Impact Factor
ISSN: 1837-9664
J Cancer 2021; 12(15):4722-4728. doi:10.7150/jca.59419 This issue Cite
Research Paper
Clinicopathologic factors affecting discrepancies in HER2 overexpression between core needle biopsy and surgical biopsy in breast cancer patients according to neoadjuvant treatment or not
1. Department of Surgery, Chung-Ang University College of Medicine, 224-1, Heukseok-dong, Dongjak-gu, Seoul 06973, Republic of Korea.
2. Department of Pathology, Chung-Ang University College of Medicine, 224-1, Heukseok-dong, Dongjak-gu, Seoul 06973, Republic of Korea.
3. Department of Radiology, Chung-Ang University College of Medicine, 224-1, Heukseok-dong, Dongjak-gu, Seoul 06973, Republic of Korea.
4. Department of Surgery, Kangnam Sacred Heart Hospital, Hallym University College of Medicine, 1 Shingil-ro, Youngdeungpo-ku, Seoul.
Abstract
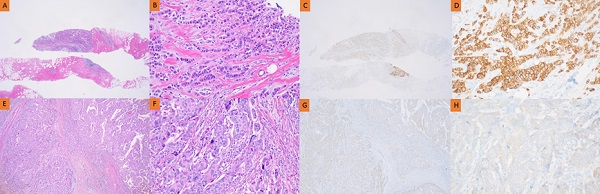
Background: Accurate determination of human epidermal growth factor receptor 2 (HER2) status on breast core needle biopsy (CNB) tissue is important for determining neoadjuvant chemotherapies (NACs) for primary breast cancer. However, HER2 discrepancies occur before and after surgery, creating difficulties in the administration of appropriate NAC. This study aimed to identify the clinical factors affecting these discrepancies.
Methods: This study was conducted on patients with primary breast cancer who underwent breast surgery from January 2012 to December 2018 at the Chung-Ang University Hospital. HER2 status was analyzed using immunohistochemistry. HER2 was graded as 0 to +3, and all 2+ cases were evaluated for gene amplification. The patients were divided into two groups based on whether or not they received chemotherapy. Patient and clinical characteristics between the two groups were compared using the χ2 test and a logistic regression model.
Results: A total of 443 patients were evaluated; 384 patients (86.7%) did not receive NAC, and 59 patients (13.3%) received NAC. The HER2 discordance rate was 12.5% in the no NAC group and 23.7% in the NAC group. Most cases showed a change in HER2 status from negative in CNB to positive in surgical biopsy (SB). Clinicopathological factors affecting HER2 change in the no NAC group were larger tumor size and higher histologic grade. Meanwhile, poor response to prior chemotherapy affected HER2 change in NAC.
Conclusion: The overall accuracy of CNB in determining HER2 status was lower in the NAC group than in the no NAC group. Some clinicopathological factors may affect HER2 changes in each group at different levels. Based on the HER2 status at the time of diagnosis, the choice of HER2-targeted therapy may vary, even if this is not true. Future research on the effects of changes in HER2 status between CNB and SB on prognosis is needed.
Keywords: HER2, breast cancer, tumor heterogeneity, neoadjuvant treatment

 Global reach, higher impact
Global reach, higher impact