Impact Factor ISSN: 1837-9664
J Cancer 2015; 6(6):519-524. doi:10.7150/jca.11404 This issue Cite
Research Paper
High EGFR_1 Inside-Out Activated Inflammation-Induced Motility through SLC2A1-CCNB2-HMMR-KIF11-NUSAP1-PRC1-UBE2C
1. Biomedical Center, School of Electronic Engineering, Beijing University of Posts and Telecommunications, Beijing, 100876, China
2. Lab of Computational Linguistics, School of Humanities and Social Sciences, Tsinghua University, Beijing, 100084, China
3. College of information, North China University of Technology, Beijing, 100043, China
* Equal contribution
Received 2014-12-20; Accepted 2015-2-20; Published 2015-4-5
Abstract
48 different Pearson mutual-positive-correlation epidermal growth factor receptor (EGFR_1)-activatory molecular feedback, up- and down-stream network was constructed from 171 overlapping of 366 GRNInfer and 223 Pearson under EGFR_1 CC ≥0.25 in high lung adenocarcinoma compared with low human normal adjacent tissues. Our identified EGFR_1 inside-out upstream activated molecular network showed SLC2A1 (solute carrier family 2 (facilitated glucose transporter) member 1), CCNB2 (cyclin B2), HMMR (hyaluronan-mediated motility receptor (RHAMM)), KIF11 (kinesin family member 11), NUSAP1 (nucleolar and spindle associated protein 1), PRC1 (protein regulator of cytokinesis 1), UBE2C (ubiquitin-conjugating enzyme E2C) in high lung adenocarcinoma. EGFR_1 inside-out upstream activated terms network includes intracellular, membrane fraction, cytoplasm, plasma membrane, integral to membrane, basolateral plasma membrane, transmembrane transport, nucleus, cytosol, cell surface; T cell homeostasis, inflammation; microtubule cytoskeleton, embryonic development (sensu Mammalia), cell cycle, mitosis, thymus development, cell division, regulation of cell cycle, Contributed--cellular process--Hs cell cycle KEGG, cytokinesis, M phase, M phase of mitotic cell cycle, estrogen-responsive protein Efp controls cell cycle and breast tumors growth, cell motility, locomotion, locomotory behavior, neoplasm metastasis, spindle pole, spindle microtubule, microtubule motor activity, microtubule-based movement, mitotic spindle organization and biogenesis, mitotic centrosome separation, spindle pole body organization and biogenesis, microtubule-based process, microtubule, cytokinesis after mitosis, mitotic chromosome condensation, establishment of mitotic spindle localization, positive regulation of mitosis, mitotic spindle elongation, spindle organization and biogenesis, positive regulation of exit from mitosis, regulation of cell proliferation, positive regulation of cell proliferation based on integrative GO, KEGG, GenMAPP, BioCarta and disease databases in high lung adenocarcinoma. Therefore, we propose high EGFR_1 inside-out activated inflammation-induced motility through SLC2A1-CCNB2-HMMR-KIF11-NUSAP1-PRC1-UBE2C in lung adenocarcinoma.
Keywords: EGFR_1 activated network, inside-out, motility, inflammation, SLC2A1-CCNB2-HMMR-KIF11-NUSAP1-PRC1-UBE2C
Introduction
EGFR_1 has EGFR activity based on GO database. EGFR positive relationship with motility has been reported in references as follows: Intercellular contact augments epidermal growth factor receptor (EGFR) and signal transducer and activator of transcription 3 (STAT3)-activatory which increases podoplanin-expression in order to promote squamous cell carcinoma motility; Ganglioside GM3 inhibits hepatoma cell motility via down-regulating activity of EGFR and PI3K/AKT signaling pathway; The ErbB4 CYT2 variant protects EGFR from ligand-induced degradation to enhance cancer cell motility; Weak power frequency magnetic field acting similarly to EGF stimulation, induces acute activations of the EGFR sensitive actin cytoskeleton motility in human amniotic cells [1-4]. Inflammation relation with motility has been reported in references as follows: Tumor necrosis factor-neuropeptide Y cross talk regulates inflammation, epithelial barrier functions, and colonic motility; Mast cells in intestinal inflammation, barrier function, and postoperative motility; Oxytocin regulates gastrointestinal motility, inflammation, macromolecular permeability, and mucosal maintenance in mice; IL-1beta and reactive oxygen species differentially regulate neutrophil directional migration and Basal random motility in a zebrafish injury-induced inflammation model [5-8]. Yet high EGFR_1 inside-out activated inflammation-induced motility through SLC2A1-CCNB2-HMMR-KIF11-NUSAP1-PRC1-UBE2C in lung adenocarcinoma is not clear.
