Impact Factor ISSN: 1837-9664
J Cancer 2022; 13(8):2594-2606. doi:10.7150/jca.72522 This issue Cite
Research Paper
Anticancer Efficacy and Mechanisms of a Dual Targeting of Norepinephrine Transporter and Thyrointegrin αvβ3 Antagonist in Neuroblastoma
1. The Pharmaceutical Research Institute, Albany College of Pharmacy and Health Sciences, Rensselaer, New York 12144, United States.
2. Institute of Engineering in Medicine, University of California San Diego, San Diego, CA.
Abstract
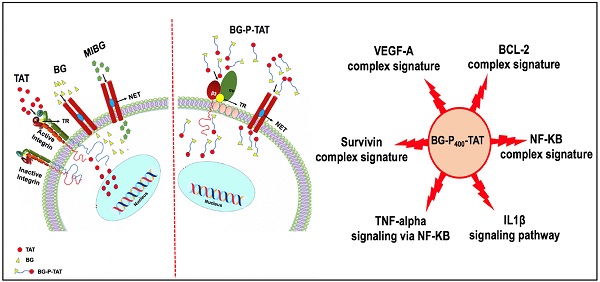
Background: In neuroendocrine tumors, the norepinephrine transporter (NET) is very active and has been exploited for diagnostic imaging purposes and/or therapy with localized radiotherapy. Integrin αvβ3 is generously expressed by and/or activated on cancer cells, but not by nonmalignant cells.
Purpose: In the present investigation, the anticancer efficacy of the dual targeting of norepinephrine transporter (NET), benzylguanidine (BG), and thyrointegrin αvβ3 receptors antagonist triazole tetraiodothyroacetic acid (TAT) conjugated via the non-cleavable linker polyethylene glycol (P, PEG400) in the treatment of human neuroblastoma was evaluated.
Experimental approach: The synthesized dual targeting compound, a novel new chemical entity named BG-P400-TAT, has purity > 98% and was formulated and tested in neuroblastoma models using neuroblastoma cell lines (SK-N-FI, SMS-KCN and SMS-KANR) implanted in SCID and NSG mice models.
Key Results: BG-P400-TAT demonstrated significant (**P<0.01, ***P< 0.001) suppression of neuroblastoma tumor progression, growth, and viability in both mice models implanted with the neuroblastoma. The pharmacokinetic and biodistribution profile of BG-P400-TAT showed a significant increase in BG-P400-TAT levels in plasma and xenografts of NSG compared to SCID mice. Further our RNAseq genome-wide expression profiling experiments in neuroblastoma cell line SKNAS results showed that BG-P400-TAT treatment altered the signal transduction pathways, intracellular multiprotein complexes and Independent GSEA.
Conclusion & Implications: BG-P400-TAT represents a potential lead candidate for the treatment of neuroblastoma and other neuroendocrine tumors.

 Global reach, higher impact
Global reach, higher impact