Impact Factor ISSN: 1837-9664
J Cancer 2020; 11(11):3106-3113. doi:10.7150/jca.38411 This issue Cite
Research Paper
The Status of the EGFR T790M Mutation is associated with the Clinical Benefits of Osimertinib Treatment in Non-small Cell Lung Cancer Patients: A Meta-Analysis
1. National Cancer Center/National Clinical Research Center for Cancer/Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, 100021, China.
2. State Key Laboratory of Molecular Oncology, Department of Medical Oncology, National Cancer Center/National Clinical Research Center for Cancer/Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, 100021, China.
3. Health Service Department of the Guard Bureau of the Joint Staff Department, Beijing, 100017, China.
Received 2019-7-14; Accepted 2019-11-30; Published 2020-3-4
Abstract
Background and Purpose: Pervious studies have demonstrated that the loss of EGFR T790M after Osimertinib treatment may be the cause of Osimertinib resistance. Here, we conducted a meta-analysis to evaluate the association between the persistence of EGFR T790M and the clinical benefits of Osimertinib in non-small cell lung cancer (NSCLC) patients with baseline EGFR T790M mutation.
Experimental design and Methods: PUBMED, EMBASE, and Cochrane databases were searched for eligible studies that provided the survival outcomes including overall survival (OS), progression-free survival (PFS) or time to discontinuation (TTD) data for each patient treated with Osimertinib with the status of the T790M mutation tested after Osimertinib resistance. The hazard ratios (HRs) and their 95% confidence intervals (CI) were calculated for each study.
Results: In total, eight eligible studies were included in the analysis, among which six studies provided the data on PFS, and the other two studies provided the TTD data. Overall, 312 patients (151 patients with the persistence of T790M) were identified. The persistence of T790M was associated with longer PFS (HR, 0.40; 95% CI, 0.19-0.84; P=0.01) and TTD (HR, 0.54; 95% CI, 0.39-0.76; P=0.0004). Furthermore, overall analysis the survival outcomes including PFS and TTD subgroups also showed preferable clinical benefits for patients with the T790M persistence (HR, 0.57; 95%CI, 0.45-0.73; P<0.00001).
Conclusions: Our findings confirm the persistence of T790M is associated with the clinical benefits of Osimertinib in NSCLC patients with baseline EGFR T790M mutation treated with Osimertinib.
Keywords: non-small cell lung cancer, EGFR T790M mutation, Osimertinib, resistance, meta-analysis
Introduction
Lung cancer is the most common cancer type and is one of the major causes of cancer-related deaths worldwide [1]. The clinical management of lung cancer has in recent years taken advantage of the increasing awareness of pathogenesis and drug resistance at the molecular level of tumors, especially in non-small cell lung cancer (NSCLC). Epidermal growth factor receptor (EGFR) gene mutations are common drivers of NSCLC. EGFR tyrosine kinase inhibitors (EGFR-TKIs) have revolutionized the treatment for NSCLC patients with EGFR driver mutations [2, 3]. However, although these new generations of TKI-targeted drugs have higher disease control rates and initial response rates in NSCLC patients with EGFR-sensitive mutations, the emergence of acquired resistance to EGFR-TKIs is inevitable. Tyrosine790 is a “gatekeeper” residue that is integral for regulating the affinity of TKI-targeted drugs in the ATP binding pocket, while about 50-60 % of patients have a secondary T790M mutation. With the Tyrosine790 mutation, the ATP binding pocket has increased affinity for ATP and confers resistance due to competition with EGFR-TKIs [4].
The EGFR T790M mutation is specifically targeted by the third-generation EGFR-TKI Osimertinib. For patients with the T790M mutation, Osimertinib has provided significant survival benefits compared to cytotoxic chemotherapy and has been approved as a standard treatment [5-7]. In the AURA3 study, the median PFS was significantly prolonged in patients receiving Osimertinib (10.1 vs. 4.4 months; P<0.001). The objective response rate of Osimertinib was significantly higher compared with chemotherapy (71% vs. 31%; P<0.001) [8]. In addition, Osimertinib was used as a first-line therapy for NSCLC patients with EGFR-mutations, and median PFS was significantly longer than first-line EGFR-TKIs (18.9 and 10.2 months, respectively; P<0.0001) in the FLAURA study [9].
