes into three domains, namely, biological process, cellular component and molecular function. Regarding biological process, a variety of processes, including extracellular structure organization (p = 1.90E-12), extracellular matrix organization (p = 6.37E-12), and collagen fibril organization (p = 4.10E-06), were shown to be involved. Regarding cellular component, collagen-containing extracellular matrix (p = 7.91E-15) and collagen trimer (p = 1.19E-13) were mostly indicated. Regarding molecular function, extracellular matrix structural constituent (p = 9.37E-11), receptor ligand activity (p = 7.41E-10) and extracellular matrix structural constituent conferring tensile strength (p = 2.67E-08) were involved. Moreover, in the GSEA, the gene signatures were shown to be linked to the metabolic functions of amine-derived hormones, the TFF pathway and transporter disorders. Moreover, KEGG analysis suggested that the DEGs pointed to pathways involving protein digestion and absorption (p = 1.50E-05) and neuroactive ligand‒receptor interaction (p = 1.1E-03). However, regarding immune infiltration, KIF-12 showed a low correlation coefficient with different types of immune cells, such as Th1, Th2, Th17, Treg cells, NK cells, B cells, aDC cells, cytotoxic cells, and macrophages. Among these immune cells, Th2 cells exhibited the highest inverse correlation with KIF-12 expression levels (Spearman r = 0.358). Furthermore, we constructed a protein‒protein interaction (PPI) regulatory network using the String database to elucidate the protein‒protein interaction in d-THCA patients and presented the two most significant molecular complex detection (MCODE) components [Figures 5].
Two most significant molecular complex detection (MCODE) components of a protein‒protein interaction (PPI) regulatory network in thyroid cancer patients. (A). MCODE 1. (B). MCODE 2.
Verification in clinical samples
Postoperative histopathologic results showed that all lesions were papillary thyroid carcinoma. There were 69% female with lateral lymph node metastases (LLNM) and 72.3% female patients without lymph node metastases (LNM). The sizes of the nodules in the patients with LLNM and patients without LNM were 0.60 ± 0.24 cm and 0.54 ± 0.25 cm, respectively. There were no significant differences in sex or tumour size (P = 0.51; P = 0.41). The average age of patients with LLNM 34.5 ± 8.3 years old, and patients without LNM were 43.8 ± 9.7 years old (P=0.001) [Table 3].
Characteristics of clinical samples
| Lateral lymph node metastasis+ | Lymph node metastasis - | P value | |
|---|---|---|---|
| No. | 29 | 22 | |
| Age, years (mean) | 34.5 | 43.8 | 0.001 |
| Sex (n, %) | 0.51 | ||
| Male | 9 (31%) | 6 (27.3%) | |
| Female | 22 (69%) | 16 (72.7%) | |
| Tumour size (cm) | 0.60 | 0.54 | 0.41 |
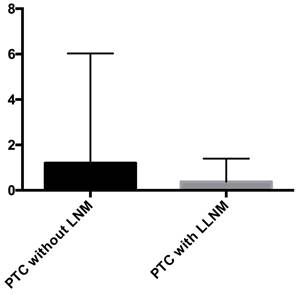
Pathological tissues were collected from PTC patients, and the KIF-12 expression of was verified using quantitative reverse transcription polymerase chain reaction (RT‒qPCR). The results showed that compared with that in the PTC samples from patients without LNM, the expression of KIF-12 in PTC samples from patients with LLNM was significantly lower (P = 0.004) (median: 0.38 vs 1.20) [Figures 6]. Our results indicated that low KIF-12 expression was related to LLNM in PTC.
RT‒PCR verification in clinical samples by comparison the patients with lateral lymph node metastases (LLNM) and without lymph node metastases (LNM).
Discussion
Although the increasing incidence of PTC has been widely reported, AS management has been considered an alternative to immediate surgery for patients with PTMC. However, some patients with PTMC have poor clinical features, such as clinically significant regional lymph nodes (LNs). However, it is not yet clear how to predict the aggressive behaviour of PTC prior to surgery. Therefore, identifying reliable biomarkers to predict the biological behaviour of PTC is clinically crucial. In this study, we explored the potential role of KIF-12 in the progression and prognosis of PTC. We found that low PTC expression was significantly related to PTC progression. Moreover, KIF-12 was markedly decreased in PTC patients with lateral lymph node metastasis (LLNM).
