Impact Factor ISSN: 1837-9664
J Cancer 2025; 16(7):2339-2352. doi:10.7150/jca.92956 This issue Cite
Research Paper
Bavachin suppresses proliferation of laryngopharyngeal cancer by regulating the STAT3 and MAPK signaling pathways
1. Department of Otorhinolaryngology Head and Neck Surgery, The First Affiliated Hospital of Anhui Medical University, Hefei, 230022, China.
2. Department of Biochemistry & Molecular Biology, Metabolic Disease Research Center, School of Basic Medicine, Anhui Medical University, Hefei, 230022, China.
3. Health Management Center, First Affiliated Hospital of the University of Science and Technology of China (Anhui Provincial Hospital), Hefei, 230022, China.
4. Department of Otorhinolaryngology Head and Neck Surgery, The People's Hospital of Xuancheng City, Xuancheng, 242000, China.
5. Department of Otorhinolaryngology Head and Neck Surgery, Fuyang Women and Children's Hospital, Fuyang, 236000, China.
6. Department of Otorhinolaryngology Head and Neck Surgery, The Fourth Affiliated Hospital of Anhui Medical University, Hefei, 230022, China.
7. Department of Otorhinolaryngology Head and Neck Surgery, The First Affiliated Hospital of Wannan Medical College, Wuhu, 241000, China.
*These authors contributed equally to this work.
Received 2023-12-5; Accepted 2024-8-21; Published 2025-3-31
Abstract
Purpose: The present study aimed to explore the underlying antitumor effects of bavachin on laryngopharyngeal cancer in-vitro and in-vivo.
Methods: Tu212 and FaDu cells were cultured in the incubator. Cells were treated with 0.1% DMSO (control group) and different concentrations of bavachin (experimental groups) for exploring the results of proliferation and apoptosis. We revealed the underlying mechanism of bavachin on laryngopharyngeal cancer through western blotting, qRT-PCR assay and immunofluorescence staining.
Results: Bavachin could suppress the proliferation and migration of laryngopharyngeal cancer cells in-vitro and in-vivo. Mechanistically, the results suggested that bavachin could downregulate the phosphorylation level of the signal transducer and activator of the transcription 3 (STAT3) and upregulate those of the mitogen-activated protein kinase (MAPK). Furthermore, bavachin also increased the expression level of Bax and suppressed those of Bcl-2, CDK4/6, and CyclinD1 in the laryngopharyngeal cancer cells. Additionally, the study also identified that bavachin promoted ferroptosis by decreasing the expression level of glutathione peroxidase 4 (GPX4) and increasing those of intracellular reactive oxygen species (ROS) and glutathione (GSH).
Conclusion: Taken together, these results demonstrated that bavachin could suppress the growth and migration of laryngopharyngeal cancer cells and induce apoptosis and cell cycle arrest of the laryngopharyngeal cancer cells by regulating the MAPK/STAT3 signaling pathway. This study demonstrated that bavachin exhibited a clinical therapeutic potential for laryngopharyngeal cancer.
Keywords: Laryngopharyngeal cancer, Bavachin, Cell cycle, Apoptosis, MAPK, STAT3
Introduction
The incidence rate of head and neck cancer has recently been increasing, especially that of head and neck squamous cell carcinoma (HNSCC), which is the sixth most common type of cancer worldwide [1]. Among the HNSCC, laryngopharyngeal carcinoma is the second most common type of cancer. It occurs in the laryngopharyngeal mucosal epithelium and accounts for approximately 90% of laryngopharyngeal carcinoma, which caused the death of 99,840 people in 2020 worldwide [2-4]. The symptoms of laryngopharyngeal cancer at the early stage are not obvious; therefore, approximately 60% of the patients are diagnosed in the late stages (III or IV), in which, the patients experience severe impairment of vocal, respiratory, and swallowing function [5]. Although the therapeutic strategies for laryngopharyngeal cancer have considerably progressed over the years, surgical resection remains the core treatment strategy [6]. However, surgery cannot guarantee the patient's survival and quality of life and its five-year survival rate has decreased from 66% to 63% due to unexplained pathogenesis and development [7]. Therefore, chemotherapy has been focused on due to its protective effects on laryngopharyngeal function [8].
Studies on and uses of herbal medicines have increased worldwide. Bavachin is a flavonoid extracted from the fruit of Psoralea corylifolia. Its chemical structure is shown in Figure 1A, its molecular formula is C20H20O4, and its molecular weight is 324.37. It has various functional activities such as anti-inflammatory [9], antiviral [10], anti-tumor [11], promoting osteogenesis and immune regulation[12,13], and also plays an important role in skin diseases [14].
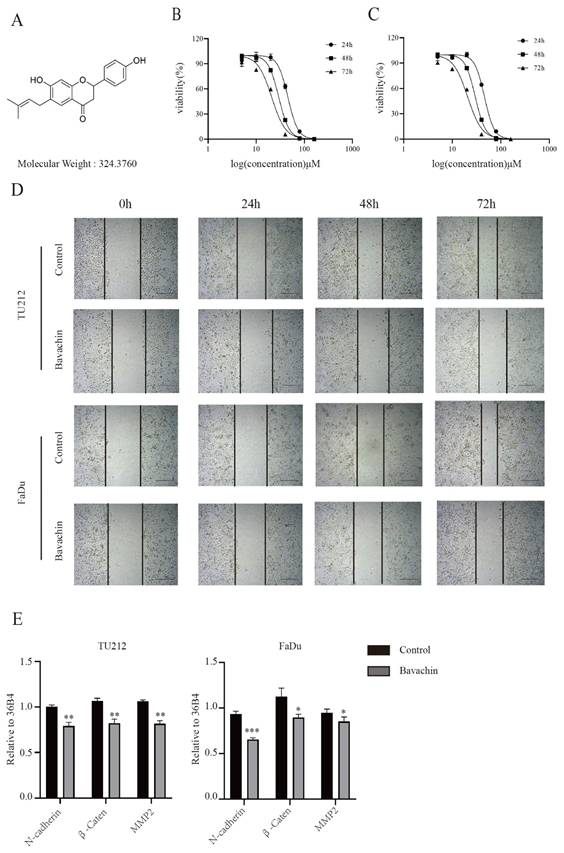
Bavachin suppressed the viability of laryngopharyngeal cancer cells and inhibited the migration of laryngopharyngeal cancer cells. (A) Chemical structure of bavachin. (B-C) MTT assays showed that bavachin (5, 10, 20, 40, 80, 160 μM/L for 24, 48 and 72h, respectively) decreased the viability of laryngopharyngeal cancer cells (n = 3). (D) Wound healing assays showed that bavachin (20 μM) significantly obstructed the migration ability of Tu212 and FaDu cells (n = 3). (E) qPCR analysis of the genes involved in migration of Tu212 and FaDu cells after bavachin treatment (n = 3). *P <0.05, **P <0.01, ***P <0.001.
Bavachin can improve asthma inflammation by inhibiting the expression of phospho-STAT6 and GATA-3 [15]. It could also inhibit the growth of various tumor cells by inducing apoptosis via interfering with mitochondria to regulate the endoplasmic reticulum stress pathway in HepG2 cells [16]. Moreover, bavachin could cause ferroptosis in the osteosarcoma cells via STAT3/P53/SLC7A11 axis [17]. However, the therapeutic effects of bavachin on human laryngopharyngeal cancer cells have not been studied.
