Impact Factor
ISSN: 1837-9664
J Cancer 2021; 12(24):7390-7398. doi:10.7150/jca.63003 This issue Cite
Research Paper
PD-L1 Expression in Chinese Patients with Advanced Non-Small Cell Lung Cancer (NSCLC): A Multi-Center Retrospective Observational Study
1. Key Laboratory of Carcinogenesis and Translational Research (Ministry of Education), Department of Pathology, Peking University Cancer Hospital & Institute, Beijing, China
2. Department of Pathology, West China Hospital, Sichuan University, Chengdu, Sichuan, China
3. Department of Pathology, Fudan University Shanghai Cancer Center, Shanghai, China
4. Department of Pathology, Sun Yat-sen University Cancer Center, Guangzhou, China
5. Zhongshan Hospital of Fudan University, Shanghai, China
6. Department of Pathology, Shanghai Pulmonary Hospital, Tongji University School of Medicine, Shanghai, China
7. Zhejiang Cancer Hospital, Hangzhou, Zhejiang, China
8. Nanjing Drum Tower Hospital, Nanjing, Jiangsu, China
9. Department of Pathology, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, Hubei, China
10. The first hospital of Jilin University - The Eastern Division, Changchun, Jilin, China
11. Medical Affairs Department, MSD China, Shanghai, China
* Authors contributed equally
Abstract
Objective: This study aimed to investigate the prevalence of tumor programmed death-ligand 1 (PD-L1) expression in Chinese patients with advanced Non-Small Cell Lung Cancer (NSCLC).
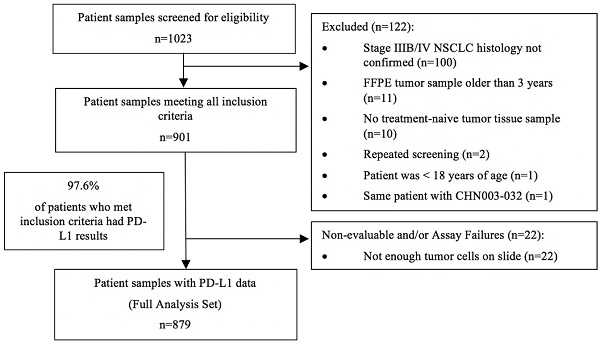
Methods: Tumor tissues with histologically confirmed stage IIIB/IV NSCLC were retrospectively obtained from 10 centers in China. PD-L1 expression was determined using the PD-L1 IHC 22C3 pharmDx kit (Agilent, Santa Clara, CA, USA) and the samples were repetitively assayed with the PD-L1 IHC 22C3 Ab concentrate (Agilent, Santa Clara, CA, USA).
Results: Out of 901 patients who met the inclusion criteria, 879 (97.6%) had evaluable PD-L1 data. The number of patients with a PD-L1 tumor proportion score (TPS) < 1%, 1-49%, and ≥ 50% (corresponding to PD-L1 non-expression, low expression, and high expression) was 424 (48.2%), 266 (30.3%), and 189 (21.5%), respectively. PD-L1 expression was more likely to be found in patients younger than 75 years, men, current or former smokers, those with good performance status (PS) scores, and those with a wild-type epidermal growth factor receptor (EGFR). PD-L1 TPS ≥ 50% and ≥ 1% were respectively 28.0% and 50.2% among patients negative for both EGFR mutation and anaplastic lymphoma kinase (ALK) rearrangement. PD-L1 expression determined using the 22C3 antibody concentrate and pharmDx kit had comparable results.
Conclusions: The prevalence of PD‑L1 expression in advanced NSCLC was consistent with that reported in the global EXPRESS study. Age, gender, smoking history, PS scores, and EGFR/ALK mutation status affected PD-L1 expression. The 22C3 antibody concentrate appears to be an alternative reagent for the PD-L1 assay.
Keywords: non-small cell lung cancer, programmed death-ligand 1, immunohistochemistry, 22C3 antibody, driver mutations

 Global reach, higher impact
Global reach, higher impact