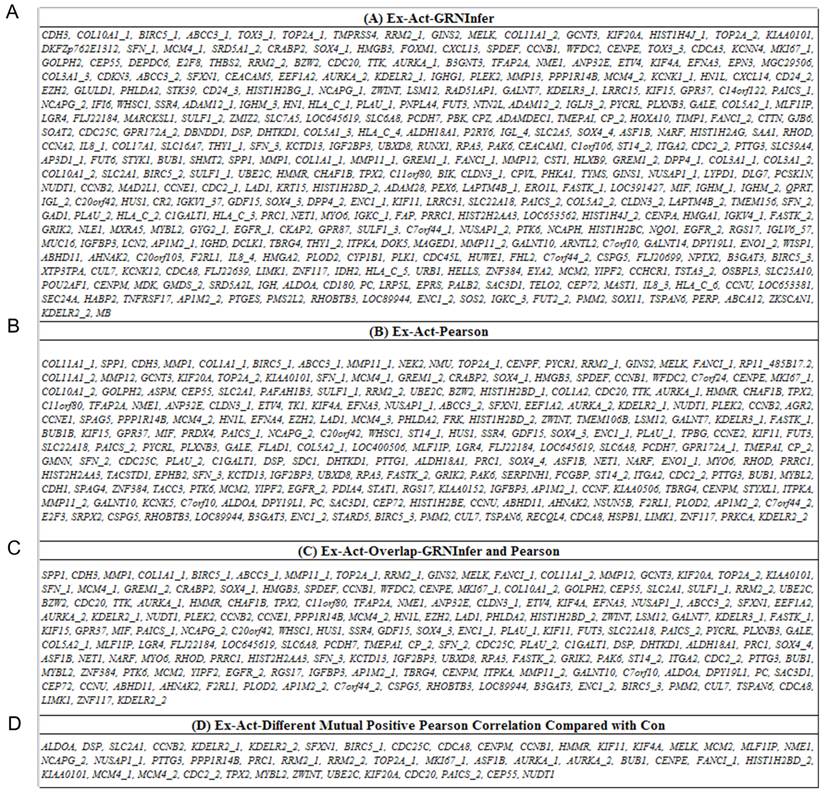
48 different Pearson mutual-positive-correlation EGFR_1-activatory molecular feedback, up- and down-stream network was constructed from 171 overlapping of 366 GRNInfer and 223 Pearson under EGFR_1 CC ≥0.25 in high lung adenocarcinoma compared with low human normal adjacent tissues (Fig. 1A-1D).
(A) EGFR_1-activatory molecules of high lung adenocarcinoma by GRNInfer. (B) EGFR_1-activatory molecules of high lung adenocarcinoma by Pearson. (C) EGFR_1-activatory overlapping molecules of high lung adenocarcinoma by GRNInfer and Pearson. (D) EGFR_1-activatory different mutual-positive-correlation molecules in high lung adenocarcinoma compared with the corresponding low human normal adjacent tissues. Con, human normal adjacent tissues; Ex, lung adenocarcinoma; Act, activation.
Materials and Methods
EGFR_1 was identified by 500 significant molecules from 22,284 genes of 25 high lung adenocarcinoma compared with 25 low human normal adjacent tissues in GEO data set GSE7670 (http://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE7670) for studying high EGFR_1 inside-out activated inflammation-induced motility using SAM [9-14]. GSE7670 raw microarray data were processed by log base 2, two classes were paired and minimum fold change ≥2 selected (the false-discovery rate 0%) [15-18].
Gene expression values of EGFR_1-activatory different molecules were computed in high lung adenocarcinoma compared with the corresponding low human normal adjacent tissues by AVERAGE and STDEV [15, 16].
EGFR_1-activatory different mutual-positive-correlation molecular Pearson coefficients were computed in high lung adenocarcinoma compared with the corresponding low human normal adjacent tissues under EGFR_1 CC ≥0.25 [17, 19, 20].
EGFR_1-activatory molecular network was further constructed in high lung adenocarcinoma by GRNInfer and GVedit tool [18, 21-26].
EGFR_1-activatory molecular knowledge network was further calculated in high lung adenocarcinoma based on terms and occurrence numbers of GO, KEGG, GenMAPP, BioCarta and Disease by MAS [27-34].