The current study showed that bavachin could mediate the cellular proliferation and migration, cell cycle arrest, apoptosis, and ROS activity of Tu212 and FaDu cells by regulating the MAPK/STAT3 pathway in-vitro. Furthermore, its effects on tumor formation in nude mice were also explored by injecting the tumor cells in-vivo. Bavachin might have the potential for the clinical treatment of laryngopharyngeal tumors.
Materials and methods
Reagents
Human laryngopharyngeal squamous cell carcinoma (Tu212) cells were obtained from BeNa Culture Collection (Beijing, China). Human pharyngeal squamous cell carcinoma (FaDu) cell lines were purchased from World Cell Factory (Shanghai, China). Bavachin (CS-6113, purity >99%) was obtained from MedChemExpress (MCE, Shanghai, China). The following primary antibodies were used in this study: β-actin (Cat. No. 4970), JNK (Cat. No. 9252), p-JNK (Cat. No. 4668), Stat3 (Cat. No. 4904), p-Stat3 (Cat. No. 9131), E-cadherin (Cat. No. 3195), MAPK (Cat. No. 9212), and p-MAPK (Cat. No. 4631) were purchased from Cell Signaling Technology (MA USA); CDK6 (Cat. No. 66278-1-1g), CDK4 (Cat. No. 66950-1-1g), Cyclin D1 (Cat. No. 60186-1-1g), MFN1 (Cat. No. 13798-1-AP), and MFN2 (Cat. No. 12186-1-AP) were purchased from Proteintech (Wuhan Sanying, China); Ki67 (Cat. No. A2094) and FITC Goat Anti-Rabbit lgG (Cat. No. AS011) were purchased from ABclonal (Wuhan, China); and Cleaved-Caspase 3 (Cat. No. AF7022) and GPX4 (Cat. No. DF6701) were purchased from Affinity Biosciences (Giangsu, China).
Cell culture
The Tu212 and FaDu cell cultures were maintained in RPMI Medium 1640 basic (Gibco, China), containing 10% fetal bovine serum (FBS, Lonsera, Australia), penicillin (100 U/ml), and streptomycin (100 mg/ml, Beyotime, China). Both the cell cultures were maintained at 37ºC in a humidified atmosphere with a 5% CO2 concentration.
Cell proliferation assay
The cells were treated with 0.1% DMSO (control group) and different concentrations of bavachin (5, 10, 20, 40, 80, and 160 μmol/L) (experimental groups) for 24, 48 and 72 h. MTT solution (25 μL) was added to each well. Then, the supernatant was sucked out and 100-μL DMSO was added to each well. Finally, the optical density (OD value) of each well was measured at 490 nm using a microplate reader.
Colony formation assay
Approximately 1500 cells were cultured into each well in a 6-well plate. After the cell adhered to the walls, the control and experimental groups were treated with 0.1% DMSO and 5, 10, and 20 μmol/L bavachin, respectively. The DMSO and bavachin were replaced every four days. On the 10th day, the cells were fixed with 4% paraformaldehyde for 30 min. Then, the cells were stained with 0.1% crystalline violet staining for 30 min and observed using digital camera.
Wound healing assay
After the monolayer cells reached the confluence state in a 6-well plate, the cells were scratched. Then, the control and experimental groups were treated with 0.1% DMSO and 20 μmol/L bavachin, respectively. The states of cell migration were observed at different times (0, 24, 48, and 72 h) using an inverted microscope.
Apoptosis detection assay
The cells were evenly spread in the wells of a 6-well plate and incubated overnight. The cells were treated with DMSO (0.1%) and bavachin (20 μmol/L) as the control and experimental groups for 24 h, respectively. The cells were subsequently processed using Annexin V Apoptosis Detection kit (Bestbio, China, BB-4101) following the manufacturer's instructions. The cellular apoptosis results were measured using a flow cytometer. The data were analyzed using FlowJo V10 software.
Cell cycle assay
The cell cycle was detected using propidium iodide (PI, Bestbio, China, BB-4104). When the number of cells reached 4×105 per well in a 6-well plate, the cells were treated with 0.1% DMSO and 20 μmol/L bavachin as the control and experimental groups and incubated for 24 h. The cells were then collected and fixed with cold anhydrous ethanol overnight. The next day, the cells were incubated with 500-μL phosphate buffered saline (PBS), containing 20 μL Rnase A, at 37ºC for 30 min. Finally, the samples were treated with 400 μL PI and incubated in dark for 30 min to 1 h at 4ºC. The cells were transferred to the flow tube. The cell cycle results were analyzed using FlowJo V10 software.
ROS and GSH assay
The ROS levels were analyzed using DCFH-DA (Beyotime, China, S0033S). Approximately 5×105 FaDu and Tu212 cells per well were spread in a 6-well plate and cultured overnight. The cells were then treated with 0.1% DMSO and 20 μM bavachin as control and experimental groups for 24 h, respectively. The harvested cells were stained with 10 μmol/L DCFH-DA for 20 min at 37ºC, immediately washed 3 times with basal medium, and detected using a flow cytometer. The intracellular GSH levels were assessed using a GSH colorimetric assay kit (Solarbio, China, BC1175).
Western blotting
In order to analyze the expression levels of intracellular proteins, β-actin, JNK, p-JNK, Stat3, p-Stat3, p-MAPK, CDK6, CDK4, Cyclin D1, MFN1, MFN2, GPX4, cells were lysed using RIPA buffer (Beyotime, China, P0013B). The total proteins were separated using sodium dodecyl sulfate-polyacrylamide gels (8%-12%) electrophoresis based on the molecular weight of the target protein. The separated proteins were transferred to polyvinylidene fluoride (PVDF, 0.45 μm, Millipore) membranes, which were blocked with 5% skim milk for 1 h. Next, the membranes were incubated at 4ºC with the homologous primary antibodies for 10 to 12 h, followed by four times washing with TBST buffer. After incubating with the respective secondary antibodies for 1.5 h, the target proteins were observed via ECL using a chemiluminescence apparatus.
qRT-PCR assay
The FaDu and Tu212 cells were divided into control and experimental groups, which were treated with 0.1% DMSO and 20 μmol/L bavachin, respectively. After washing the cells twice with cold PBS, the intracellular RNA was extracted using TRIzol reagent (Ambion, USA) and reverse transcribed into cDNA using gradient thermal cycler (LongGene, China). The 20-μL PCR reaction mixture included cDNA as a template, qPCR Master Mix (TOLOBIO, China), diethyl pyrocarbonate (DEPC)-treated water (Sangon Biotech, China), and relevant primers. The PCR reaction was run on a Real-Time PCR detection system (Roche, China).
