Impact Factor
ISSN: 1837-9664
J Cancer 2021; 12(18):5593-5604. doi:10.7150/jca.56709 This issue Cite
Research Paper
The Differential Antitumor Activity of 5-Aza-2'-deoxycytidine in Prostate Cancer DU145, 22RV1, and LNCaP Cells
1. Aoyang Institute of Cancer, Affiliated Aoyang Hospital of Jiangsu University, 279 Jingang Blvd., Zhangjiagang, Suzhou, 215600, China
2. Dept of Urology, the Affiliated Aoyang Hospital of Jiangsu University, 279 Jingang Blvd., Zhangjiagang, Suzhou, 215600, China
3. Perinatology Research Branch, Eunice Kennedy Shriver National Institute of Child Health and Human Development, National Institutes of Health, Detroit 48201, MI, USA
4. The Laboratory of Clinical Genomics, Hefei KingMed Diagnostics Ltd., 2800 Chuangxin Blvd., Building H4, Hefei 230088, China
5. National Center for Gene Testing Technology Application & Demonstration(Hefei), 2800 Chuangxin Blvd., Building H4, Hefei 230088, China
*Equal Contribution
#Current address: Zhengzhou University, 100 Kexue Blvd., Zhengzhou, 450001, China
Abstract
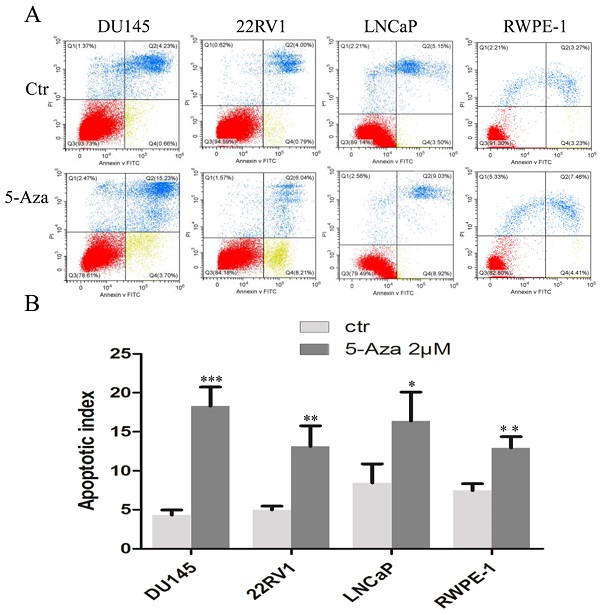
DNA methylation is a DNA methyltransferase-mediated epigenetic modification affecting gene expression. This process is involved in the initiation and development of malignant disease. 5‐Aza‐2′‐deoxycytidine (5‐Aza), a classic DNA methyltransferase inhibitor, possesses antitumor proliferation activity. However, whether 5-Aza induces cytotoxicity in solid tumors warrants further investigated. In this study, human prostate cancer (CaP) cells were treated with 5-Aza and subjected to cell viability and cytotoxicity analysis. Reverse transcription-polymerase chain reaction and methylation-specific polymerase chain reaction assay were utilized to test the gene expression and methylation status of the p53 and p21 gene promoters. The results showed that 5-Aza differentially inhibited spontaneous proliferation, arrested the cell cycle at S phase in DU145, at G1 phase in 22RV1 and LNCaP cells, and G2 phase in normal RWPE-1 cells, as well as induced the expression of phospho-H2A.X and tumor suppressive mammary serine protease inhibitor (maspin) in all three types of CaP cells. 5-Aza also increased p53 and p21 transcription through promoter demethylation, and decreased the expression of oncogene c-Myc in 22RV1 and LNCaP cells. Western blotting analysis showed that the poly (ADP-ribose) polymerase cleavage was detected in DU145 and 22RV1 cells. Moreover, there were no significant changes in p53, p21 and c-Myc expression in DU145 cells following treatment with 5-Aza. Thus, in responsible for its apoptotic induction and DNA damage, the mechanism of the antitumor activities of 5-Aza may involve in an increase of tumor suppressive maspin, upregulation of wild type p53-mediated p21 expression and a decrease of oncogene c-Myc level in 22RV1 and LNCaP cells, and enhancing the tumor suppressive maspin expression in DU145 cells. These results enriched our understanding of the multifaceted antitumor activity of 5-Aza, and provided the expression basis of biomarkers for its possible clinical application in prostate cancer.
Keywords: 5-Aza-2′-deoxycytidine, prostate cancer cell, antitumor activity, p53, p21

 Global reach, higher impact
Global reach, higher impact