Impact Factor ISSN: 1837-9664
J Cancer 2021; 12(18):5622-5632. doi:10.7150/jca.58824 This issue Cite
Research Paper
NEAT1/miR-101-dependent Up-regulation of DNA-PKcs Enhances Malignant Behaviors of Pancreatic Ductal Adenocarcinoma Cells
1. Hepatobiliary and Pancreatic Surgery, Affiliated Hospital of Jiangnan University, 585 Xingyuan Rd, Liangxi District, Wuxi, 214041, China
2. School of Medicine, Jiangnan University, Wuxi 214122, China
3. Hepatobiliary and Pancreatic Surgery, The Third Hospital Affiliated to Nantong University, Wuxi 214041, China
4. Medical School, Nantong University, Nantong 226001, China
5. Wuxi Institute of Hepatobiliary Surgery, Wuxi 214041, China
6. Department of Gastroenterology, Affiliated Hospital of Jiangnan University, Wuxi 214041, China
*These authors contribute equally to the manuscript
Abstract
Background: Although we previously revealed that DNA-dependent protein kinase catalytic subunit (DNA-PKcs) is overexpressed in pancreatic ductal adenocarcinoma (PDAC) and important for gemcitabine resistance, the role of DNA-PKcs in the progression and metastasis of PDAC remain unclear. To date, the upstream signaling events stimulating DNA-PKcs overexpression in PDAC are still not well characterized.
Methods: Expression of DNA-PKcs was measured by western blot. The levels of miRNA-101 and lncRNA nuclear paraspeckle assembly transcript 1 (NEAT1) were detected by real-time PCR. Cell viability was determined by CCK-8. Cell migration and cell invasion were measured by transwell assay. The regulatory relationship between NEAT1 and miR-101 was determined by a luciferase assay.
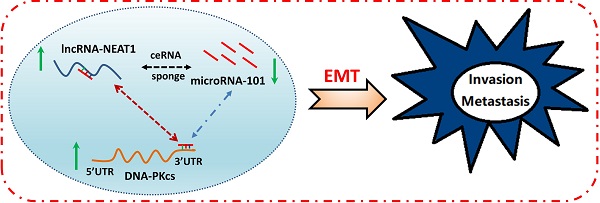
Results: DNA-PKcs expression was significantly elevated in human PDAC tissues and cells. DNA-PKcs overexpression was correlated with TNM stage and lymph node metastasis. Higher expression of DNA-PKcs was closely correlated with patients of worse overall survival (OS). DNA-PKcs knockdown suppresses malignant behaviors of PDAC cells. Further study showed that miRNA-101 level was decreased in PDAC tissues and cells, which could be responsible for DNA-PKcs overexpression and DNA-PKcs mediated oncogenic actions in PDAC cells. Moreover, NEAT1 functions as an oncogene influencing cell proliferation, migration and invasion in part by serving as a competing endogenous RNA (ceRNAs) modulating miR-101 expression, leading to up-regulation of DNA-PKcs.
Conclusion: These findings suggest that NEAT1/miR-101-dependent up-regulation of DNA-PKcs promotes the malignant behaviors of PDAC cells. The NEAT1/miR-101/DNA-PKcs axis may serve as a viable prognostic marker and therapeutic target for PDAC.
Keywords: DNA-PKcs, NEAT1, miR-101, pancreatic ductal adenocarcinoma

 Global reach, higher impact
Global reach, higher impact