3.2
Impact Factor
ISSN: 1837-9664
J Cancer 2023; 14(1):140-151. doi:10.7150/jca.77965 This issue Cite
Review
Pyroptosis Provides New Strategies for the Treatment of Cancer
1. Department of Hepatobiliary Surgery, The Fourth Hospital of Hebei Medical University, Shijiazhuang, China.
2. Emergency Department, The Fourth Hospital of Hebei Medical University, Shijiazhuang, China.
3. Department of Clinical Laboratory, The Fourth Hospital of Hebei Medical University, Shijiazhuang, China.
4. Department of Pancreatic and Hepatobiliary Surgery, Fudan University Shanghai Cancer Center, Shanghai, China.
5. Pancreatic Cancer Institute, Fudan University, Shanghai, China.
*These authors have contributed equally to this work and share first authorship.
Abstract
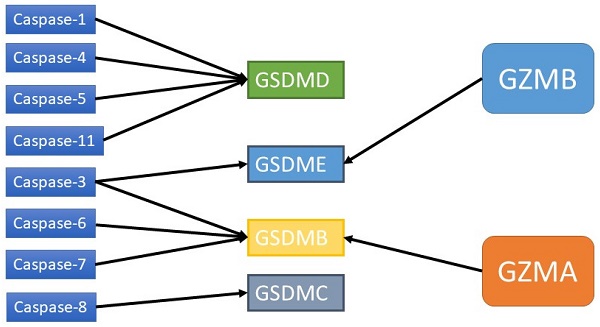
Cancer is an important cause of death worldwide. The main types of cancer treatment are still surgery, chemotherapy and radiotherapy, and immunotherapy is becoming an important cancer treatment. Pyroptosis is a type of programmed cell death that accompanies an inflammatory response. This paper reviews the recent research progress in pyroptosis in tumors. Pyroptosis has been observed since 1986 and until recently has been recognized as programmed cell death mediated by GSDM family proteins. The molecular pathway of pyroptosis depends on the inflammasome-mediated caspase-1/GSDMD pathway, which is the canonical pathway, and the caspase-4/5/11/GSDMD pathway, which is the noncanonical pathway. Other pathways include caspase3/GSDME. Pyroptosis is a double-edged sword that is closely related to the tumor immune microenvironment. On the one hand, pyroptosis produces a chronic inflammatory environment, promotes the transition of normal cells to tumor cells, helps tumor cells achieve immune escape, and promotes tumor growth and metastasis. On the other hand, some tumor cell treatments can induce pyroptosis, which is a nonapoptotic form of cell death. Additionally, pyroptosis releases inflammatory molecules that promote lymphocyte recruitment and enhance the immune system's ability to kill tumor cells. With the advent of immunotherapy, pyroptosis has been shown to enhance the antitumor efficacy of immune checkpoint inhibitors. Some antineoplastic agents, such as chemotherapeutic agents, can also exert antineoplastic effects through the pyroptosis pathway. Pyroptosis, which is a programmed cell death mode, has been the focus of research in recent years, and the relationship between pyroptosis, tumors and tumor immunity has attracted attention, but there are still some questions to be answered regarding the specific mechanism. Further study of pyroptosis would aid in developing new antitumor therapies and has great clinical prospects.
Keywords: pyroptosis, cancer, tumor immunity, gasdermin, tumor treatment

 Global reach, higher impact
Global reach, higher impact