Result
EGFR_1-activatory different Pearson mutual-positive-correlation molecular gene expression values were illustrated column diagrams by AVERAGE and STDEV in high lung adenocarcinoma and the corresponding low human normal adjacent tissues, including ALDOA, DSP, SLC2A1, CCNB2, KDELR2_1, KDELR2_2, SFXN1, BIRC5_1, CDC25C, CDCA8, CENPM, CCNB1, HMMR, KIF11, KIF4A, MELK, MCM2, MLF1IP, NME1, NCAPG_2, NUSAP1_1, PTTG3, PPP1R14B, PRC1, RRM2_1, RRM2_2, TOP2A_1, MKI67_1, ASF1B, AURKA_1, AURKA_2, BUB1, CENPE, FANCI_1, HIST1H2BD_2, KIAA0101, MCM4_1, MCM4_2, CDC2_2, TPX2, MYBL2, ZWINT, UBE2C, KIF20A, CDC20, PAICS_2, CEP55, NUDT1, as shown in Supplementary Figure 1A.
EGFR_1-activatory different mutual-positive-correlation molecular Pearson coefficients were illustrated column diagrams in high lung adenocarcinoma showing every molecular positive relationships with others, compared with the corresponding low human normal adjacent tissues containing some molecular negative relationships with others within each group, as shown in Supplementary Figure 1B and 1C, respectively.
EGFR_1-activatory molecular network was further constructed by GRNInfer in high lung adenocarcinoma. EGFR_1 feedback molecular network contained TOP2A_1, MELK, MCM4_1, CCNB1, CENPE, CEP55, RRM2_2, CDC20, NME1, MCM4_2, NCAPG_2, MLF1IP, CDC2_2, PTTG3, BUB1, CDCA8; EGFR_1 upstream SLC2A1, CCNB2, HMMR, KIF11, NUSAP1_1, PRC1, FANCI_1, HIST1H2BD_2, TPX2, MYBL2, UBE2C, PAICS_2, NUDT1; EGFR_1 downstream BIRC5_1, RRM2_1, KIF20A, KIAA0101, MKI67_1, AURKA_1, KIF4A, SFXN1, AURKA_2, KDELR2_1, PPP1R14B, ZWINT, CDC25C, DSP, ASF1B, MCM2, CENPM, ALDOA, KDELR2_2, as shown in Supplementary Figure 2.
EGFR_1-activatory knowledge terms network was further identified by MAS 3.0 in high lung adenocarcinoma. EGFR_1 feedback function network included chromosome, cell cycle, mitosis, cell division, cytokinesis, centrosome, G2/M transition of mitotic cell cycle, positive regulation of ubiquitin ligase activity during mitotic cell cycle, regulation of cell cycle, Contributed--cellular process--Hs cell cycle KEGG, M phase, M phase of mitotic cell cycle, Contributed--cellular process--Hs Cell Cycle-G1 to S control Reactome, AKAP95 role in mitosis and chromosome dynamics, Cell Cycle: G2/M Checkpoint, Cyclins and Cell Cycle Regulation, Estrogen-responsive protein Efp controls cell cycle and breast tumors growth, Sonic Hedgehog (SHH) Receptor Ptc1 Regulates cell cycle, Stathmin and breast cancer resistance to antimicrotubule agents, condensed chromosome kinetochore, negative regulation of cell proliferation, regulation of apoptosis, negative regulation of progression through cell cycle, positive regulation of epithelial cell proliferation, regulation of cell proliferation, Granzyme A mediated Apoptosis Pathway, mitotic chromosome condensation, chromosome organization and biogenesis, DNA replication, neoplasm metastasis, centriole, chromatin-binding, chromosome segregation, apoptotic chromosome condensation, positive regulation of apoptosis, condensed nuclear chromosome, spindle pole body, mitotic spindle checkpoint, cell proliferation, microtubule cytoskeleton, chromosome aberrations, outer kinetochore of condensed chromosome, spindle, microtubule, microtubule associated complex, microtubule motor activity, microtubule-based movement, mitotic chromosome movement towards spindle pole, development, kinetochore assembly, microtubule-based process, DNA replication initiation, Contributed--cellular process--Hs DNA replication Reactome, S phase of mitotic cell cycle, negative regulation of ubiquitin ligase activity during mitotic cell cycle; EGFR_1 upstream microtubule cytoskeleton, embryonic development (sensu Mammalia), cell cycle, mitosis, thymus development, cell division, regulation of cell cycle, Contributed--cellular process--Hs cell cycle KEGG, cytokinesis, M phase, M phase of mitotic cell cycle, Estrogen-responsive protein Efp controls cell cycle and breast tumors growth, cell motility, locomotion, locomotory behavior, neoplasm metastasis, spindle pole, spindle microtubule, microtubule motor activity, microtubule-based movement, mitotic spindle organization and biogenesis, mitotic centrosome separation, spindle pole body organization and biogenesis, microtubule-based process, microtubule, cytokinesis after mitosis, mitotic chromosome condensation, establishment of mitotic spindle localization, positive regulation of mitosis, mitotic spindle elongation, chromosome, nucleosome assembly, establishment and or maintenance of chromatin architecture, chromatin assembly or disassembly, protein complex assembly, chromatin, chromatin assembly, parkinson disease, cell proliferation, Role of Ran in mitotic spindle regulation, negative regulation of apoptosis, negative regulation of programmed