Unfortunately, even in T790M-positive tumors, the response to Osimertinib is not permanent and drug resistance will occur sooner or later. Complex mechanisms that mediate resistance to Osimertinib have been demonstrated, such as the acquisition of tertiary EGFR mutations (e.g. EGFR 797S, L718Q mutations), HER2 or MET amplification, BRAF mutations, and the histologic changes to small cell transformation [10]. It is worth noting that even though the mechanisms of Osimertinib resistance are heterogeneous, they can be conceptualized as binary variables: some patients lose secondary T790M mutations when they acquire Osimertinib resistance, while some patients remain a T790M positive status [11]. Previous reports have shown that changes in T790M mutation status appear to be associated with the clinical benefits of Osimertinib in baseline EGFR T790M-positive NSCLC patients [12-14]. A study by Lin et al. consistently found that loss of the T790 M mutation was associated with the shortest PFS (median 2.6 months, 95% CI 1.3-NR) of the Osimertinib resistance group [12]. Similarly, a retrospective study conducted by Zhou et al (2018) also demonstrated a shorter PFS in patients with T790M loss when patients acquired Osimertinib resistance [13]. Overall, studies have found that the presence or absence of the T790M mutation may be associated with resistance to Osimertinib, but the mechanism of this action remains unknown [15-17]. Hence, we conducted a meta-analysis of the published literatures to explore the association between the persistence of T790M and the clinical benefits of Osimertinib in NSCLC patients with baseline EGFR T790M mutation.
Methods
Search strategy
This study was conducted in compliance with the recommendations of the Cochrane Handbook for Systematic Reviews of Interventions and reported based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement guidelines [18]. PUBMED, EMBASE, and Cochrane databases were searched for eligible published articles that reported survival time for each Osimertinib treated patient with both positive baseline EGFR T790M and EGFR T790M detection after Osimertinib resistance nearly 5 years from January 1st, 2014 to September 1st, 2019. The search terms utilized included: non-small cell lung cancer, T790M, epidermal growth factor receptor, and Osimertinib. Case reports, letters, conference abstracts, comments, editorials, proceedings, and personal communications were excluded. Moreover, the reference lists of all trials fulfilling the eligibility criteria were examined for any relevant studies missed in initial searches.
Data extraction and quality assessment
The following information was extracted from the chosen studies: first author or correspondent author's name, publication year, study design, baseline characteristics, number of participants and major outcomes of T790M persistence and loss when Osimertinib progressed, and survival outcome for each patient. Two researchers independently extracted the data, and any discrepancy was resolved by discussion. The quality of the included studies was assessed using the Newcastle-Ottawa Scale (NOS) independently by two researchers [19].
Statistical analysis
For each individual study, HR with a survival result of 95% CI was extracted. If there was no HR of 95% CI, the hazard ratios and corresponding 95% confident intervals for each included study were calculated using the Cox regression model. The method of fixed or random-effects inverse-variance- weighted was then used to pool the hazard ratios. A χ2-based homogeneity test was performed and the inconsistency index (I2) and Q statistics were determined. We classified the I2 value<50% as having homogeneity and a fixed-effect model was well accepted. An I2 value>50% predicted potential heterogeneity. If heterogeneity existed, subgroup analysis was used to weaken its effects. Else, we synthesized the results with a randomized effect model if no definite heterogeneity was detected. Pooling effects were calculated and a two-sided p-value<0.05 was considered to indicate statistical significance. Sensitivity analysis was performed using the leave-one method. At last, the publication bias was analyzed. All analyses were performed using the comprehensive Meta-analysis statistical software Review manager 5.2 (The Nordic Cochrane Centre, Rigshospitalet 2008).