Tumor formation assay in nude mouse
Five-week-old BALB/c male nude mice were obtained from GemPharmatech LLC (Nanjing, China) and housed in a Specific pathogen Free (SPF) environment. All the animal treatments and experiments were performed as per the guidelines of the Animal Center of Anhui Medical University and approved by the Animal Ethical Committee of Anhui Medical. The FaDu cells (4×105) were resuspended in a basic medium and subcutaneously administered into the lower right flank of each mouse. The mice's weight and tumor size were measured every two. When the tumor volume reached approximately 100 mm3, the experimental and control group were administered 60 mg/kg of bavachin and equal volume of DMSO solvent every two days, respectively. After 3 weeks, the tumor tissues were excised from the nude mice and completely preserved for further analysis.
Immunofluorescence staining
The tumor tissues excised from the nude mice were fixed with 4% paraformaldehyde for two days, paraffinized, and cut into 5-μm-thick sections, which were then fixed on adhesive slides. After the slides were completely dried at 65ºC, they were orderly put into xylene, ethanol, and distilled water. The antigens were restored in ethylenediamine tetraacetic acid disodium salt (EDTA) in the microwave. After cooling the slides naturally to room temperature, the restored antigens were incubated with 5% normal goat serum for 2 h at 37ºC, followed by incubation with the primary antibodies for cleaved-Caspase3 and Ki67 at 4ºC overnight. The tissue sections were allowed to reach room temperature and then incubated with the corresponding secondary antibodies at 37ºC for 1.5 h. Then, the tissue sections were stained with DAPI at room temperature for 10 min. Finally, the stained sections were observed under a fluorescence microscope.
Histopathological analysis
For the histopathological analysis of the tumor tissues, they were immersed in 4% formalin for two days and paraffinized. The tumor tissues were cut into 5-μm-thick sections using a paraffin cutting machine and then stained with hematoxylin and eosin (H&E). Finally, the histopathology of tumor tissues was observed under a microscope.
Statistical analyses
All the data were analyzed with the paired t-test and one-way analysis of variance for multiple comparisons using GraphPad Prism to estimate the statistical significance. A P-value of <0.05 was considered statistically significant.
Results
Bavachin could inhibit the proliferation of laryngopharyngeal cancer cells in-vitro
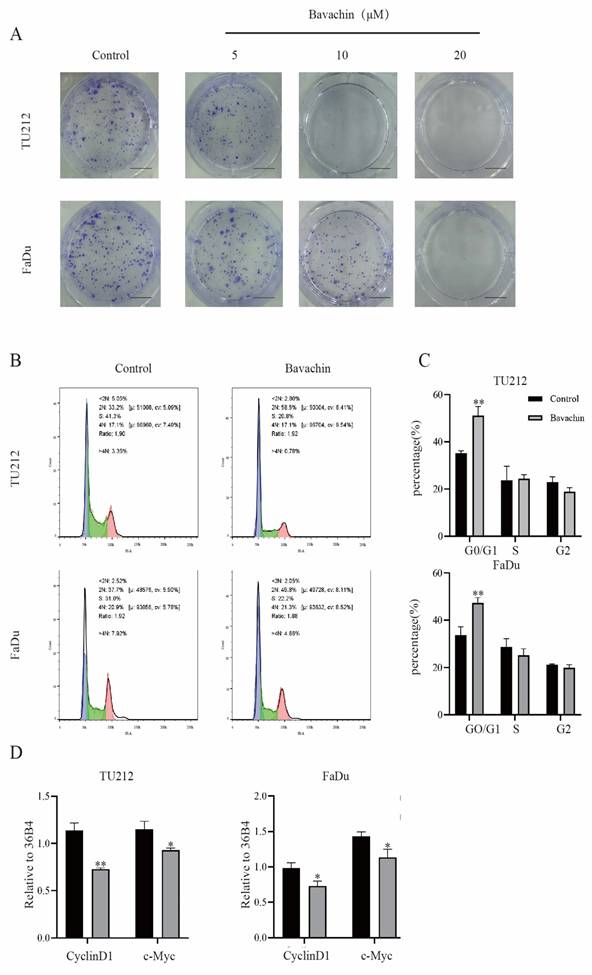
Previous studies indicated that bavachin could suppress the growth of a wide variety of tumors [14, 15]. In order to analyze the effects of bavachin on laryngopharyngeal cancer cells, the Tu212 and FaDu cells were treated with different concentrations of bavachin. Then, the viability and proliferation ability of the cells were analyzed using MTT and colony formation assays, respectively. The MTT assay showed that the viability of Tu212 and FaDu cells gradually reduced with the increase in the concentration of bavachin. By fitting the data, the IC50 values of bavachin for Tu212 and FaDu cells were identified to be 46.09 μM and 52.26 μΜ, respectively after 24 h (Figure 1. B-C). For the colony formation assay, different bavachin concentrations (5, 10, and 20 μM/L) were selected. The colony formation ability of Tu212 and FaDu cells gradually decreased with the increase in the concentration of bavachin (Figure 2. A). The results showed that bavachin could inhibit the proliferation of laryngopharyngeal cancer cells.
Bavachin inhibited cellular proliferation and caused cell cycle arrest of laryngopharyngeal cancer cells. (A) Effect of bavachin on colony formation (n = 3). (B-C) Laryngopharyngeal cancer cells were treated with bavachin (20 μM) for 24 h and analyzed using a flow cytometer. Bavachin induced G0/G1 cell cycle arrest as compared to the control group. (E) qPCR results of genes related to the cell cycle of Tu212 and FaDu cells after treatment with bavachin (20 μM/L) for 24 h (n = 3). *P <0.05, **P <0.01, ***P <0.001.
Bavachin could suppress the migration of laryngopharyngeal cancer cells in-vitro
A wound healing assay was performed to explore the effects of bavachin on the migration of laryngopharyngeal cancer cells. The result showed that bavachin treatment significantly inhibited the migration distance of laryngopharyngeal cancer cells as compared with the control group (Figure 1. D). Previous studies showed that the lower expression level of E-cadherin and higher expression levels of N-cadherin, β-catenin, and MMP2 were correlated with an increase in tumor cell migration and worse prognosis [18-20]. Interestingly, this study showed that the protein levels of E-cadherin increased significantly after treating the Tu212 and FaDu cells with bavachin (Figure 5. A-B). Additionally, q-RT-PCR analysis also showed that the bavachin treatment decreased the mRNA levels of MMP2 and N-cadherin in the Tu212 and FaDu cells as compared to the control group (Figure 1. E). These results suggested that bavachin could suppress the migration of Tu212 and FaDu cells in-vitro.