cell death, spindle organization and biogenesis, positive regulation of exit from mitosis, negative regulation of ubiquitin ligase activity during mitotic cell cycle, positive regulation of ubiquitin ligase activity during mitotic cell cycle, regulation of cell proliferation, positive regulation of cell proliferation; EGFR_1 downstreamactin filament organization, skeletal development, development, erythrocyte differentiation, chromosome, centriole, spindle microtubule, cytoplasmic microtubule, interphase microtubule organizing center, microtubule-binding, G2/M transition of mitotic cell cycle, cytokinesis, apoptosis, anti-apoptosis, cell cycle, mitosis, positive regulation of exit from mitosis, spindle checkpoint, positive regulation of progression through mitotic cell cycle, establishment of chromosome localization, negative regulation of apoptosis, microtubule cytoskeleton, Contributed--cellular process--Hs Apoptosis, negative regulation of programmed cell death, bcellsurvivalPathway, DNA replication, regulation of mitosis, traversing start control point of mitotic cell cycle, cell proliferation, cell division, Contributed--cellular process--Hs cell cycle KEGG, M phase, M phase of mitotic cell cycle, Cell Cycle: G2/M Checkpoint, Regulation of cell cycle progression by Plk3, Sonic Hedgehog (SHH) Receptor Ptc1 Regulates cell cycle, condensed chromosome kinetochore, epidermis development, keratinocyte differentiation, microtubule associated complex, microtubule motor activity, organelle organization and biogenesis, microtubule-based movement, microtubule-based process, chromatin, DNA replication initiation, Contributed--cellular process--Hs DNA replication Reactome, Contributed--cellular process--Hs Cell Cycle-G1 to S control Reactome, neoplasm metastasis, regulation of cell cycle, chromatin assembly or disassembly, nucleosome assembly, spermatogenesis, chromatin modification, cell differentiation, centrosome, spindle, mitotic cell cycle, spindle organization and biogenesis, Role of Ran in mitotic spindle regulation, DNA replication origin-binding, microtubule, as shown in Supplementary Figure 3.
Discussion
EGFR_1-activatory different molecular Pearson mutual-positive-correlation feedback, up- and down-stream network was setup in high lung adenocarcinoma compared with the corresponding low human normal adjacent tissues (Supplementary Figure 2). Our identified EGFR_1 inside-out upstream activated molecular network showed SLC2A1 (solute carrier family 2 (facilitated glucose transporter) member 1), CCNB2 (cyclin B2), HMMR (hyaluronan-mediated motility receptor (RHAMM)), KIF11 (kinesin family member 11), NUSAP1 (nucleolar and spindle associated protein 1), PRC1 (protein regulator of cytokinesis 1), UBE2C (ubiquitin-conjugating enzyme E2C) in high lung adenocarcinoma. EGFR_1 inside-out upstream activated terms network includes intracellular, membrane fraction, cytoplasm, plasma membrane, integral to membrane, basolateral plasma membrane, transmembrane transport, nucleus, cytosol, cell surface; T cell homeostasis, inflammation; microtubule cytoskeleton, embryonic development (sensu Mammalia), cell cycle, mitosis, thymus development, cell division, regulation of cell cycle, Contributed--cellular process--Hs cell cycle KEGG, cytokinesis, M phase, M phase of mitotic cell cycle, estrogen-responsive protein Efp controls cell cycle and breast tumors growth, cell motility, locomotion, locomotory behavior, neoplasm metastasis, spindle pole, spindle microtubule, microtubule motor activity, microtubule-based movement, mitotic spindle organization and biogenesis, mitotic centrosome separation, spindle pole body organization and biogenesis, microtubule-based process, microtubule, cytokinesis after mitosis, mitotic chromosome condensation, establishment of mitotic spindle localization, positive regulation of mitosis, mitotic spindle elongation, spindle organization and biogenesis, positive regulation of exit from mitosis, negative regulation of ubiquitin ligase activity during mitotic cell cycle, positive regulation of ubiquitin ligase activity during mitotic cell cycle, regulation of cell proliferation, positive regulation of cell proliferation; transporter activity, sugar porter activity, glucose transporter activity, protein-binding, substrate-specific transmembrane transporter activity, hyaluronic acid-binding, carbohydrate-binding, nucleotide-binding, ATP-binding, motor activity, DNA-binding, ubiquitin-protein ligase activity, ligase activity in high lung adenocarcinoma based on integrative GO, KEGG, GenMAPP, BioCarta and disease databases (Supplementary Figure 3). Therefore, we propose high EGFR_1 inside-out activated inflammation-induced motility through SLC2A1-CCNB2-HMMR-KIF11-NUSAP1-PRC1-UBE2C in lung adenocarcinoma.
