3.2
Impact Factor
ISSN: 1837-9664
J Cancer 2021; 12(17):5296-5309. doi:10.7150/jca.56138 This issue Cite
Research Paper
MicroRNA-133 Targets Phosphodiesterase 1C in Drosophila and Human Oral Cancer Cells to Regulate Epithelial-Mesenchymal Transition
1. Department of Life Science in Dentistry, School of Dentistry, Pusan National University, Yangsan 50612, Korea.
2. BK21 FOUR Project, School of Dentistry, Pusan National University, Yangsan 50612, Korea.
3. Dental and Life Science Institute, Pusan National University, Yangsan 50612, Korea.
4. Department of Oral Pathology, School of Dentistry, Pusan National University, Yangsan 50612, Korea.
5. Interdisciplinary Program of Genomic Science, Pusan National University, Yangsan 50612, Korea.
6. Department of Anatomy, Department of Biomedical Informatics, School of Medicine, Pusan National University, Yangsan 50612, Korea.
Abstract
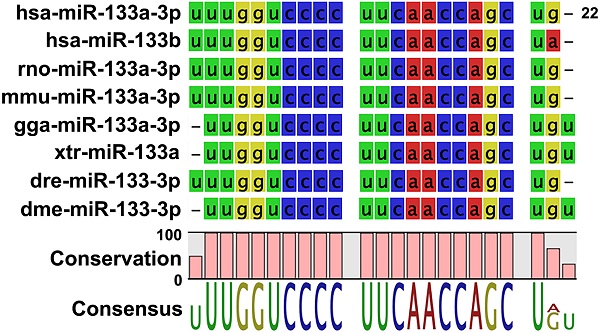
Non-coding microRNAs (miRNAs) have been proposed to play diverse roles in cancer biology, including epithelial-mesenchymal transition (EMT) crucial for cancer progression. Previous comparative studies revealed distinct expression profiles of miRNAs relevant to tumorigenesis and progression of oral cancer. With putative targets of these miRNAs mostly validated in vitro, it remains unclear whether similar miRNA-target relationships exist in vivo. In this study, we employed a hybrid approach, utilizing both Drosophila melanogaster and human oral cancer cells, to validate projected miRNA-target relationships relevant to EMT. Notably, overexpression of dme-miR-133 resulted in significant tissue growth in Drosophila larval wing discs. The RT-PCR analysis successfully validated a subset of its putative targets, including Pde1c. Subsequent experiments performed in oral cancer cells confirmed conserved targeting of human PDE1C by hsa-miR-133. Furthermore, the elevated level of miR-133 and its targeting of PDE1C was positively correlated with enhanced migrative ability of oral cancer cells treated with LPS, along with the molecular signature of a facilitated EMT process induced by LPS and TGF-β. The analysis on the RNAseq data also revealed a negative correlation between the expression level of hsa-miR-133 and the survival of oral cancer patients. Taken together, our mammal-to-Drosophila-to-mammal approach successfully validates targeting of PDE1C by miR-133 both in vivo and in vitro, underlying the promoted EMT phenotypes and potentially influencing the prognosis of oral cancer patients. This hybrid approach will further aid to widen our scope in investigation of intractable human malignancies, including oral cancer.
Keywords: microRNA-133, Drosophila melanogaster, oral cancer, PDE1C, Epithelial-Mesenchymal Transition, mammal-to-Drosophila-to-mammal approach

 Global reach, higher impact
Global reach, higher impact