Impact Factor ISSN: 1837-9664
J Cancer 2021; 12(15):4463-4477. doi:10.7150/jca.60008 This issue Cite
Research Paper
MST4 negatively regulates the EMT, invasion and metastasis of HCC cells by inactivating PI3K/AKT/Snail1 axis
1. Cancer Research Institute, School of Basic Medical Sciences, Southern Medical University, Guangzhou 510515, China.
2. Institute of Comparative Medicine & Laboratory Animal Center, Southern Medical University, Guangzhou 510515, China.
3. Radiotherapy Center, the First People's Hospital of Chenzhou, Xiangnan University, Chenzhou 423000, China.
4. Department of Physiology, Faculty of Basic Medical Sciences, Guilin Medical University, Guilin 541004, China.
5. State Key Laboratory of Organ Failure Research, Key Laboratory of Mental Health of the Ministry of Education, Guangdong-Hong Kong-Macao Greater Bay Area Center for Brain Science and Brain-Inspired Intelligence, Guangdong Province Key Laboratory of Psychiatric Disorders, Collaborative Innovation Center for Brain Science, Department of Neurobiology, School of Basic Medical Sciences, Southern Medical University, Guangzhou 510515, China.
6. The third people's hospital of Kunming (The Sixth Affiliated Hospital of Dali University), Kunming 650041, China.
7. Department of Gastrointestinal Oncology, The Third Affiliated Hospital of Kunming Medical University (Yunnan Cancer Hospital, Yunnan Cancer Center), Kunming 650118, China.
8. Department of Pathology, the Second Affiliated Hospital, Guilin Medical University, Guilin 541199, China.
9. Department of Oncology, The First Affiliation Hospital of Guangdong Pharmaceutical University, Guangzhou 510062, China.
*These authors contributed equally to this work.
Abstract
Background: Hepatocellular carcinoma (HCC) is one of the most common cancers worldwide and has a poor prognosis due to the high incidence of invasion and metastasis-related progression. However, the underlying mechanism remains elusive, and valuable biomarkers for predicting invasion, metastasis, and poor prognosis of HCC patients are still lacking.
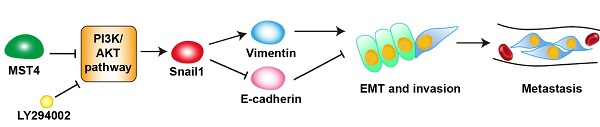
Methods: Immunohistochemistry (IHC) was performed on HCC tissues (n = 325), and the correlations between MST4 expression of the clinical HCC tissues, the clinicopathologic features, and survival were further evaluated. The effects of MST4 on HCC cell migratory and invasive properties in vitro were evaluated by Transwell and Boyden assays. The intrahepatic metastasis mouse model was established to evaluate the HCC metastasis in vivo. The PI3K inhibitor, LY294002, and a specific siRNA against Snail1 were used to investigate the roles of PI3K/AKT pathway and Snail1 in MST4-regulated EMT, migration, and invasion of HCC cells, respectively.
Results: In this study, by comprehensively analyzing our clinical data, we discovered that low MST4 expression is highly associated with the advanced progression of HCC and serves as a prognostic biomarker for HCC patients of clinical-stage III-IV. Functional studies indicate that MST4 inactivation induces epithelial-to-mesenchymal transition (EMT) of HCC cells, promotes their migratory and invasive potential in vitro, and facilitates their intrahepatic metastasis in vivo, whereas MST4 overexpression exhibits the opposite phenotypes. Mechanistically, MST4 inactivation elevates the expression and nuclear translocation of Snail1, a key EMT transcription factor (EMT-TF), through the PI3K/AKT signaling pathway, thus inducing the EMT phenotype of HCC cells, and enhancing their invasive and metastatic potential. Moreover, a negative correlation between MST4 and p-AKT, Snail1, and Ki67 and a positive correlation between MST4 and E-cadherin were determined in clinical HCC samples.
Conclusions: Our findings indicate that MST4 suppresses EMT, invasion, and metastasis of HCC cells by modulating the PI3K/AKT/Snail1 axis, suggesting that MST4 may be a potential prognostic biomarker for aggressive and metastatic HCC.
Keywords: MST4, Hepatocellular carcinoma, Metastasis, Prognosis, EMT, PI3K/AKT, Snail1

 Global reach, higher impact
Global reach, higher impact