Motility with clinical correlation has been reported in references. Such as, swimming motility in a longitudinal collection of clinical isolates of Burkholderia cepacia complex bacteria from people with cystic fibrosis; Identification of clinical outcome measures for recovery of gastrointestinal motility in postoperative ileus; Integrin-free tetraspanin CD151 can inhibit tumor cell motility upon clustering and is a clinical indicator of prostate cancer progression; 4D tracking of clinical seminal samples for quantitative characterization of motility parameters; Clinical Utility of Wireless Motility Capsule in Patients with Suspected Multiregional Gastrointestinal Dysmotility [35-39].
Motility positive relationship with cyclin, kinesin has been reported in references as follows: Over-expression of cyclin D1 induces glioma invasion by increasing matrix metalloproteinase activity and cell motility; Cyclin D1 interacts and collaborates with Ral GTPases enhancing cell detachment and motility; Cyclin D1 governs adhesion and motility of macrophages; The regulation of SIRT2 function by cyclin-dependent kinases affects cell motility; Cyclin-dependent kinase 5 activity controls cell motility and metastatic potential of prostate cancer cells [40-44]. Kinesin-dependent motility generation as target mechanism of cadmium intoxication; Effects of alpha-tubulin K40 acetylation and detyrosination on kinesin-1 motility in a purified system; TRIM3 regulates the motility of the kinesin motor protein KIF21B; Control and gating of kinesin-microtubule motility on electrically heated thermo-chips [45-48].
In summary, EGFR_1-activatory molecular Pearson mutual-positive-correlation network was constructed in high lung adenocarcinoma from the overlapping molecules of GRNInfer with Pearson. We propose and verify high EGFR_1 inside-out activated inflammation-induced motility through SLC2A1-CCNB2-HMMR-KIF11-NUSAP1-PRC1-UBE2C in lung adenocarcinoma. High EGFR_1 inside-out activated inflammation-induced motility through SLC2A1-CCNB2-HMMR-KIF11-NUSAP1-PRC1-UBE2C is very useful to develop a new route and identify novel markers and potential drugs for prognosis and therapy of lung adenocarcinoma.
Supplementary Material
Supplementary Figures 1 - 3.
Acknowledgements
This work was supported by the National Natural Science Fund (61171114) and Key Fund (61433015), the National Social Science Major Fund (14ZDB154) of China, Fundamental Research Funds for the Central Universities (BUPT Project No: 2014RC0201), Research Innovation Fund of Beijing University of Posts and Telecommunications (BUPT Project No: 2014XD-01), Research Innovation Fund for College Students of Beijing University of Posts and Telecommunications.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Fujii M, Honma M, Takahashi H, Ishida-Yamamoto A, Iizuka H. Intercellular contact augments epidermal growth factor receptor (EGFR) and signal transducer and activator of transcription 3 (STAT3)-activation which increases podoplanin-expression in order to promote squamous cell carcinoma motility. Cell Signal. 2013;25:760-5 doi:S0898-6568(12)00341-5 [pii] 10.1016/j.cellsig.2012.12.004
2. Huang X, Li Y, Zhang J, Xu Y, Tian Y, Ma K. Ganglioside GM3 inhibits hepatoma cell motility via down-regulating activity of EGFR and PI3K/AKT signaling pathway. J Cell Biochem. 2013;114:1616-24 doi:10.1002/jcb.24503
3. Kiuchi T, Ortiz-Zapater E, Monypenny J, Matthews DR, Nguyen LK, Barbeau J. et al. The ErbB4 CYT2 variant protects EGFR from ligand-induced degradation to enhance cancer cell motility. Sci Signal. 2014;7:ra78. doi:7/339/ra78 [pii] 10.1126/scisignal.2005157
4. Wu X, Cao MP, Shen YY, Chu KP, Tao WB, Song WT. et al. Weak power frequency magnetic field acting similarly to EGF stimulation, induces acute activations of the EGFR sensitive actin cytoskeleton motility in human amniotic cells. PLoS One. 2014;9:e87626. doi:10.1371/journal.pone.0087626 PONE-D-13-30033 [pii]
