Impact Factor ISSN: 1837-9664
J Cancer 2023; 14(4):573-590. doi:10.7150/jca.82291 This issue Cite
Editorial Commentary
The most likely but largely ignored triggering factor for breast (or all) cancer invasion
1. Department of Pathology, Hackensack Meridian School of Medicine, Nutley, NJ, USA
2. LumaBridge, San Antonio, TX, USA
3. Queen Elizabeth Hospital, Department of Clinical Oncology, Hong Kong, China
4. Division of Genomic Medicine, Department of Medicine, and of Microbiology, Immunology & Tropical Medicine, George Washington University Medical Center, Washington DC, USA
5. Department of Obstetrics and Gynecology, Changhua Christian Hospital, Changhua, Taiwan
6. Department of Pathology, Chinese PLA General Hospital 7 th Medical Center, Beijing, China
7. IIND Department of Gynecology, Lublin Medical University, Lublin, Poland
8. Pulmonary-Oncology Department, "Theageneio" Cancer Hospital, Thessaloniki, Greece
9. Laboratory of Biological Effects of Non-Ionizing Radiation, Institute of Cell Biophysics, Russian Academy of Sciences, Russian Federation
10. Department of Pathology, Hunan Normal University School of Medicine, Changsha, Hunan, China
11. Department of Genetics and Genomics Sciences, Institute for Genomics and Multiscale Biology, Icahn School of Medicine at Mount Sinai, New York, NY, USA
12. Laboratory of Mammalian Cell Biology and Development, The Rockefeller University, New York, NY, USA
13. Department of Sema4 Health Informatics, Stamford, CT, USA
14. Department of Medical Oncology, Peking University Cancer Hospital and Institute, China
15. Department of Gastrointestinal Surgery, The Second Affiliated Hospital of Nanchang University and National Engineering Research Center for Bioengineering Drugs and the Technologies, Institute of Translational Medicine, Nanchang University, Nanchang, China
Abstract
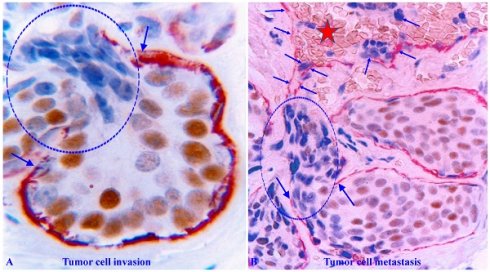
Breast cancer development and progression are believed to be a sequential process, from normal to hyperplastic, to in situ, and to invasive and metastatic stages. Given that over 90% of cancer deaths are caused by invasive and metastatic lesions, countless factors and multiple theories have been proposed as the triggering factor for the cascade of actions of cancer invasion. However, those factors and theories are largely based on the studies of cell lines or animal models. In addition, corresponding interventions based on these factors and theories have failed to reduce the incidence rate of invasive and metastatic lesions, suggesting that previous efforts may have failed to arm at the right target. Considering these facts and observations, we are proposing “A focal aberrant degeneration in the myoepithelial cell layer (MECL) as the most likely triggering factor for breast cancer invasion”. Our hypothesis is based on our recent studies of breast and multiple other cancers. Our commentary provides the rationale, morphologic, immunohistochemical, and molecular data to support our hypotheses. As all epithelium-derived cancers share a very similar architecture, our hypothesis is likely to be applicable to invasion of all cancer types. We believe that human tissue-derived data may provide a more realistic roadmap to guide the clinic practice.
Keywords: Breast myoepithelial cell layer, Tumor capsule, Tumor invasion, Cell interactions.

 Global reach, higher impact
Global reach, higher impact