The role of KIF-12 in tumours has seldom been reported. On the basis of data in the TCGA database, we found the potential role of KIF-12 as a tumour suppressor gene in differentiated thyroid carcinoma, and KIF-12 expression was an independent prognostic factor for thyroid cancer. The results showed that the downregulation of KIF-12 expression enhances the progression of differentiated thyroid carcinoma, and the effect may be more significant among younger female patients with unifocal PTC showing lymph node metastasis and BRAF mutation. The results were also successfully validated in clinical samples from PTC patients with LLNM. The kinesin superfamily (KIFs) is a class of molecular motors that mainly mediates intracellular molecular transport. Kinesins are motor proteins that transport cargoes by walking unidirectionally along microtubule tracks, hydrolyzing one molecule of ATP at each step. Abnormal expression of genes in the KIF protein family in tumours may alter the distribution of genetic material, affect spindle formation, and cause defects in cell division, thereby mediating tumour development. In addition, kinesins are key participants in chromosomal and spindle movements during mitosis. KIF12 is an orphan kinesin-12 family motor protein [11]. KIF12 is an orphan kinesin-12 family motor protein, which consists of microtubule-associated motor proteins involved in organizing the cytoskeleton and intracellular transport [18]. In addition, KIF12 has been shown to be required for normal mitosis, Myosin II localization in the cleavage furrow, and cytokinesis in Dictyostelium [19]. KIF12 has previously been reported to be involved in the pathogenesis of polycystic kidney disease as a modifier gene [20], affecting insulin secretion from β-cells and thus mediating the progression of diabetes mellitus [21]. Previous cases described pathogenic KIF12 variants as a potential cause for cholestatic liver disease [22]. Its function in tumours has not been reported, and only a part of the KIF12 function is understood. This is the first study to report KIF-12 as a prognostic factor for thyroid cancer.
Furthermore, to understand the molecular mechanism of KIF-12 in thyroid cancer, GO and KEGG functional enrichment analysis were performed on the DEGs. The molecular function terms included extracellular matrix structural constituent, receptor ligand activity and extracellular matrix structural constituent conferring tensile strength. GSEA was conducted to better understand the potential function of KIF-12 in thyroid cancer. GSEA demonstrated that gene signatures were linked to the metabolic functions of amine-derived hormones, the TFF pathway and transporter disorders, which may provide some hints for interactions of KIF-12 on thyroid cancer development and progression.
Although the prognostic value of KIF-12 in thyroid cancer has been demonstrated in our study for the first time, there were still some limitations in our work. The data in the TCGA database and clinical cases are limited, and access to more public data and patient data will be helpful toward strengthening our results. Second, the underlying mechanisms regarding KIF-12 actions are not well investigated, further mechanistic study to assess the impact of KIF-12 on thyroid cancer are needed.
In conclusion, our study explored the prognostic role of KIF-12 in thyroid cancer for the first time. On the basis of integrated bioinformatics analyses of data in TCGA database, we also conducted further validation experiments in clinical cases to further confirm that low KIF-12 expression was correlated with the progression and prognosis of thyroid cancer. Collectively, our findings indicate that KIF-12 is a promising prognostic suppressor of thyroid cancer.
Abbreviations
PTC: Papillary thyroid carcinoma; TCGA-THCA: Cancer Genome Atlas-Thyroid Cancer; DEGs: Differentially expressed genes; GESA: Gene set enrichment analysis; PFI: Progression-free interval; AS: Active surveillance; ATA: American Thyroid Association; TI-RADS: Thyroid Imaging Reporting and Data System; PTMC: Papillary thyroid microcarcinoma; TPM: Transcripts per million reads; GO: Gene ontology; KEGG: Kyoto Encyclopedia of Genes and Genomes; FDR: False discovery rate; ssGSEA: Single-sample gene set enrichment analysis; MCODE: Molecular Complex Detection; PPI: Protein‒protein interaction; STRING: Search Tool for the Retrieval of Interacting Genes; LLNM: Lateral lymph node metastases; LNM: Lymph node metastases; LNs: Lymph nodes; KIFs: Kinesin superfamily.
Acknowledgements
Funding
National High Level Hospital Clinical Research Funding (2022-PUMCH-B-066) and Fundamental Research Funds for the Central Universities (3332023011).
Ethical approval
Informed consent was obtained, and the study protocol conformed to the ethical guidelines of the 1975 Declaration of Helsinki. This study was approved by the ethics committee of Peking Union Medical College Hospital (87-JS-2881).