Results
Selection of eligible studies
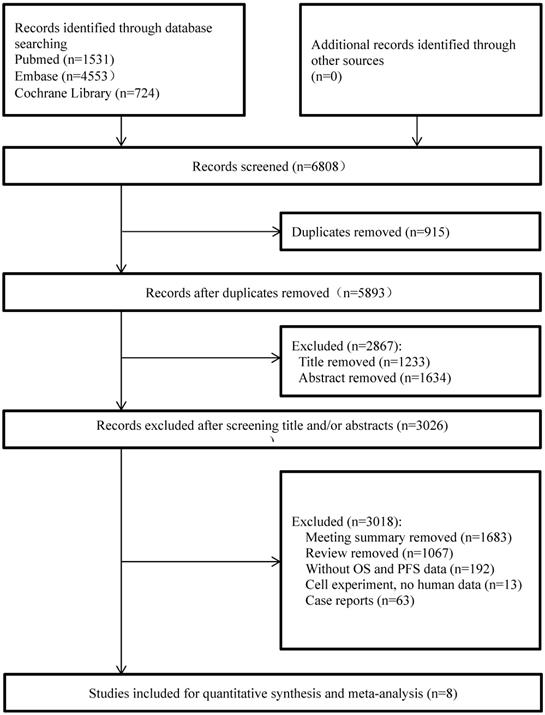
The initial search strategy identified a total of 6,808 related articles, of which 1,531 were from PUBMED, 4,553 were from EMBASE, and 724 were from the Cochrane Library. Due to duplications, we deleted 915 studies. After the titles and abstracts were screened, 2,867 studies were excluded because they did not meet the inclusion criteria. Then, we carefully reviewed the full text of the remaining 3,026 eligible studies and excluded 1,683 conference abstracts, 1,067 reviews, 63 case reports, 192 articles without specific PFS or TTD data, and 13 studies related to cell experiments. After filtering, a total of eight clinical studies were selected for final analysis. A flow chart describing the study selection eligibility is shown in Figure 1.
Study flow diagram.
Characteristics of included studies and quality assessment
A total of eight studies with 312 patients were included in the analysis. The characteristics of the included studies are outlined in Table 1. These studies included 312 patients with baseline T790M mutations who were orally treated with Osimertinib to resistance and were re-biopsied for T790M mutation status in the tissue or blood, of which outcomes included PFS and TTD, respectively. A total of 6 studies reported the outcome of PFS and 2 studies reported TTD. The NOS scores of the eight studies are listed in Table 2. In general, the studies are of good quality with the NOS scores ranging from 6 to 8 points.
Characteristics of included studies.
| Studies | Type | Year | Nation | Age | Gender | Re- biopsies | T790M positive | T790M negative | Outcome | Pos/Neg HR | 95% CIs |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Caicun 2019 [13] | retrospective | 2019 | China | <65(20) >=65(11) | F(16), M(15) | 31 | 16 | 15 | PFS | 0.12 | 0.039-0.368 |
| GeoffreyR 2018 [14] | retrospective | 2018 | American | Unknown | F(28), M(13) | 41 | 14 | 27 | TTD | 0.411 | 0.21-0.804 |
| James 2018 [34] | retrospective | 2017 | American | Unknown | uncategorized | 110 | 58 | 52 | TTD | 0.597 | 0.406-0.878 |
| Vassiliki 2018 [35] | prospective | 2018 | UK | Unknown | uncategorized | 64 | 28 | 36 | PFS | 0.779 | 0.472-1.287 |
| Michael E 2018 [36] | retrospective | 2018 | American | 59 | F(5), M(4) | 9 | 7 | 2 | PFS | 0.118 | 0.011-1.327 |
| Keke 2018 [10] | retrospective | 2018 | China | 66 | F(5), M(4) | 9 | 7 | 2 | PFS | 0.282 | 0.039-2.027 |
| Chia 2017 [12] | prospective | 2017 | Taiwan | 59 | uncategorized | 41 | 18 | 23 | PFS | 0.849 | 0.452-1.595 |
| Young-Chul 2019 [15] | prospective | 2019 | Korea | Unknown | uncategorized | 7 | 3 | 4 | PFS | 0.24 | 0.03-2.20 |
Quality assessment included in the study - using the NOS score.
| Studies | Selection 1 | Selection 2 | Selection 3 | Selection 4 | Comparability | Outcome assessment | Score | ||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | |||||||
| Caicun 2019[13] | * | * | * | * | * | * | * | * | ******** |
| GeoffreyR 2018[14] | * | * | * | * | * | * | * | * | ******** |
| James 2018[34] | * | * | * | * | * | * | * | ******* | |
| Vassiliki 2018[35] | * | * | * | * | * | * | * | * | ******** |
| Michael E 2018[36] | * | * | * | * | * | * | * | ******* | |
| Keke 2018[10] | * | * | * | * | * | * | ****** | ||
| Chia 2017[12] | * | * | * | ** | * | * | * | ******** | |
| Young-Chul 2019[15] | * | * | * | * | * | * | * | ******* | |
Notes In this evaluation form, *represents one point, ** represents two points, ******or more indicates that the quality of the article is high and credible out of 9 points.