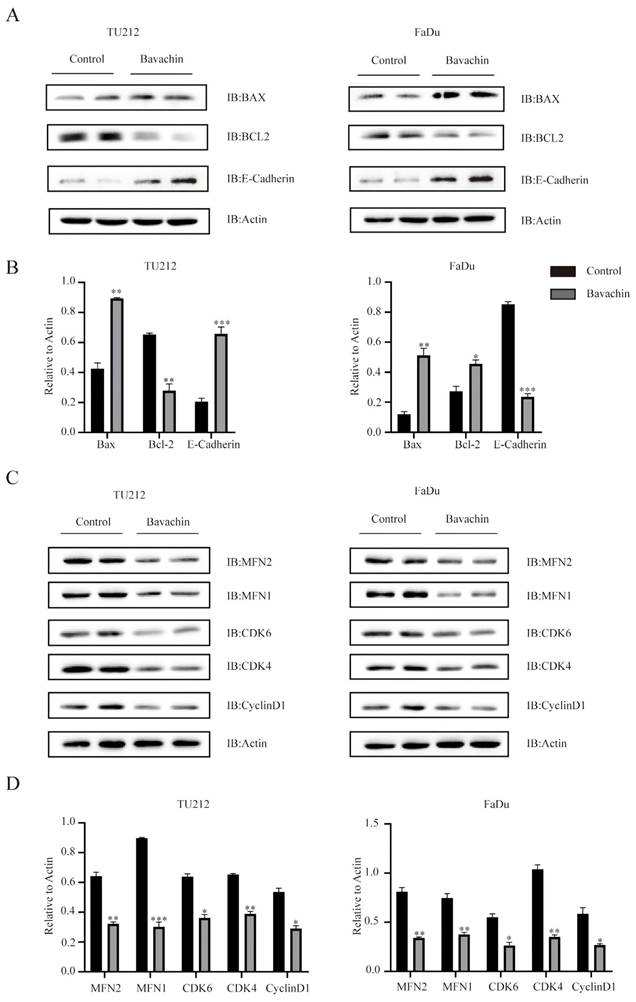
(A) Bavachin (20 μM/L) downregulated the protein expression level of Bcl-2 and upregulated those of Bax and E-cadherin in the Tu212 and FaDu cells. (B) Statistical results of the protein expression indicated in A. (C) Western blot results of protein related to the cell cycle of Tu212 and FaDu cells after treatment with bavachin (20 μM/L) for 24 h. (D) Statistical results of the protein expression indicated in C. *P <0.05, **P <0.01, ***P <0.001.
Bavachin could arrest laryngopharyngeal cancer cells in G0/G1 phase
The cell cycle assay results were analyzed using flow cytometry to determine the effects of bavachin on the progression of cell cycles. The result showed that the bavachin treatment arrested the Tu212 and FaDu cells in the G0/G1 phase as compared to the control group (Figure 2. B). The 20-μM/L bavachin treatment for 24 h significantly increased the proportion of Tu212 and FaDu cells in the G0/G1 stage as compared to the control group, and the proportion of cells in the S and G2/G0 stage was reduced (Figure 2. C). Among the stages of the cell cycle, G0/G1 phase is tightly regulated by cyclin D1, CDK4/6, and other related proteins [21]. In this study, Western blot analysis also showed that the bavachin treatment lowered the expression levels of cyclin D1 and CDK4/6 in both the Tu212 and FaDu cells as compared to those of the control group (Figure 5. C-D). Moreover, the qRT-PCR also demonstrated that bavachin treatment decreased the expression levels of Cyclin D1 and c-Myc (Figure 2. D). These results suggested that bavachin could arrest the Tu212 and FaDu cells in G0/G1 phase.
Bavachin could promote the apoptosis of laryngopharyngeal cancer cells in-vitro
The apoptosis detection assay results were analyzed using flow cytometry to explore the effects of bavachin on the apoptosis of Tu212 and FaDu cells. The results showed that bavachin significantly increased the early and late apoptotic rate of Tu212 and FaDu cells. In the Tu212 cells, the experimental (20 μM/L bavachin) and control groups showed cellular apoptosis of 25.5% and 11.43%, respectively, while those in the FaDu cells were 20.83% and 9.81%, respectively (Figure 3. A-B). The mechanism of the bavachin-induced apoptosis was further elucidated by analyzing the alterations in apoptosis-related proteins using Western blotting. These results showed that the expression level of BAX was higher in the experimental group as compared to the control group (Figure 5. A-B). Meanwhile, the expression level of Bcl-2 decreased in the experimental group in both the Tu212 and FaDu cells (Figure 5. A-B). These results demonstrated that bavachin could promote the apoptosis of the laryngopharyngeal cancer cells.
Bavachin induced the apoptosis of laryngopharyngeal cancer cells. (A-B) Bavachin (20 μM/L) significantly induced the apoptosis of laryngopharyngeal cancer cells (25.5% in the bavachin treatment group vs. 11.43% in the control group in the Tu212 cells and 20.83% in the bavachin treatment group vs. 9.81% in the control group in the FaDu cells, respectively) after 24 h (n = 3). (C-D) Bavachin (20 μM/L) promoted the accumulation of ROS in Tu212 and FaDu cells after 24 h (n = 3). (E) Bavachin (20 μM/L) treatment after 24 h decreased the intracellular level of GSH in the Tu212 and FaDu cells (n = 3). (F) Bavachin (20 μM/L) treatment remarkably suppressed the expression levels of GPX4 in the Tu212 and FaDu cells after 24 h (n = 3). (G) Statistical results of the protein expression indicated in F. *P <0.05, **P <0.01.
