Impact Factor
ISSN: 1837-9664
J Cancer 2022; 13(1):112-123. doi:10.7150/jca.62630 This issue Cite
Research Paper
Long non-coding RNA NNT-AS1 positively regulates NPM1 expression to affect the proliferation of estrogen-mediated endometrial carcinoma by interacting
1. Department of Gynecology, The First Affiliated Hospital, College of Medicine, Zhejiang University, Hangzhou, Zhejiang, China.
2. Department of Obstetrics and Gynecology, the Fourth Affiliated Hospital, College of Medicine, Zhejiang University, Hangzhou, Zhejiang, China.
3. Department of Gynecology, Yiwu Central Hospital, Yiwu, Zhejiang, China.
4. Department of Pathology, The First Affiliated Hospital, College of Medicine, Zhejiang University, Hangzhou, Zhejiang, China.
Abstract
Objective: This study aims to investigate the mechanism of long non-coding RNA NNT-AS1 in the proliferation of estrogen-mediated endometrial carcinoma (EC).
Materials and methods: NNT-AS1, miR-30c, and Nucleophosmin 1 (NPM1) expressions were measured by quantitative real-time PCR and Western blotting. Cell Counting Kit-8 assay and 5-Ethynyl-2'-deoxyuridine (EdU) assay were used to detect the viability and proliferation of Ishikawa and HEC-1-A cells, respectively. RNA immunoprecipitation assay was used to confirm the interaction between NNT-AS1 and miR-30c. Luciferase reporter assay was performed to confirm the interaction between miR-30c and NPM1.
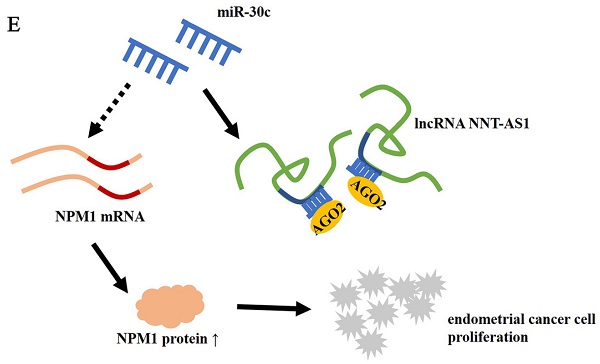
Results: NNT-AS1 and NPM1 expressions in EC tissues and cell lines were higher than in benign endometrium and normal endometrial epithelial cells (EECs). miR-30c expression in EC tissues and cell lines was lower than in benign endometrium and normal EECs. NNT-AS1 interacted with miR-30c, and miR-30c negatively regulated NPM1 expression. Overexpression of NNT-AS1 increased NPM1 expression in EC cells, while overexpression of miR-30c reversed the effect. NNT-AS1 interference inhibited the mRNA level of NPM1, while the miR-30c inhibitor reversed the result. Estradiol (E2) promoted the proliferation of EC cells, small interfering RNA (siRNA) against NNT-AS1 inhibited EC cell proliferation, miR-30c inhibitor promoted cell proliferation, and NPM1 siRNA inhibited cell proliferation. E2 increased tumor volume, and NNT-AS1 interference reduced tumor volume in vivo.
Conclusion: NNT-AS1 promoted the proliferation of estrogen-mediated EC by regulating miR-30c/NPM1.
Keywords: NNT-AS1, miR-30c, NPM1, endometrial carcinoma, estrogen

 Global reach, higher impact
Global reach, higher impact