Meta-analysis
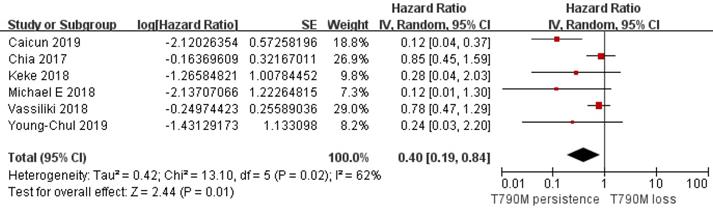
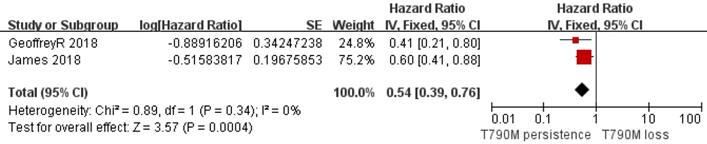
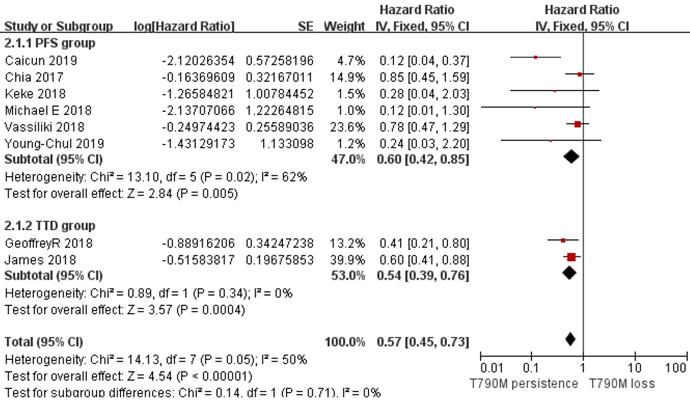
Of the 312 patients, 151 had T790M mutation persistence after Osimertinib resistance, while 161 lost the T790M mutation with resistance to Osimertinib. Analysis of the PFS data provided by 6 clinical studies showed that the T790M persistence group had a longer PFS than the T790M loss group (pooled HR=0.40; 95% CI 0.19 to 0.84; P=0.01; I2=62%; Figure 2). Considering the very nature of the retrospective design, there was potential heterogeneity among included studies. We classified I2 higher than 50% which is acceptable and does not affect the reliability of the results. In addition, an analysis of the other two studies on TTD also supported a longer TTD in the T790M persistence group vs. the T790M loss group (pooled HR=0.54; 95% CI 0.39 to 0.76; P=0.0004; Figure 3) without heterogeneity (I2=0). Although only two studies provided the TTD data, the study showed that the heterogeneity between them was zero, which supported the result was credible. Considering that both PFS and TTD belonged to the survival outcomes, we comprehensively analyzed the survival outcomes of the two subgroups, which showed that the T790M persistence group had longer survival outcomes than the T790M loss group (HR=0.57; 95% CI 0.45 to 0.73; P<0.00001; Figure 4), whose I2 equaled 50% due to the potentially acceptable heterogeneity from the included retrospective study. Only one study showed specific OS data, so we did not include it in our meta-analysis. However, the study also showed that the OS data of the T790M loss group was also decreased than that of the persistence group (P=0.021)[13]. From this we believe that the persistence of T790M is associated with the benefits of clinical outcomes in NSCLC patients treated with Osimertinib.
Forest plot of PFS included in the study in the persistence of T790M NSCLC patients compared with the T790M loss group when they acquired resistance to Osimertinib and had a positive T790M mutation at baseline.
Forest plot of TTD included in the study in the persistence of T790M NSCLC patients compared with the T790M loss group when they acquired resistance to Osimertinib and had a positive T790M mutation at baseline.
Forest plot of survival outcomes included in the study in the persistence of T790M NSCLC patients compared with the T790M loss group when they acquired resistance to Osimertinib and had a positive T790M mutation at baseline.
Sensitivity analysis
Sensitivity analysis assesses the stability and reliability of the results by omitting one study at a time. The results showed that the combined effect size and heterogeneity analysis in Review Manager 5.2 were not significantly affected by individual studies, indicating that the meta-analysis results were stable and reliable.