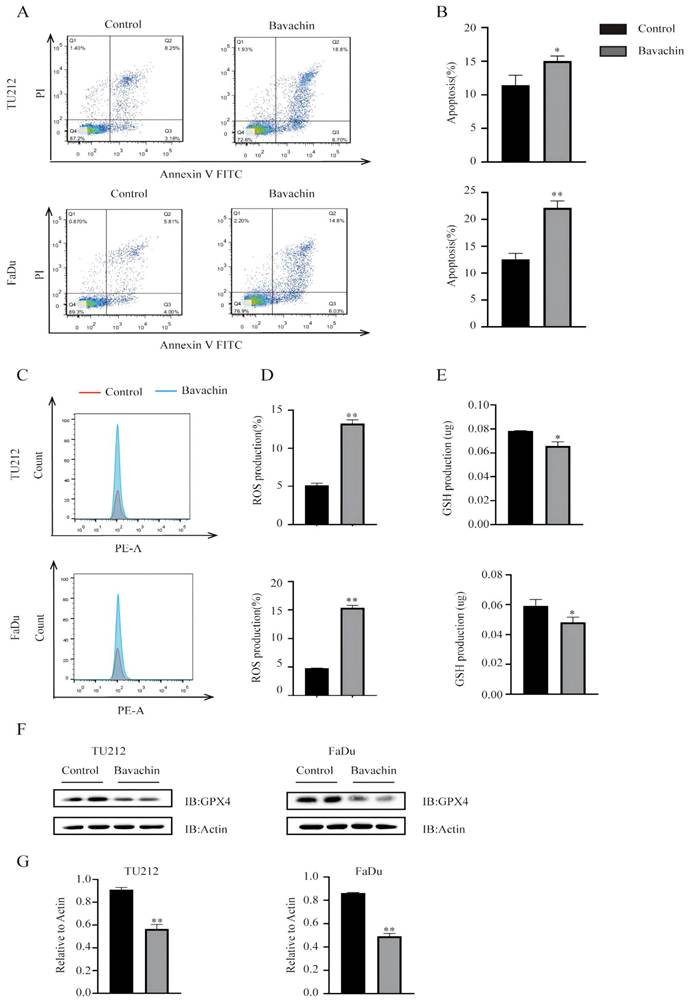
Bavachin could promote ferroptosis by accumulating ROS and consuming GSH regulated by MAPK and STAT3
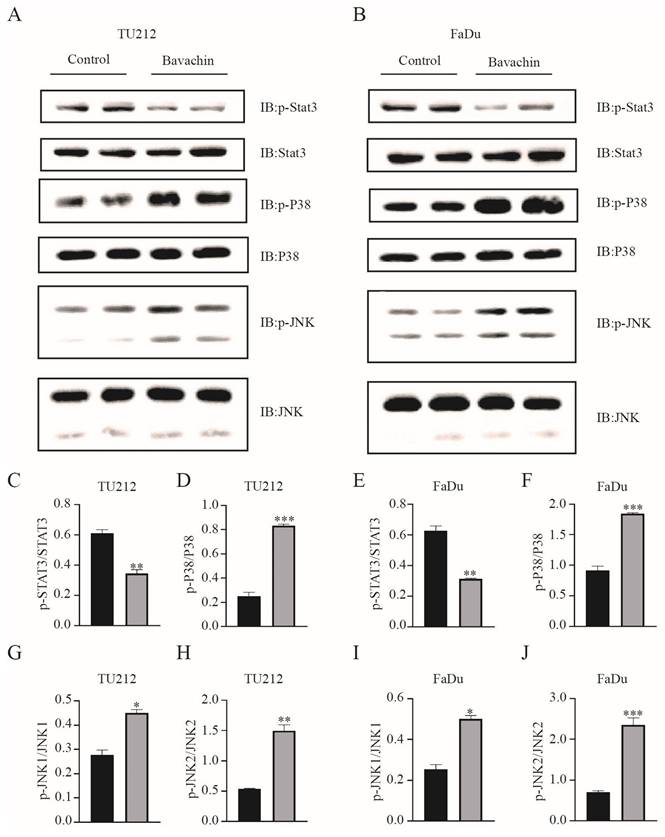
A previous study demonstrated that bavachin could induce ferroptosis in osteosarcoma cells by regulating the STAT3/P53/SLC7A11 signaling pathway [15]. Therefore, this study explored the effects of bavachin on inducing ferroptosis in the Tu212 and FaDu cells. The results showed that the bavachin treatment increased the level of intracellular ROS (13.6% and 15.7%) as compared to the control group (4.88% and 4.81%) in the Tu212 and FaDu cells, respectively (Figure 3. C-D). The bavachin treatment also increased the consumption of intracellular GSH as compared to the control group (Figure 3. E). The expression levels of the key pathway components of Tu212 and FaDu cells were analyzed using Western blotting. The results showed that the bavachin treatment suppressed the phosphorylation level of STAT3 and promoted that of P38 and JNK (Figure 4. A-B). Meanwhile, the bavachin treatment downregulated the expression of GPX4 in the Tu212 and FaDu cells (Figure 3. F). These results demonstrated that bavachin could promote ferroptosis by increasing the level of intracellular ROS and inhibiting the expression levels of GPX4 and GSH.
Bavachin (20 μM/L) treatment remarkably suppressed the expression levels of p-STAT3 and promoted the phosphorylation levels of P38 and JNK in the Tu212 and FaDu cells after 24 h (n = 3). (C-J) Statistical results of the protein expression indicated in A and B. *P <0.05, **P <0.01, ***P <0.001.
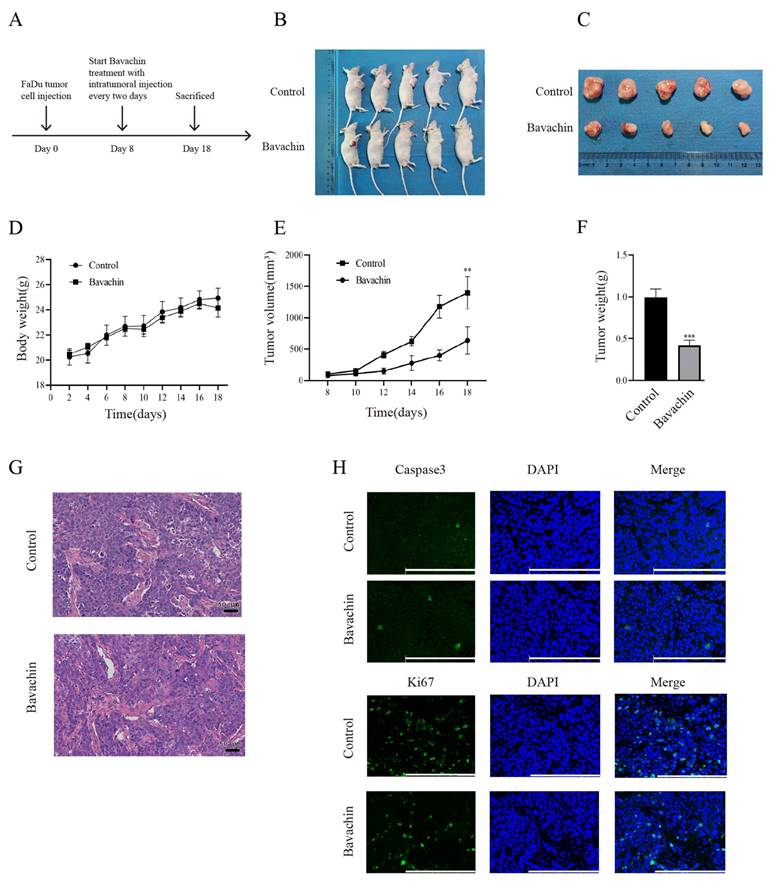
Bavachin could inhibit the growth of laryngopharyngeal cancer cells in-vivo
The FaDu cells (4×105) were subcutaneously injected into the male BALB/c nude mice to further explore the suppressing effects of bavachin on the growth of laryngopharyngeal carcinoma cells in vivo (Figure 6. B-C). When tumor volume reached approximately 100 mm3, bavachin was injected at the tumor site after every two days (Figure 6. A). The results showed that the bavachin treatment markedly suppressed the tumor size and tumor weight as compared to the control group (Figure 6. E-F). Moreover, immunofluorescence analysis of the tumor tissues showed a significant decrease in the number of Ki67-positive cells in the experimental group as compared to the control group. Meanwhile, the proportion of cleaved-caspase3-positive cells was higher in the tumor tissues in the experimental group as compared to the control group (Figure 6. H). Collectively, these results indicated that bavachin could suppress the growth of human laryngopharyngeal cancer cells in-vivo by regulating the expression levels of Ki67 and cleaved-caspase3.
Bavachin inhibited tumor growth in-vivo. (A) Schematic diagram of the in-vivo experiment. (B-C) State of tumor growth in the human laryngopharyngeal cancer cells. The FaDu (4×106) cells were injected into nude mice (n = 6). (D) Changes in mice body weight with time. (E) Changes in tumor volume with time. (F) Comparison of tumor weight between the experimental and control group. (G-H) Immunofluorescence staining of tumor tissues was performed for the detection of c-Caspase3 and Ki67. (Scale bar: 20 μm). *P <0.05, **P <0.01, ***P <0.001.
