Impact Factor ISSN: 1837-9664
J Cancer 2022; 13(9):2884-2892. doi:10.7150/jca.71992 This issue Cite
Research Paper
From Immunosuppression to Immunomodulation - Turning Cold Tumours into Hot
1. Department of Pharmaceutical and Pharmacological Sciences, University of Padova, Via F. Marzolo 5, 35131 Padova, Italy
2. Department of Virology, National Institute of Public Health NIH —National Research Institute, Chocimska 24, 00-791 Warsaw, Poland
3. Centre for Advanced Materials and Technologies, Warsaw University of Technology, Poleczki 19, Warsaw 02-822, Poland
Abstract
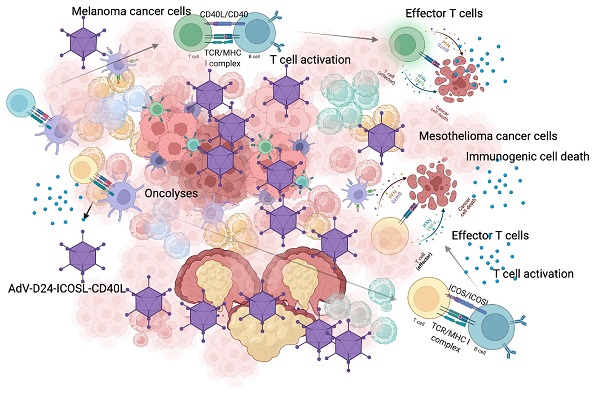
Cancer cells employ various mechanisms to evade and suppress anti-cancer immune responses generating a “cold” immunosuppressive tumour microenvironment. Oncolytic viruses are a promising tool to convert tumour immunosuppression to immunomodulation and improve the efficacy of cancer treatment. Emerging preclinical and clinical findings confirm that oncolytic viruses act in a multimodal scheme, triggering lyses, immunogenic cell death and finally inducing anti-cancer immune responses. In this paper, we tested the local administration of a novel oncolytic adenovirus AdV-D24-ICOSL-CD40L expressing co-stimulatory molecules ICOSL and CD40L to induce the production of tumour infiltrating lymphocytes to the site of injection. Subsequently, in immunocompetent mouse models, we studied possible correlation between tumour infiltrates and anti-cancer efficacy. Described results showed that the delivery of oncolytic viruses encoding immunomodulatory transgenes in combination with anti-PD1 resulted in synergistic inhibition of both melanoma and mesothelioma tumours. Importantly anti-cancer effect positively correlated with cytotoxic CD8+ tumour-infiltrating lymphocytes exerting a central role in the tumour volume control thus generating beneficial outcomes that will undoubtedly provide new insights into possible future treatment strategies to combat cancer. Altogether our findings highlight the importance of oncolytic vectors able to modulate anti-cancer immune responses that can correlate with efficacy in solid malignancies.
Keywords: oncolytic virus, TILs, immunotherapy, mesothelioma, melanoma, drug delivery

 Global reach, higher impact
Global reach, higher impact