Publication bias
Due to the small number of studies involving PFS and TTD, the publication bias was not assessed with the Egger test. However, given the objective existence of publication bias in the retrospective study and the overall heterogeneity between them was favorable, we believe that publication bias would not affect the reliability of our results.
Discussion
In the study, we demonstrated that the presence of T790M after Osimertinib resistance was associated with longer survival benefits of Osimertinib in T790M positive NSCLC patients treated with Osimeritinb. These results suggest that the detection of T790M after treatment may serve as a potential predictor for the benefit of Osimertinib in patients with NSCLC.
Although more and more studies have confirmed the superior efficacy of Osimertinib in NSCLC paitents with EGFR driver mutations [5, 12, 20-22], the tumor response and survival outcomes after the treatment of Osimertinib are usually different in patients. The mechanisms of resistance to Osimertinib have been the subject of several clinical studies [23-25]. Previously prospective or retrospective clinical studies have compared the relationship between treatment outcomes and T790M mutation status in disease progression, but without a definitive conclusion. Our meta-analysis focused on the NSCLC patients with baseline T790M mutation who had re-tested for T790M mutations in disease progression after single-agent Osimertinib treatment. We found that the T790M mutation could not be detected in 51.6% (161/312) of the patients when they were resistant to Osimertinib. Importantly, the loss of the T790M mutation was significantly associated with poor survival. Conversely, the persistence of T790M-positive mutations in clinical progression of oral Osimertinib appeared to be a good predictor of treatment outcome.
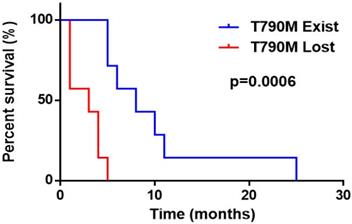
What's more, in order to verify our conclusions, we further reviewed the clinical information of 214 NSCLC patients with the EGFR T790M mutation at baseline treated with Osimertinib in our hospital. Of these patients, 38 patients were excluded from the analysis due to irregularly taking Osimertinib. 79 patients had neither disease progression nor resistance and 3 patients discontinued Osimertinib because of adverse reactions. The remaining 94 patients with the EGFR T790M mutation at baseline suffered from disease progression after Osimertinib treatment. While, tissue or blood genetic detections were re-executed on 14 of the 94 patients with complete clinical data after resistance to Osimertinib. The genetic status of these 14 patients and their clinical progression-free survival data were used to validate our conclusions. Of the 14 patients, 7 had T790M mutation persistence after Osimertinib resistance, while 7 lost the T790M mutation with resistance to Osimertinib. Analysis of the PFS data showed that the T790M persistence group had a longer median PFS than the T790M loss group (8 months vs. 3 months, respectively; pooled HR=0.098; 95% CI 0.019 to 0.513; P=0.0006) (Figure 5), which validated our main findings and conclusions.
Progression free survival of T790M persistence vs. T790M loss after Osimertinib resistance in NSCLC patients with the EGFR T790M mutation at baseline in our hospital.
In addition, the mechanism of the difference in T790M mutation status when patients acquired resistance to Osimertinib remains unclear. The most likely explanation may be the genomic heterogeneity inherent in the tumor before the treatment with Osimertinib [26, 27]. As previously reported, concomitant genomic alterations are widespread in lung cancer [28], and the T790M-positive and wild-type cell clones may co-exist in tumors at baseline levels or after acquired resistance to pre-EGFR TKI [29]. Osimertinib is effective for patients who have EGFR T790M mutations and can exert selective pressure, resulting in an increase in pre-existing T790M wild-type clones with additional EGFR-independent resistance mechanisms. This makes them more visible than T790M mutant cells in primary “T790M positive tumors”, which may further result in the loss of T790M under resistance to Osimertinib [30]. Consistent with this hypothesis, we statistically analyzed the results of second-generation sequencing of tumors that have lost T790M mutations and found multiple EGFR-independent resistance mechanisms, such as alternative signaling pathways for bypassing activation and histological transformation [31, 32]. We included a summary of 14 studies with Osimertinib resistance mechanisms and summarized 134 patients who had T790M loss after Osimertinib resistance and re-tested for blood or tissue. The results showed the occurrence of the following mutations: 18.7% (25/134) had TP53 mutations; 17.8% (24/134) had MET mutations; 11.2% (15/134) had small cell lung cancer histological pathology transformation; 5.6% (8/134) had the C797S mutation; 5.6% (8/134) had the KRAS mutation; 5.6% (8/134) had the PIK3CA mutation; 3.7% (5/134) had the BRAF mutation; 2.8% (4/134) had the CCNE1 amplification; 2.8% (4/134) had the CDK6 amplification; 2.8% (4/134) had the ratio of CCDC6-RET fusion; and the ratio of HER2 amplification occurred in 1.9% (3/134). Besides, there were many other mutations that were difficult to count one by one, but it had to be considered that the occurrence of these downstream mutations may provide a potential mechanism for Osimertinib resistance after the T790M mutation disappears. What's more, due to the lack of T790M mutations, there may be EGFR-independent resistance mechanisms, which need further study.
