3.2
Impact Factor
ISSN: 1837-9664
J Cancer 2024; 15(8):2329-2339. doi:10.7150/jca.93766 This issue Cite
Research Paper
c-Rel is a Novel Oncogene in Lung Squamous Cell Carcinoma Regulating Cell Proliferation and Migration
1. Department of histology and embryology, School of Medicine, Shenzhen Campus of Sun Yat-Sen University, Sun Yat-Sen University, Shenzhen, Guangdong, China.
2. Department of Pulmonary and Critical Care Medicine, Guangzhou First People's Hospital, School of Medicine, South China University of Technology, Guangzhou, Guangdong, China.
3. School of Ophthalmology and Optometry, Wenzhou Medical University, Wenzhou, Zhejiang, China.
Abstract
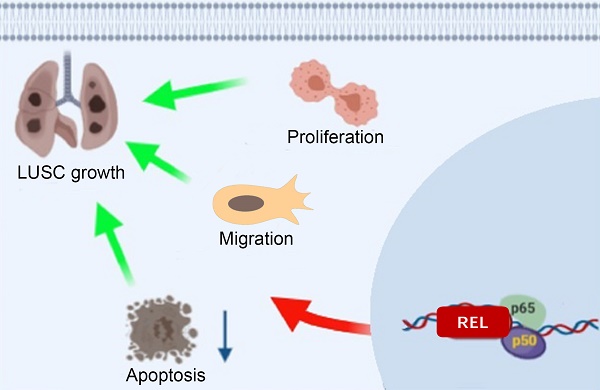
Lung squamous cell carcinoma (LUSC) accounts for approximately 25% to 30% of lung cancers, but largely no targeted therapy is available against it, calling for identification of new oncogenes in LUSC growth for new therapeutic targets. In this study, REL was identified through a screening for oncogenes that are highly amplified in human LUSC. Its expression was associated with poor prognosis in LUSC patients. Furthermore, knockdown of c-Rel in LUSC cell lines lead to significant decrease in cell proliferation and migration. Mechanistically, c-Rel knockdown suppressed NFκB pathway by blocking phosphorylation of IκB. Consistently, pharmaceutic inhibition of c-Rel also. In orthotopic xenograft lung cancer mouse model, c-Rel knockdown inhibited the tumor growth. Cancer cell proliferation and epithelial-mesenchymal-transition (EMT) of the tumors were impaired by c-Rel knockdown. Finally, it's confirmed in precision-cut tumor slices of LUSC that deletion of c-Rel inhibits the NFκB pathway and cancer cell growth. Accordingly, we hypothesize that c-Rel promotes the activation of the NFκB pathway by promoting the phosphorylation of IκB in LUSC. Our study reveals REL as a novel LUSC oncogene and provides new insights into the molecular regulation of LUSC, which will provide new therapeutic targets for the treatment of squamous lung cancer.
Keywords: c-Rel, REL, NFκB pathway, LUSC.

 Global reach, higher impact
Global reach, higher impact