3.2
Impact Factor
ISSN: 1837-9664
J Cancer 2021; 12(24):7334-7348. doi:10.7150/jca.61031 This issue Cite
Research Paper
Targeting ADAR1 suppresses progression and peritoneal metastasis of gastric cancer through Wnt / β-catenin pathway
1. General Surgery Department, The First Affiliated Hospital of Xi'an Jiaotong University, Xi'an 710061, Shaanxi, China.
2. Department of Gastrointestinal Surgery, People's Hospital of Ningxia Hui Autonomous Region, Yinchuan 750002, Ningxia, China.
3. Department of Pathology, School of Basic Medicine, Ningxia Medical University, Yinchuan 750004, Ningxia, China.
4. Department of Pathology, North China University of Science and Technology Affiliated Hospital, Tangshan 063000, Hebei, China.
5. Department of Pathology, People's Hospital of Ningxia Hui Autonomous Region, Yinchuan 750002, Ningxia, China.
6. Digestive Endoscopy Center, People's Hospital of Ningxia Hui Autonomous Region, Yinchuan 750002, Ningxia, China.
7. Department of Surgery, School of Clinical Medicine, Ningxia Medical University, Yinchuan 750004, Ningxia, China.
* Equal contributors
Abstract
Objective: Peritoneal metastasis frequently occurs in advanced gastric cancer, which is typically not eligible for radical surgery. Here, this study observed the function and regulatory mechanism of ADAR1 in peritoneal metastasis of gastric cancer.
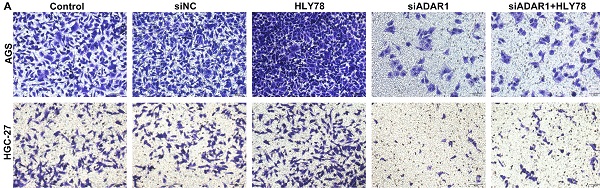
Methods: ADAR1, CALR and β-catenin proteins were detected in normal mucosa, primary gastric cancer, metastatic lymph node and metastatic omentum tissues by immunohistochemistry, western blot, and immunofluorescence. After silencing ADAR1 by siADAR1, the effect and mechanism of ADAR1 on gastric cancer metastasis were observed in nude mouse models of gastric cancer with peritoneal metastasis as well as HGC-27 and AGS gastric cancer cells.
Result: Our results showed that ADAR1 was significantly up-regulated in gastric cancer, metastatic lymph node and metastatic omentum tissues. Its up-regulation was significantly correlated to lymph node metastasis and peritoneal metastasis. Silencing ADAR1 significantly reduced the volume of peritoneal metastatic tumors and weakened oncogene CALR expression, Wnt / β-catenin pathway and epithelial-mesenchymal transition (EMT) process in vivo. Furthermore, ADAR1 knockdown distinctly suppressed cell viability, colony formation and migration of HGC-27 and AGS cells and ameliorated the effects of Wnt pathway activator on tumor progression. The similar findings were investigated when treated with ADAR1 inhibitor 8-Azaadenosine.
Conclusion: Collectively, this study identified a novel oncogenic function of ADAR1 in peritoneal metastasis of gastric cancer via Wnt / β-catenin pathway. Hence, ADAR1 could be a novel marker and therapeutic target against gastric cancer metastasis.
Keywords: ADAR1, gastric cancer, peritoneal metastasis, Wnt / β-catenin pathway, CALR

 Global reach, higher impact
Global reach, higher impact