Impact Factor
ISSN: 1837-9664
J Cancer 2021; 12(18):5486-5493. doi:10.7150/jca.45262 This issue Cite
Research Paper
PHLDA1 Modulates the Endoplasmic Reticulum Stress Response and is required for Resistance to Oxidative Stress-induced Cell Death in Human Ovarian Cancer Cells
1. Department of Obstetrics and Gynecology, The Third Affiliated Hospital of Chongqing Medical University, Chongqing 401120, PR China.
2. Department of Urology, Daping Hospital, Army Medical University, Chongqing 400042, PR China.
3. Department of Obstetrics and Gynecology, Daping Hospital, Army Medical University, Chongqing 400042, PR China.
4. The Ohio State University Comprehensive Cancer Center, Columbus, OH 43210, USA.
5. Cancer Research Institute of Jilin University, The First Hospital of Jilin University, Changchun 130021, PR China.
6. Department of Hematology and Hematopoietic Cell Transplantation, Comprehensive Cancer Center, City of Hope National Medical Center, Duarte, California 91010, USA.
* These authors equally contributed.
Abstract
Objective: Pleckstrin homology-like domain family A member 1 (PHLDA1) has been implicated in the regulation of apoptosis in a variety of normal cell types and cancers. However, its precise pathophysiological functions remain unclear. Here, we examined the expression of PHLDA1 in human ovarian cancer (OvCa), the most lethal gynecologic malignancy, and investigated its functions in vitro.
Materials and Methods: The expression of PHLDA1 was detected by reverse-transcription quantitative PCR (RT-qPCR), immunohistochemical analysis, or western blotting, silencing of PHLDA was achieved by shRNA, cell proliferation was detected by MTT assay, apoptosis was detected by flow cytometric analysis, PHLDA1 transcriptional activity was detected by dual luciferase reporter assay.
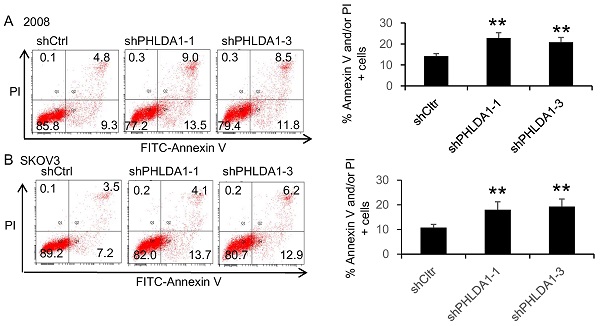
Results: PHLDA1 mRNA levels were significantly higher in serous OvCa specimens compared with normal ovarian tissue, confirmed by immunohistochemical staining of PHLDA1 protein, which also indicated the expression was predominantly cytoplasmic. Bioinformatics analysis of publicly available datasets indicated that PHLDA1 expression in clinical specimens was significantly associated with disease stage, progression-free survival, and overall survival. In human OvCa cell lines, shRNA-mediated silencing of PHLDA1 expression enhanced apoptosis after exposure to oxidative stress- and endoplasmic reticulum stress-inducing agents. PHLDA1 silencing increased not the expression of anti-apoptotic or autophagy-related proteins, but the expression of ER stress response-associated proteins.
Conclusion: PHLDA1 modulates the susceptibility of human OvCa cells to apoptosis via the endoplasmic reticulum stress response pathway.
Keywords: PHLDA1, endoplasmic reticulum stress, oxidative stress, apoptosis, ovarian cancer

 Global reach, higher impact
Global reach, higher impact