Previous studies have shown that the dynamic quantitative assessment of the T790M mutation load is related to the extent of response to third-generation EGFR inhibitors [13-15, 33-36]. Our results provide further evidence that the current binary assessment (presence or loss) of T790M status alone may not be a uniform biomarker for Osimertinib treatment, but the detailed analysis of tumor genomic changes before and after treatment highlight the role of the development of Osimertinib resistance. The results of this study may provide a new understanding of the mechanisms that drive early Osimertinib resistance. Overall, an understanding of the detailed genomic alterations of tumors before and after the Osimertinib treatment is not yet fully understood. More clinical data are needed to reveal the difference in T790M mutation status after Osimertinib treatment failure.
Our study has several limitations. First, for the studies were retrospective in nature, and there might have been potential selection bias. Second, the sample size of included studies was relatively small. Third, because of the limited numbers of studies and the types of publications included, publication bias was not assessed with the Egger test.
In summary, our meta-analysis indicates that persistence of the T790M mutation is associated with longer survival benefits of the use of Osimertinib in non-small cell lung cancer with a T790M mutation at baseline. Dynamic detection of T790M mutation status may help to indicate and predict disease progression in a timely manner.
Abbreviations
EGFR, epidermal growth factor receptor; EGFR- TKI, epidermal growth factor receptor-tyrosine kinase inhibitor; OS, overall survival; PFS, progression-free survival; TTD, time to discontinuation; HR, hazard ratio; NSCLC, non-small cell lung cancer; ATP, adenosine triphosphate.
Acknowledgements
This work was supported by the National Natural Sciences Foundation Key Program (81630071); CAMS Innovation Fund for Medical Sciences (CIFMS 2016-I2M-3-008); Aiyou foundation (KY201701); Ministry of Education Innovation Team development project (IRT-17R10); CAMS Key lab of translational research on lung cancer (2018PT31035).
Competing Interests
The authors have declared that no competing interest exists.
References
1. Meza R, Meernik C, Jeon J, Cote ML. Lung cancer incidence trends by gender, race and histology in the United States, 1973-2010. PLoS One. 2015;10:e0121323 1-14
2. Zhou C, Wu YL, Chen G, Feng J, Liu XQ, Wang C. et al. Erlotinib versus chemotherapy as first-line treatment for patients with advanced EGFR mutation-positive non-small-cell lung cancer (OPTIMAL, CTONG-0802): a multicentre, open-label, randomised, phase 3 study. Lancet Oncol. 2011;12:735-42
3. Kris MG, Johnson BE, Berry LD, Kwiatkowski DJ, Iafrate AJ, Wistuba II. et al. Using multiplexed assays of oncogenic drivers in lung cancers to select targeted drugs. JAMA. 2014;311:1998-2006
4. Nagano T, Tachihara M, Nishimura Y. Mechanism of Resistance to Epidermal Growth Factor Receptor-Tyrosine Kinase Inhibitors and a Potential Treatment Strategy. Cells. 2018:7 212:1-16
5. Chen P, Chen F, Lei J, Zhou B. Curative effectiveness and safety of osimertinib in the treatment for non-small-cell lung cancer: a meta-analysis of the experimental evidence. Onco Targets Ther. 2018;11:9033-47
6. Goss G, Tsai CM, Shepherd FA, Ahn MJ, Bazhenova L, Crinò L. et al. CNS response to osimertinib in patients with T790M-positive advanced NSCLC: pooled data from two phase II trials. Ann Oncol. 2018;29:687-93
