3.2
Impact Factor
ISSN: 1837-9664
J Cancer 2024; 15(8):2442-2447. doi:10.7150/jca.91530 This issue Cite
Research Paper
A Retrospective Analysis of Azvudine in Patients with COVID-19 and Pre-existing Cancer
1. School of Pharmacy, Shanxi Medical University, Taiyuan 030001, China.
2. Department of Pharmacy, Shanxi Province Cancer Hospital/Shanxi Hospital Affiliated to Cancer Hospital, Chinese Academy of Medical Sciences/Cancer Hospital, Affiliated with Shanxi Medical University, Taiyuan, 030002, China.
Abstract
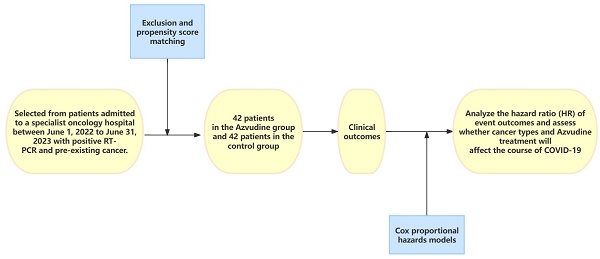
Objectives: Azvudine has been recommended as a potential treatment for the recently discovered Coronavirus disease (COVID-19) in 2019. However, the effectiveness of Azvudine in individuals who have both COVID-19 and pre-existing cancer remains uncertain. Consequently, we undertook a retrospective analysis to evaluate the clinical efficacy of Azvudine therapy in hospitalized patients with COVID-19 and pre-existing cancer.
Methods: This is a single-center retrospective analysis of patients diagnosed with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection, selected from patients admitted to a specialist oncology hospital between June 1, 2022 to June 31, 2023 with positive RT-PCR and pre-existing cancer. After exclusion and propensity score matching, patients in the test group treated with Azvudine and control patients treated with standard antiviral therapy were included. The primary outcome is the interval time from the first dose of Azvudine to the attainment of the first negative result for nucleic acid. Secondary outcomes included the rate of nucleic acid conversion, the duration of hospitalization, and the admission to the intensive care unit (ICU). Cox proportional hazards models were used to analyze the hazard ratio (HR) of event outcomes and to assess whether cancer types and Azvudine treatment will affect the course of COVID-19, specifically the time it takes for primary symptoms to alleviate.
Results: In this study, a total of 84 patients were included for analysis. Among them, 42 patients received Azvudine treatment after hospitalization, and the rest were treated with standard antiviral therapy. The results expressed that the time taken for the first negative nucleic acid test was significantly shorter in the Azvudine group compared to the control group [5 (IQR3-7) d vs 12 (IQR9-15) d], p<0.0001. This difference was statistically significant. Furthermore, a multivariate COX analysis indicated that Azvudine treatment could effectively reduce the time required for nucleic acid conversion in cancer patients (HR 1.994, 95% CI 1.064-3.736, p=0.031). And the type of cancer also had an impact on the course of COVID-19 in patients. (HR 3.442, 95%CI 1.214-9.756, p=0.020; HR 3.246, 95% CI 1.925-7.209, p=0.036).
Conclusion: Azvudine was correlated with a reduced duration for achieving nucleic acid conversion in individuals diagnosed with cancer. And different types of cancer have a certain impact on the course of COVID-19 for patients.
Keywords: COVID-19, Azvudine, cancer, RT-PCR, retrospective study

 Global reach, higher impact
Global reach, higher impact