Discussion
Laryngopharyngeal cancer is a common type of head and neck cancer in otorhinolaryngology. Although studies have focused on preventing laryngopharyngeal cancer, its incidence has been continuously increasing annually. Numerous therapeutic strategies have been designed for the treatment of laryngopharyngeal cancer, among which, surgical resection is the core treatment strategy. Nevertheless, the patients have a poor prognosis and impaired laryngopharyngeal function after surgery. Recently, increasing evidence suggested that traditional Chinese medicine and natural products might have significant function potential [22]. Bavachin is a flavonoid extracted from the fruit of Psoralea corylifolia [14]. Previous studies showed that bavachin exhibited positive antitumor effects through different mechanisms. For instance, bavachin could promote ROS generation and induce ferroptosis in the osteosarcoma cells by regulating the STAT3/P53/SLC7A11 signaling pathway [17]. Additionally, bavachin could also suppress the growth of human placental choriocarcinoma cells and human hepatocellular carcinoma cells by causing mitochondrial dysfunction and endoplasmic reticulum stress [16, 23]. Other studies reported that bavachin could induce the apoptosis of multiple myeloma cells by downregulating the expression levels of nuclear transcription factor-kappa B (NF-κB) and STAT3 and upregulating that of cleaved-caspase3 [13]. This study indicated that bavachin suppressed the growth of laryngopharyngeal cancer.
This study has demonstrated that bavachin could inhibit the proliferation of laryngopharyngeal cancer cells in-vitro. Moreover, bavachin could also significantly suppress the migration ability of laryngopharyngeal cells. Previous studies suggested that the loss of E-cadherin, an epithelial adhesion molecule, resulted in metastasis by breaking the intercellular contacts [24]. Studies showed that the levels of N-cadherin, β-catenin, and MMP2 were markedly increased in the late tumor stages [18, 20]. This study confirmed that the bavachin treatment could inhibit the expression level of N-cadherin, β-catenin, and MMP2 and increase that of E-cadherin in the laryngopharyngeal cancer cells.
Apoptosis is related to the mitochondrial and death receptor pathways [25]. The mitochondrial apoptotic pathway is controlled by the Bcl-2 protein family, including pro-apoptotic proteins (Bax and Bad) and anti-apoptotic proteins (Bcl-2 and Bcl-xl) [26, 27]. In the process of apoptosis, the fusion/fission proteins regulate the breakdown of mitochondria into fragments, causing cell death [28]. Mitochondrial fusion is mainly mediated by three crucial proteins, including mitofusin-1 (MFN1), mitofusin-2 (MFN2), and optic atrophy 1 (OPA1) [29]. This study showed that the bavachin treatment downregulated the protein expression levels of Bcl-2, MFN1, and MFN2 and upregulated that of Bax in the laryngopharyngeal cancer cells. These results indicated that bavachin could induce apoptosis by regulating the mitochondrial pathway.
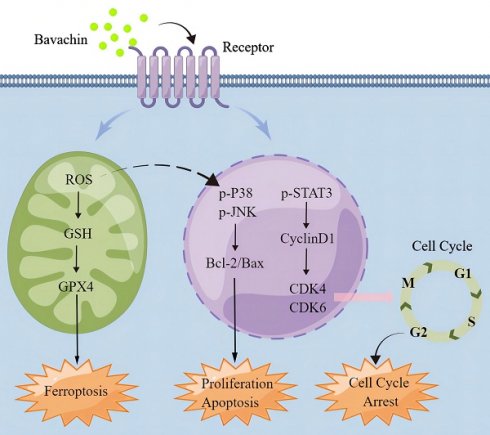
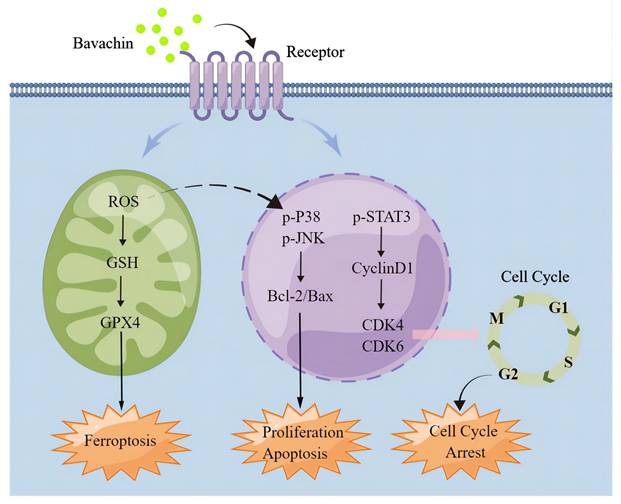
Schematic representation of the signaling pathways in laryngopharyngeal cancer cells affected by bavachin. This image was supported by Figdraw.
The cell cycle arrest is correlated with apoptosis and regulation of cellular proliferation [30, 31]. Numerous regulatory proteins are involved in the complex process of the cell cycle, among which, cyclin-dependent kinases (CDKs), including the cyclin proteins, are the main proteins [32]. CDKs are the most vital regulatory proteins involved in the cell cycle, and the specific cyclins play important roles in the different phases of the cell cycle [33]. In the current study, bavachin induced the arrest of Tu212 and FaDu cells at the G0/G1 phase, and significantly reduced the protein expression levels of CDK4/6, cyclin D1, and c-Myc. Therefore, the results suggested that bavachin could suppress the proliferation of Tu212 and FaDu cells by inhibiting the growth of cells entering the S stage of the cell cycle.
The MAPK family plays a crucial role in regulating the growth, differentiation, apoptosis, and stress response of the cells [34]. The diverse stimuli can activate different MAPK pathways and regulate various biological processes; for instance, p38 and JNK are associated with oxidative stress response and cellular apoptosis [35]. p38 and JNK regulate multiple transcription factors, leading to the upregulation of pro-apoptotic proteins and downregulation of anti-apoptotic proteins [36]. A previous study showed that exogenous NO could induce the apoptosis of hepatocellular carcinoma cells by positive regulation of p38/JNK signaling pathways [37]. Moreover, the STAT3 signaling pathway is also a vital intracellular signaling pathway, which mediates cellular growth and cycle [38]. In general, the expression level of STAT3 is higher in tumor cells as compared to the normal cells and is beneficial for the proliferation of tumor cells and anti-apoptosis [39]. This study showed that bavachin treatment downregulated the phosphorylation level of STAT3 and upregulated that of p38 and JNK in the Tu212 and FaDu cells. These results demonstrated that bavachin could suppress the proliferation of tumor cells and cause cell cycle arrest and apoptosis through the MAPK and STAT3 signaling pathways.
Previous studies reported that tumor cells had higher levels of ROS as compared to normal cells [40]. Nevertheless, some chemotherapeutic drugs can cause oxidative stress injury and apoptosis of tumor cells by increasing the oxidative stress level in tumor cells [41]. Recently, ferroptosis has been defined as the reaction of intracellular iron with H2O2, producing ROS and causing lipid peroxidation (LP) [42]. In particular, the system xc- -GSH-GPX4-dependent antioxidant defense induces the accumulation of LP [43]. A previous study reported that bavachin could induce ferroptosis in the osteosarcoma cells by inhibiting the expression level of STAT3 [17] and inhibit human liver cancer cells by inducing apoptosis and concomitant accumulation of reactive oxygen species [44]. Consistent, this study also showed that the bavachin-induced ferroptosis was correlated with the accumulation of ROS and consumption of GSH in the Tu212 and FaDu cells.