5. Chandrasekharan B, Jeppsson S, Pienkowski S, Belsham DD, Sitaraman SV, Merlin D. et al. Tumor necrosis factor-neuropeptide Y cross talk regulates inflammation, epithelial barrier functions, and colonic motility. Inflamm Bowel Dis. 2013;19:2535-46 doi:10.1097/01.MIB.0000437042.59208.9f
6. de Jonge WJ. Mast cells in intestinal inflammation, barrier function, and postoperative motility. J Pediatr Gastroenterol Nutr. 2011;53(Suppl 2):S56-7
7. Welch MG, Margolis KG, Li Z, Gershon MD. Oxytocin regulates gastrointestinal motility, inflammation, macromolecular permeability, and mucosal maintenance in mice. Am J Physiol Gastrointest Liver Physiol. 2014;307:G848-62 doi:ajpgi.00176.2014 [pii] 10.1152/ajpgi.00176.2014
8. Yan B, Han P, Pan L, Lu W, Xiong J, Zhang M. et al. IL-1beta and reactive oxygen species differentially regulate neutrophil directional migration and Basal random motility in a zebrafish injury-induced inflammation model. J Immunol. 2014;192:5998-6008 doi:jimmunol.1301645 [pii] 10.4049/jimmunol.1301645
9. Storey. JD. A direct approach to false discovery rates. J Roy Stat Soc, Ser B. 2002;64:479-98
10. Wang L, Huang J, Jiang M, Sun L. MYBPC1 computational phosphoprotein network construction and analysis between frontal cortex of HIV encephalitis (HIVE) and HIVE-control patients. Cell Mol Neurobiol. 2011;31:233-41 doi:10.1007/s10571-010-9613-x
11. Wang L, Huang J, Jiang M. CREB5 computational regulation network construction and analysis between frontal cortex of HIV encephalitis (HIVE) and HIVE-control patients. Cell Biochem Biophys. 2011;60:199-207
12. Wang L, Huang J, Jiang M, Zheng X. AFP computational secreted network construction and analysis between human hepatocellular carcinoma (HCC) and no-tumor hepatitis/cirrhotic liver tissues. Tumor Biol. 2010;31:417-25
13. Wang L, Huang J, Jiang M. RRM2 computational phosphoprotein network construction and analysis between no-tumor hepatitis/cirrhotic liver tissues and human hepatocellular carcinoma (HCC). Cell Physiol Biochem. 2010;26:303-10
14. Huang JX, Wang L, Jiang MH. TNFRSF11B computational development network construction and analysis between frontal cortex of HIV encephalitis (HIVE) and HIVE-control patients. J Inflamm (Lond). 2010;7:50
15. You J, Wang L, Huang J, Jiang M, Chen Q, Wang Y. et al. Low glucose transporter SLC2A5-inhibited human normal adjacent lung adenocarcinoma cytoplasmic pro-B cell development mechanism network. Mol Cell Biochem. 2015;399:71-6 doi:10.1007/s11010-014-2233-x
16. Huang J, Wang L, Jiang M, Chen Q, Jiang Z, Feng H. AGR2-mediated lung adenocarcinoma metastasis novel mechanism network through repression with interferon coupling cytoskeleton to steroid metabolism-dependent humoral immune response. Cell Immunol. 2014;290:102-6 doi:S0008-8749(14)00093-8 [pii] 10.1016/j.cellimm.2014.05.008
17. Wang L, Huang J, Jiang M, Diao H, Zhou H, Li X. et al. Cartilage oligomeric matrix protein (COMP)-mediated cell differentiation to proteolysis mechanism networks from human normal adjacent tissues to lung adenocarcinoma. Anal Cell Pathol (Amst). 2013;36:93-105 doi:ER0276047N6RPK77 [pii] 10.3233/ACP-130084
18. Sun Y, Wang L, Jiang M, Huang J, Liu Z, Wolfl S. Secreted Phosphoprotein 1 Upstream Invasive Network Construction and Analysis of Lung Adenocarcinoma Compared with Human Normal Adjacent Tissues by Integrative Biocomputation. Cell Biochem Biophys. 2010;56:59-71
19. Wang L, Huang J, Jiang M, Diao H, Zhou H, Li X. et al. Adenosylmethionine Decarboxylase 1 (AMD1)-Mediated mRNA Processing and Cell Adhesion Activated & Inhibited Transition Mechanisms by Different Comparisons Between Chimpanzee and Human Left Hemisphere. Cell Biochem Biophys. 2014;70:279-88 doi:10.1007/s12013-014-9902-y