Author contributions
XY conceived of and designed the study. G-LY and L-RF were major contributors in performed the analysis, writing the manuscript, and prepared the figures and tables. M-LY, J-YX, L-ZY, L-XL, Z-LR and L-XY participated in the study design and edited the manuscript. HY, S-XL, JJ, P-AN, A-YA and L-NW participated in the verification of data, image quality verification, selection and collection of samples. All authors read and approved the final manuscript.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Bray F, Ferlay J, Soerjomataram I. et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394-424
2. Kim J, Gosnell JE, Roman SA. Geographic influences in the global rise of thyroid cancer. Nat Rev Endocrinol. 2020;16:17-29
3. Vaccarella S, Franceschi S, Bray F. et al. Worldwide thyroid-cancer epidemic?. The increasing impact of overdiagnosis. N Engl J Med. 2016;375:614-617
4. Yu XM, Wan Y, Sippel RS. et al. Should all papillary thyroid microcarcinomas be aggressively treated?. An analysis of 18,445 cases. Ann Surg. 2011;254:653-660
5. Cho SJ, Suh CH, Baek JH. et al. Active surveillance for small papillary thyroid cancer: a systematic review and meta-analysis. Thyroid. 2019;29:1399-1408
6. Jeon MJ, Lee YM, Sung TY. et al. Quality of life in patients with papillary thyroid microcarcinoma managed by active surveillance or lobectomy: a cross-sectional study. Thyroid. 2019;29:956-962
7. Haugen BR, Alexander EK, Bible KC. et al. 2015 American thyroid association management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: the American thyroid association guidelines task force on thyroid nodules and differentiated thyroid cancer. Thyroid. 2016;26:1-133
8. Choi KY, Kim JH, Park IS. et al. Predictive gene signatures of nodal metastasis in papillary thyroid carcinoma. Cancer Biomark. 2018;22:35-42
9. Wu M, Yuan H, Li X. et al. Identification of a five-gene signature and establishment of a prognostic nomogram to predict progression-free interval of papillary thyroid carcinoma. Front Endocrinol (Lausanne). 2019;10:790
10. Lin P, Guo YN, Shi L. et al. Development of a prognostic index based on an immunogenomic landscape analysis of papillary thyroid cancer. Aging. 2019;11:480-500
11. Maddirevula S, Alhebbi H, Alqahtani A. et al. Identification of novel loci for pediatric cholestatic liver disease defined by KIF12, PPM1F, USP53, LSR, and WDR83OS pathogenic variants. Genet Med. 2019;21:1164-1172
12. Love MI, Huber W, Anders S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014;15:550
13. Yu G, Wang LG, Han Y. et al. ClusterProfiler: an R package for comparing biological themes among gene clusters. OMICS. 2012;16:284-287
14. Subramanian A, Tamayo P, Mootha VK. et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci USA. 2005;102:15545-15550
15. Bindea G, Mlecnik B, Tosolini M. et al. Spatiotemporal dynamics of intratumoral immune cells reveal the immune landscape in human cancer. Immunity. 2013;39:782-795
16. Szklarczyk D, Gable AL, Lyon D. et al. STRING v11: protein-protein association networks with increased coverage, supporting functional discovery in genome-wide experimental datasets. Nucleic Acids Res. 2019;47:D607-D613
17. Zhou Y, Zhou B, Pache L. et al. Metascape provides a biologist-oriented resource for the analysis of systems-level datasets. Nat Commun. 2019;10:1523
18. Mrug M, Zhou J, Yang C. et al. Genetic and informatic analyses implicate Kif12 as a candidate gene within the Mpkd2 locus that modulates renal cystic disease severity in the Cys1cpk mouse. PLoS One. 2015;10:e0135678
19. Hirokawa N, Noda Y. Intracellular transport and kinesin superfamily proteins, KIFs: structure, function, and dynamics. Physiol Rev. 2008;88:1089-1118
20. Lakshmikanth GS, Warrick HM, Spudich JA. A mitotic kinesin-like protein required for normal karyokinesis, myosin localization to the furrow, and cytokinesis in dictyostelium. Proc Natl Acad Sci USA. 2004;101:16519
21. Yang W, Tanaka Y, Bundo M. et al. Antioxidant signaling involving the microtubule motor KIF12 is an intracellular target of nutrition excess in beta cells. Dev Cell. 2014;31:202-214
22. Stalke A, Sgodda M, Cantz T. et al. KIF12 Variants and Disturbed Hepatocyte Polarity in Children with a Phenotypic Spectrum of Cholestatic Liver Disease. J Pediatr. 2022;240:284-291.e9
Author contact
Corresponding authors: Yu Xia and Yuxin Jiang. Email addresses: xiayupumchcom (Yu Xia), jiangyuxinxhcom (Yuxin Jiang); Tel.: +86 106 915 9311; Fax: +86 106 915 5402.

 Global reach, higher impact
Global reach, higher impact