Altogether, bavachin could suppress the proliferation and migration of laryngopharyngeal cancer cells, resulting in the apoptosis and cell cycle arrest of the laryngopharyngeal cancer cells by regulating the MAPK/STAT3 signaling pathway. These results suggested a direct effect of bavachin on laryngopharyngeal cancer cells, which might be reversed by an antioxidant, thereby requiring further study. In the future, the invasion ability of bavachin on the laryngopharyngeal cancer cells should be further explored by constructing a lung metastasis model.
Conclusions
Taken together, our findings suggested that bavachin could significantly inhibit the growth, migration, cell-cycle arrest, and induced apoptosis of laryngopharyngeal cells in-vivo and in-vitro. Furthermore, we also revealed that bavachin exhibited anti-tumor effects on laryngopharyngeal cells by regulating the MAPK and STAT3 signaling pathways. This study provided a basis for using bavachin as an anti-tumor agent for laryngopharyngeal cancer in the future.
Abbreviations
CDKs: cyclin-dependent kinases; DEPC: diethyl pyrocarbonate; EDTA: ethylenediamine tetraacetic acid disodium salt; GPX4: glutathione peroxidase 4; GSH: glutathione; HNSCC: head and neck squamous cell carcinoma; LP: lipid peroxidation; MAPK: mitogen-activated protein kinase; PBS: phosphate buffered saline; PI: propidium iodide; PVDF: polyvinylidene fluoride; ROS: reactive oxygen species; SPF: Specific pathogen Free; STAT3: signal transducer and activator of the transcription 3.
Acknowledgements
Funding
This work was supported by the Provincial Natural Science Foundation of Anhui, China (Nos. 2108085MH282).
Author contributions
H.B.Z., M.Z. and C.B.G. designed and supervised the study. Z.M.D. and Y.Z. performed the cell related experiment. H.X. and D.W. performed the animal line experiment. W.J.J., X.N.Y. and D.Y.S performed the analysis of data. X.W. and H.T.H. performed the graphing mechanism. All authors checked the results and wrote the manuscript. All authors have read and approved the final manuscript. All data were generated in-house, and no paper mill was used. All authors agree to be accountable for all aspects of work ensuring integrity and accuracy.
Data sharing statement
The data supporting the current study are available from the corresponding author upon reasonable request.
Ethics statement
No human subjects were involved in the study. All the animal treatments and experiments were performed as per the guidelines of the Animal Center of Anhui Medical University and approved by the Animal Ethical Committee of Anhui Medical.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Fitzmaurice C, Allen C, Barber RM, Barregard L, Bhutta ZA, Brenner H. et al. Global, Regional, and National Cancer Incidence, Mortality, Years of Life Lost, Years Lived With Disability, and Disability-Adjusted Life-years for 32 Cancer Groups, 1990 to 2015. JAMA Oncology. 2017;3:524-48
2. Genden EM, Ferlito A, Silver CE, Jacobson AS, Werner JA, Suárez C. et al. Evolution of the management of laryngeal cancer. Oral Oncology. 2007;43:431-9
3. Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A. et al. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA: a cancer journal for clinicians. 2021;71:209-49
4. Wu Y, Zhang Y, Zheng X, Dai F, Lu Y, Dai L. et al. Circular RNA circCORO1C promotes laryngeal squamous cell carcinoma progression by modulating the let-7c-5p/PBX3 axis. Molecular Cancer. 2023;22:51
5. Morgan RL, Eguchi MM, McDermott J, Mueller AC, Amini A, Goddard JA. et al. Comparative effectiveness of posttreatment imaging modalities for Medicare patients with advanced head and neck cancer. Cancer. 2021;127:535-43
6. Song L, Zhang S, Yu S, Ma F, Wang B, Zhang C. et al. Cellular heterogeneity landscape in laryngeal squamous cell carcinoma. International Journal of Cancer. 2020;147:2879-90
7. Ohi M, Toiyama Y, Yasuda H, Ichikawa T, Imaoka H, Okugawa Y. et al. Preoperative computed tomography predicts the risk of recurrent laryngeal nerve paralysis in patients with esophageal cancer undergoing thoracoscopic esophagectomy in the prone position. Esophagus. 2021;18:228-38
8. Verma A, Schwartz N, Cohen DJ, Boyan BD, Schwartz Z. Estrogen signaling and estrogen receptors as prognostic indicators in laryngeal cancer. Steroids. 2019;152:108498
9. Hung YL, Wang SC, Suzuki K, Fang SH, Chen CS, Cheng WC. et al. Bavachin attenuates LPS-induced inflammatory response and inhibits the activation of NLRP3 inflammasome in macrophages. Phytomedicine. 2019;59:152785
10. Cheng C, Yu-Feng S, Yang H, Lei L, Wei-Chao C, Gao-Xue W. et al. Highly efficient inhibition of spring viraemia of carp virus replication in vitro mediated by bavachin, a major constituent of psoralea corlifonia Lynn. Virus Res. 2018;255:24-35
11. Takeda T, Tsubaki M, Tomonari Y, Kawashima K, Itoh T, Imano M. et al. Bavachin induces the apoptosis of multiple myeloma cell lines by inhibiting the activation of nuclear factor kappa B and signal transducer and activator of transcription 3. Biomedicine & Pharmacotherapy. 2018;100:486-94
12. He HQ, Law BYK, Zhang N, Qiu CL, Qu YQ, Wu AG. et al. Bavachin Protects Human Aortic Smooth Muscle Cells Against beta-Glycerophosphate-Mediated Vascular Calcification and Apoptosis via Activation of mTOR-Dependent Autophagy and Suppression of beta-Catenin Signaling. Front Pharmacol. 2019;10:1427
13. Weng ZB, Gao QQ, Wang F, Zhao GH, Yin FZ, Cai BC. et al. Positive skeletal effect of two ingredients of Psoralea corylifolia L. on estrogen deficiency-induced osteoporosis and the possible mechanisms of action. Mol Cell Endocrinol. 2015;417:103-13
14. Chopra B, Dhingra A K, Dhar K L. Psoralea corylifolia L. (Buguchi) - Folklore to modern evidence: Review. Fitoterapia. 2013;90:44-56
15. Liang Z, Luo Z, Chen J, Li B, Li L, Shen C. Bavachin inhibits IL-4 expression by downregulating STAT6 phosphorylation and GATA-3 expression and ameliorates asthma inflammation in an animal model. Immunobiology. 2022;227:152182
16. Yang Y, Tang X, Hao F, Ma Z, Wang Y, Wang L. et al. Bavachin Induces Apoptosis through Mitochondrial Regulated ER Stress Pathway in HepG2 Cells. Biological and Pharmaceutical Bulletin. 2018;41:198-207