7. Remon J, Caramella C, Jovelet C, Lacroix L, Lawson A, Smalley S. et al. Osimertinib benefit in EGFR-mutant NSCLC patients with T790M-mutation detected by circulating tumour DNA. Ann Oncol. 2017;28:784-90
8. Mok TS, Wu Y-L, Ahn M-J, Garassino MC, Kim HR, Ramalingam SS. et al. Osimertinib or Platinum-Pemetrexed in EGFR T790M-Positive Lung Cancer. N Engl J Med. 2017;376:629-40
9. Soria JC, Ohe Y, Vansteenkiste J, Reungwetwattana T, Chewaskulyong B, Lee KH. et al. Osimertinib in Untreated EGFR-Mutated Advanced Non-Small-Cell Lung Cancer. N Engl J Med. 2018;378:113-25
10. Nie K, Jiang H, Zhang C, Geng C, Xu X, Zhang L. et al. Mutational Profiling of Non-Small-Cell Lung Cancer Resistant to Osimertinib Using Next-Generation Sequencing in Chinese Patients. Biomed Res Int. 2018;2018:9010353 1-6
11. Rotow J, Bivona TG. Understanding and targeting resistance mechanisms in NSCLC. Nat Rev Cancer. 2017;17:637-58
12. Lin CC, Shih JY, Yu CJ, Ho CC, Liao WY, Lee JH. et al. Outcomes in patients with non-small-cell lung cancer and acquired Thr790Met mutation treated with osimertinib: a genomic study. Lancet Respir Med. 2018;6:107-16
13. Zhao S, Li X, Zhao C, Jiang T, Jia Y, Shi J. et al. Loss of T790M mutation is associated with early progression to osimertinib in Chinese patients with advanced NSCLC who are harboring EGFR T790M. Lung Cancer. 2019;128:33-9
14. Oxnard GR, Hu Y, Mileham KF, Husain H, Costa DB, Tracy P. et al. Assessment of Resistance Mechanisms and Clinical Implications in Patients With EGFR T790M-Positive Lung Cancer and Acquired Resistance to Osimertinib. JAMA Oncol. 2018;4:1527-34
15. Park CK, Cho HJ, Choi YD, Oh IJ, Kim YC. A Phase II Trial of Osimertinib in the Second-Line Treatment of Non-small Cell Lung Cancer with the EGFR T790M Mutation, Detected from Circulating Tumor DNA: LiquidLung-O-Cohort 2. Cancer Res Treat. 2019;51:777-87
16. Carlisle JW, Ramalingam SS. Role of osimertinib in the treatment of EGFR-mutation positive non-small-cell lung cancer. Future Oncol. 2019;15:805-16
17. Hong MH, Kim HR, Ahn BC, Heo SJ, Kim JH, Cho BC. Real-World Analysis of the Efficacy of Rebiopsy and EGFR Mutation Test of Tissue and Plasma Samples in Drug-Resistant Non-Small Cell Lung Cancer. Yonsei Med J. 2019;60:525-34
18. Hutton B, Salanti G, Caldwell DM, Chaimani A, Schmid CH, Cameron C. et al. The PRISMA extension statement for reporting of systematic reviews incorporating network meta-analyses of health care interventions: checklist and explanations. Ann Intern Med. 2015;162:777-84
19. Dugani SB, Ayala Melendez AP, Reka R, Hydoub YM, McCafferty SN, Murad MH. et al. Risk factors associated with premature myocardial infarction: a systematic review protocol. BMJ Open. 2019;9:e023647 1-5
20. Fang W, Huang Y, Hong S, Zhang Z, Wang M, Gan J. et al. EGFR exon 20 insertion mutations and response to osimertinib in non-small-cell lung cancer. BMC Cancer. 2019;19:595 1-9
21. Scott LJ. Osimertinib as first-line therapy in advanced NSCLC: a profile of its use. Drugs Ther Perspect. 2018;34:351-7
22. Stratmann JA, Michels S, Hornetz S, Christoph DC, Sackmann S, Spengler W. et al. Efficacy and safety analysis of the German expanded access program of osimertinib in patients with advanced, T790M-positive non-small cell lung cancer. J Cancer Res Clin Oncol. 2018;144:2457-63
23. Zhang Q, Zhang XC, Yang JJ, Yang ZF, Bai Y, Su J. et al. EGFR L792H and G796R: Two Novel Mutations Mediating Resistance to the Third-Generation EGFR Tyrosine Kinase Inhibitor Osimertinib. J Thorac Oncol. 2018;13:1415-21