20. Diao H, Wang L, Huang J, Jiang M, Zhou H, Li X. et al. BRCA1-mediated inflammation and growth activated & inhibited transition mechanisms by different comparisons between no-tumor hepatitis/cirrhotic tissues and HCC. J Cell Biochem. 2013;115:641-50 doi:10.1002/jcb.24699
21. Wang Y, Joshi T, Zhang XS, Xu D, Chen L. Inferring gene regulatory networks from multiple microarray datasets. Bioinformatics (Oxford, England). 2006;22:2413-20
22. Sun L, Wang L, Jiang M, Huang J, Lin H. Glycogen debranching enzyme 6 (AGL), enolase 1 (ENOSF1), ectonucleotide pyrophosphatase 2 (ENPP2_1), glutathione S-transferase 3 (GSTM3_3) and mannosidase (MAN2B2) metabolism computational network analysis between chimpanzee and human left cerebrum. Cell Biochem Biophys. 2011;61:493-505
23. Wang L, Sun L, Huang J, Jiang M. Cyclin-dependent kinase inhibitor 3 (CDKN3) novel cell cycle computational network between human non-malignancy associated hepatitis/cirrhosis and hepatocellular carcinoma (HCC) transformation. CELL PROLIFERAT. 2011;44:291-9
24. Wang L, Huang J, Jiang M, Sun L. Survivin (BIRC5) cell cycle computational network in human no-tumor hepatitis/cirrhosis and hepatocellular carcinoma transformation. J Cell Biochem. 2011;112:1286-94
25. Wang L, Sun Y, Jiang M, Zheng X. Integrative decomposition procedure and Kappa statistics for the distinguished single molecular network construction and analysis. J Biomed Biotechnol. 2009;2009:726728
26. Wang L, Sun Y, Jiang M, Zhang S, Wolfl S. FOS proliferating network construction in early colorectal cancer (CRC) based on integrative significant function cluster and inferring analysis. Cancer Invest. 2009;27:816-24
27. Wang L, Huang J, Jiang M, Chen Q, Jiang Z, Feng H. CAMK1 phosphoinositide signal-mediated protein sorting and transport network in human hepatocellular carcinoma (HCC) by biocomputation. Cell Biochem Biophys. 2014;70:1011-6 doi:10.1007/s12013-014-0011-8
28. Qi L, Wang L, Huang J, Jiang M, Diao H, Zhou H. et al. Activated amelogenin Y-linked (AMELY) regulation and angiogenesis in human hepatocellular carcinoma by biocomputation. Oncol Lett. 2013;5:1075-9 doi:10.3892/ol.2013.1122 ol-05-03-1075 [pii]
29. Wang L, Huang J, Jiang M, Lin H, Qi L, Diao H. Activated PTHLH Coupling Feedback Phosphoinositide to G-Protein Receptor Signal-Induced Cell Adhesion Network in Human Hepatocellular Carcinoma by Systems-Theoretic Analysis. ScientificWorldJournal. 2012;2012:428979. doi:10.1100/2012/428979
30. Wang L, Huang J, Jiang M, Lin H, Qi L, Diao H. Inhibited PTHLH downstream leukocyte adhesion-mediated protein amino acid N-linked glycosylation coupling Notch and JAK-STAT cascade to iron-sulfur cluster assembly-induced aging network in no-tumor hepatitis/cirrhotic tissues (HBV or HCV infection) by systems-theoretical analysis. Integr Biol (Camb). 2012;4:1256-62 doi:10.1039/c2ib20148h
31. Wang L, Huang J, Jiang M, Lin H. Tissue-specific transplantation antigen P35B (TSTA3) immune response-mediated metabolism coupling cell cycle to postreplication repair network in no-tumor hepatitis/cirrhotic tissues (HBV or HCV infection) by biocomputation. Immunol Res. 2012;52:258-68
32. Wang L, Huang J, Jiang M, Lin H. Signal transducer and activator of transcription 2 (STAT2) metabolism coupling postmitotic outgrowth to visual and sound perception network in human left cerebrum by biocomputation. J Mol Neurosci. 2012;47:649-58 doi:10.1007/s12031-011-9702-4
33. Lin H, Wang L, Jiang M, Huang J, Qi L. P-glycoprotein (ABCB1) inhibited network of mitochondrion transport along microtubule and BMP signal-induced cell shape in chimpanzee left cerebrum by systems-theoretical analysis. Cell Biochem Funct. 2012;30:582-7 doi:10.1002/cbf.2837