17. Luo Y, Gao X, Zou L, Lei M, Feng J, Hu Z. Bavachin Induces Ferroptosis through the STAT3/P53/SLC7A11 Axis in Osteosarcoma Cells. Oxidative Medicine and Cellular Longevity. 2021;2021:1783485
18. Zhu GJ, Song PP, Zhou H, Shen XH, Wang JG, Ma XF. et al. Role of epithelial-mesenchymal transition markers E-cadherin, N-cadherin, beta-catenin and ZEB2 in laryngeal squamous cell carcinoma. Oncol Lett. 2018;15:3472-81
19. Re M, Gioacchini FM, Scarpa A, Cassandro C, Tulli M, Cassandro E. The prognostic significance of E-cadherin expression in laryngeal squamous-cell carcinoma: a systematic review. Acta Otorhinolaryngologica Italica. 2018;38:504-10
20. Zhu Y, Yan L, Zhu W, Song X, Yang G, Wang S. et al. MMP2/3 promote the growth and migration of laryngeal squamous cell carcinoma via PI3K/Akt-NF-κB-mediated epithelial-mesenchymal transformation. Journal of Cellular Physiology. 2019;234:15847-55
21. Zamir-Nasta T, Razi M, Shapour H, Malekinejad H. Roles of p21, p53, cyclin D1, CDK-4, estrogen receptor α in aflatoxin B1-induced cytotoxicity in testicular tissue of mice. Environmental Toxicology. 2018;33:385-95
22. Liu Y, Yang S, Wang K, Lu J, Bao X, Wang R. et al. Cellular senescence and cancer: Focusing on traditional Chinese medicine and natural products. Cell Proliferation. 2020;53:e12894
23. Lee J, Lim W, Song G. Bavachin suppresses human placental choriocarcinoma cells by targeting electron transport chain complexes and mitochondrial dysfunction. Free Radical Biology and Medicine. 2020;156:26-35
24. Onder TT, Gupta PB, Mani SA, Yang J, Lander ES, Weinberg RA. Loss of E-Cadherin Promotes Metastasis via Multiple Downstream Transcriptional Pathways. Cancer Research. 2008;68:3645-54
25. Zhou J, Xu XZ, Hu YR, Hu AR, Zhu CL, Gao GS. Cryptotanshinone Induces Inhibition of Breast Tumor Growth by Cytotoxic CD4+ T Cells through the JAK2/STAT4/ Perforin Pathway. Asian Pacific Journal of Cancer Prevention. 2014;15:2439-45
26. Antonsson B. Mitochondria and the Bcl-2 family proteins in apoptosis signaling pathways. Molecular and cellular biochemistry. 2004;256-257:141-55
27. Song X, Wang B, Lin S, Jing L, Mao C, Xu P. et al. Astaxanthin inhibits apoptosis in alveolar epithelial cells typeIIin vivo and in vitro through theROS -dependent mitochondrial signalling pathway. Journal of Cellular and Molecular Medicine. 2014;18:2198-212
28. Brooks C, Cho SG, Wang CY, Yang T, Dong Z. Fragmented mitochondria are sensitized to Bax insertion and activation during apoptosis. American Journal of Physiology-Cell Physiology. 2011;300:C447-C455
29. van der Bliek A M, Shen Q, Kawajiri S. Mechanisms of mitochondrial fission and fusion. Cold Spring Harb Perspect Biol. 2013;5:a011072
30. Romanov V, Whyard TC, Waltzer WC, Grollman AP, Rosenquist T. Aristolochic acid-induced apoptosis and G2 cell cycle arrest depends on ROS generation and MAP kinases activation. Archives of Toxicology. 2015;89:47-56
31. He D, Guo X, Zhang E, Zi F, Chen J, Chen Q. et al. Quercetin induces cell apoptosis of myeloma and displays a synergistic effect with dexamethasone in vitro and in vivo xenograft models. Oncotarget. 2016;7:45489-99
32. Bendris N, Lemmers B, Blanchard J M. Cell cycle, cytoskeleton dynamics and beyond: the many functions of cyclins and CDK inhibitors. Cell Cycle. 2015;14:1786-98
33. Lim S, Kaldis P. Cdks, cyclins and CKIs: roles beyond cell cycle regulation. Development. 2013;140:3079-93
34. Plotnikov A, Zehorai E, Procaccia S, Seger R. The MAPK cascades: Signaling components, nuclear roles and mechanisms of nuclear translocation. Biochimica et Biophysica Acta (BBA) - Molecular Cell Research. 2011;1813:1619-33
35. Zheng HY, Shen FJ, Tong YQ, Li Y. PP2A Inhibits Cervical Cancer Cell Migration by Dephosphorylation of p-JNK, p-p38 and the p-ERK/MAPK Signaling Pathway. Curr Med Sci. 2018;38:115-23
36. Yue J, López J M. Understanding MAPK Signaling Pathways in Apoptosis. International Journal of Molecular Sciences. 2020;21:2346
37. Liu L, Xing Y, Cao M, Xu J, Chen J. Exogenous NO induces apoptosis of hepatocellular carcinoma cells via positive p38/JNK signaling pathway and negative ERK signaling pathways. Molecular and Cellular Biochemistry. 2021;476:1651-61
38. Lai PS, Rosa DA, Magdy Ali A, Gómez-Biagi RF, Ball DP, Shouksmith AE. et al. A STAT inhibitor patent review: progress since 2011. Expert Opinion on Therapeutic Patents. 2015;25:1397-421
39. Fathi N, Rashidi G, Khodadadi A, Shahi S, Sharifi S. STAT3 and apoptosis challenges in cancer. Int J Biol Macromol. 2018;117:993-1001
40. Panieri E, Santoro M M. ROS homeostasis and metabolism: a dangerous liason in cancer cells. Cell Death & Disease. 2016;7:e2253
41. Alexandre J, Hu Y, Lu W, Pelicano H, Huang P. Novel action of paclitaxel against cancer cells: bystander effect mediated by reactive oxygen species. Cancer Res. 2007;67:3512-7
42. Chen X, Yu C, Kang R, Tang D. Iron Metabolism in Ferroptosis. Frontiers in Cell and Developmental Biology. 2020;8:590226
43. Chen X, Li J, Kang R, Klionsky DJ, Tang D. Ferroptosis: machinery and regulation. Autophagy. 2021;17:2054-81
44. Yang Y, Tang X, Hao F, Ma Z, Wang Y, Wang L. et al. Bavachin Induces Apoptosis through Mitochondrial Regulated ER Stress Pathway in HepG2 Cells. Biol Pharm Bull. 2018;41:198-207
Author contact
Corresponding authors: Mei Zhang, Health Management Center, First Affiliated Hospital of the University of Science and Technology of China (Anhui Provincial Hospital), Lujiang Road 17, Hefei, 230011, China; E-mail address: syzm110011com; Phone: 86-13721063819; Fax: 0551-2283409. Huabing Zhang, Ph.D. Address: Department of Biochemistry & Molecular Biology, School of Basic Medicine, Anhui Medical University, Meishan Road 81, Hefei 230032, China; E-mail address: slzhang1977com and huabingzhangedu.cn (H. Zhang); Phone: 86-551-65161131; Fax: 86-551-65142940. Chaobing Gao, MD, Department of Otorhinolaryngology Head and Neck Surgery, First Affiliated Hospital of Anhui Medical University, Jixi Road 218, Hefei, Anhui 230022, China; E-mail address: gcb110011com; Phone: 86-13856951551; Fax: 0551-63633742.

 Global reach, higher impact
Global reach, higher impact