24. Yang Z, Yang N, Ou Q, Xiang Y, Jiang T, Wu X. et al. Investigating Novel Resistance Mechanisms to Third-Generation EGFR Tyrosine Kinase Inhibitor Osimertinib in Non-Small Cell Lung Cancer Patients. Clin Cancer Res. 2018;24:3097-107
25. Nishino M, Suda K, Kobayashi Y, Ohara S, Fujino T, Koga T. et al. Effects of secondary EGFR mutations on resistance against upfront osimertinib in cells with EGFR-activating mutations in vitro. Lung Cancer. 2018;126:149-55
26. Hao S, Lu CH, Lin CC, Chen HY, Li L, Wang YB. et al. [The role and mechanism of 2-deoxyglucose in reversing osimertinib-acquired resistance of non-small cell lung cancer cell line]. Zhonghua Jie He He Hu Xi Za Zhi. 2019;42:198-205
27. Xing P, Han X, Wang S, Liu Y, Yang S, Hao X. et al. Co-mutational assessment of circulating tumour DNA (ctDNA) during osimertinib treatment for T790M mutant lung cancer. J Cell Mol Med. 2019;00:1-10
28. Blakely CM, Watkins TBK, Wu W, Gini B, Chabon JJ, McCoach CE. et al. Evolution and clinical impact of co-occurring genetic alterations in advanced-stage EGFR-mutant lung cancers. Nat Genet. 2017;49:1693-704
29. Piotrowska Z, Niederst MJ, Karlovich CA, Wakelee HA, Neal JW, Mino-Kenudson M. et al. Heterogeneity Underlies the Emergence of EGFRT790 Wild-Type Clones Following Treatment of T790M-Positive Cancers with a Third-Generation EGFR Inhibitor. Cancer Discov. 2015;5:713-22
30. Wu S, Shi X, Si X, Liu Y, Lu T, Zhang L. et al. EGFR T790M detection in formalin-fixed paraffin-embedded tissues of patients with lung cancer using RNA-based in situ hybridization: A preliminary feasibility study. Thorac Cancer. 2019 [Epub ahead of print]
31. Li XY, Huang LT, Wu JQ, He MF, Zhu SH, Zhan P. et al. Zebrafish Xenograft Model of Human Lung Cancer for Evaluating Osimertinib Resistance. Biomed Res Int. 2019;2019:3129748 1-11
32. Kato Y, Hosomi Y, Watanabe K, Yomota M, Kawai S, Okuma Y. et al. Impact of clinical features on the efficacy of osimertinib therapy in patients with T790M-positive non-small cell lung cancer and acquired resistance to epidermal growth factor receptor tyrosine kinase inhibitors. J Thorac Dis. 2019;11:2350-60
33. Ariyasu R, Nishikawa S, Uchibori K, Oh-Hara T, Yoshizawa T, Dotsu Y. et al. High ratio of T790M to EGFR activating mutations correlate with the osimertinib response in non-small-cell lung cancer. Lung Cancer. 2018;117:1-6
34. Yang JC, Ahn MJ, Kim DW, Ramalingam SS, Sequist LV, Su WC. et al. Osimertinib in Pretreated T790M-Positive Advanced Non-Small-Cell Lung Cancer: AURA Study Phase II Extension Component. J Clin Oncol. 2017;35:1288-96
35. Papadimitrakopoulou VA, Wu Y-L, Han J-Y, Ahn M-J, Ramalingam SS, John T. et al. LBA51Analysis of resistance mechanisms to osimertinib in patients with EGFR T790M advanced NSCLC from the AURA3 study. Ann Oncol. 2018;29(suppl 4):viii741
36. Goldberg ME, Montesion M, Young L, Suh J, Greenbowe J, Kennedy M. et al. Multiple configurations of EGFR exon 20 resistance mutations after first- and third-generation EGFR TKI treatment affect treatment options in NSCLC. PLoS One. 2018;13:e0208097 1-8
Author contact
Corresponding authors: Hua Bai, State Key Laboratory of Molecular Oncology, Department of Medical Oncology, National Cancer Center/National Clinical Research Center for Cancer/Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, 100021, China. E-mail: wangjieac.cn. Or Jie Wang, State Key Laboratory of Molecular Oncology, Department of Medical Oncology, National Cancer Center/National Clinical Research Center for Cancer/Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, 100021, China. E-mail: wangjieac.cn.

 Global reach, higher impact
Global reach, higher impact