34. Huang J, Wang L, Jiang M, Lin H, Qi L, Diao H. PTHLH coupling upstream negative regulation of fatty acid biosynthesis and Wnt receptor signal to downstream peptidase activity-induced apoptosis network in human hepatocellular carcinoma by systems-theoretical analysis. J Recept Signal Transduct Res. 2012;32:250-6 doi:10.3109/10799893.2012.700717
35. Zlosnik JE, Mori PY, To D, Leung J, Hird TJ, Speert DP. Swimming motility in a longitudinal collection of clinical isolates of Burkholderia cepacia complex bacteria from people with cystic fibrosis. PLoS One. 2014;9:e106428. doi:10.1371/journal.pone.0106428 PONE-D-14-24988 [pii]
36. van Bree SH, Bemelman WA, Hollmann MW, Zwinderman AH, Matteoli G, El Temna S. et al. Identification of clinical outcome measures for recovery of gastrointestinal motility in postoperative ileus. Ann Surg. 2014;259:708-14 doi:10.1097/SLA.0b013e318293ee55
37. Palmer TD, Martinez CH, Vasquez C, Hebron KE, Jones-Paris C, Arnold SA. et al. Integrin-free tetraspanin CD151 can inhibit tumor cell motility upon clustering and is a clinical indicator of prostate cancer progression. Cancer Res. 2014;74:173-87 doi:0008-5472.CAN-13-0275 [pii] 10.1158/0008-5472.CAN-13-0275
38. Di Caprio G, El Mallahi A, Ferraro P, Dale R, Coppola G, Dale B. et al. 4D tracking of clinical seminal samples for quantitative characterization of motility parameters. Biomed Opt Express. 2014;5:690-700 doi:10.1364/BOE.5.000690 199475 [pii]
39. Arora Z, Parungao JM, Lopez R, Heinlein C, Santisi J, Birgisson S. Clinical Utility of Wireless Motility Capsule in Patients with Suspected Multiregional Gastrointestinal Dysmotility. Dig Dis Sci. 2014 doi:10.1007/s10620-014-3431-9
40. Arato-Ohshima T, Sawa H. Over-expression of cyclin D1 induces glioma invasion by increasing matrix metalloproteinase activity and cell motility. Int J Cancer. 1999;83:387-92 doi:10.1002/(SICI)1097-0215(19991029)83:3<387::AID-IJC15>3.0.CO;2-O [pii]
41. Fernandez RM, Ruiz-Miro M, Dolcet X, Aldea M, Gari E. Cyclin D1 interacts and collaborates with Ral GTPases enhancing cell detachment and motility. Oncogene. 2011;30:1936-46 doi:onc2010577 [pii] 10.1038/onc.2010.577
42. Neumeister P, Pixley FJ, Xiong Y, Xie H, Wu K, Ashton A. et al. Cyclin D1 governs adhesion and motility of macrophages. Mol Biol Cell. 2003;14:2005-15 doi:10.1091/mbc.02-07-0102 02-07-0102 [pii]
43. Pandithage R, Lilischkis R, Harting K, Wolf A, Jedamzik B, Luscher-Firzlaff J. et al. The regulation of SIRT2 function by cyclin-dependent kinases affects cell motility. J Cell Biol. 2008;180:915-29 doi:jcb.200707126 [pii] 10.1083/jcb.200707126
44. Strock CJ, Park JI, Nakakura EK, Bova GS, Isaacs JT, Ball DW. et al. Cyclin-dependent kinase 5 activity controls cell motility and metastatic potential of prostate cancer cells. Cancer Res. 2006;66:7509-15 doi:66/15/7509 [pii] 10.1158/0008-5472.CAN-05-3048
45. Bohm KJ. Kinesin-dependent motility generation as target mechanism of cadmium intoxication. Toxicol Lett. 2014;224:356-61 doi:S0378-4274(13)01395-7 [pii] 10.1016/j.toxlet.2013.11.004
46. Kaul N, Soppina V, Verhey KJ. Effects of alpha-tubulin K40 acetylation and detyrosination on kinesin-1 motility in a purified system. Biophys J. 2014;106:2636-43 doi:S0006-3495(14)00505-0 [pii] 10.1016/j.bpj.2014.05.008
47. Labonte D, Thies E, Pechmann Y, Groffen AJ, Verhage M, Smit AB. et al. TRIM3 regulates the motility of the kinesin motor protein KIF21B. PLoS One. 2013;8:e75603. doi:10.1371/journal.pone.0075603 PONE-D-13-16444 [pii]
48. Ramsey L, Schroeder V, van Zalinge H, Berndt M, Korten T, Diez S. et al. Control and gating of kinesin-microtubule motility on electrically heated thermo-chips. Biomed Microdevices. 2014;16:459-63 doi:10.1007/s10544-014-9848-2
Author contact
Corresponding author: Lin Wang (Prof. Dr.), Biomedical Center, School of Electronics Engineering, Beijing University of Posts and Telecommunications, Beijing, 100876, China. Email: wanglin98org.cn Tel: 8610-13240981826

 Global reach, higher impact
Global reach, higher impact