Impact Factor
ISSN: 1837-9664
J Cancer 2023; 14(6):935-942. doi:10.7150/jca.81384 This issue Cite
Research Paper
High levels of baseline serum IL-10 are associated with reduced clinical benefit from first-line immune checkpoint inhibitor therapy in advanced renal cell carcinoma
1. CHA University School of Medicine, Seongnam, Korea
2. Medical Oncology, Department of Internal Medicine, CHA Bundang Medical Center, CHA University School of Medicine, Seongnam, Korea
* These authors equally contributed to this work.
Abstract
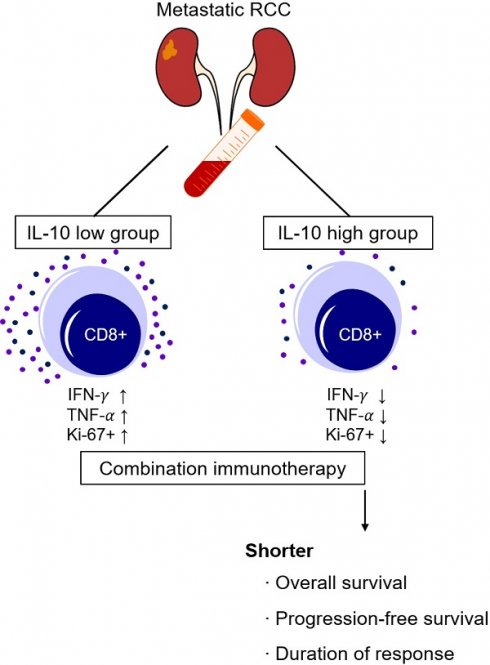
Immune checkpoint inhibitor (ICI) became a standard treatment for advanced renal cell carcinoma (RCC). However, clinically valid biomarkers of therapeutic outcome are lacking. We investigated the role of interleukin-10 (IL-10) as a predictive biomarker for first-line ICI therapy in patients with advanced RCC. Baseline serum samples were prospectively collected and analyzed using a cytometric bead assay. Patients were divided into two groups according to their serum IL-10 levels using maximally selected rank statistics. A fraction (13.0%) of patients had high levels of serum IL-10 at baseline. High serum IL-10 levels (> 4.3 ng/mL) were associated with a significantly shorter progression-free (median: 5.2 months vs. not reached, P = 0.007) and overall survival (median: 13.9 months vs. not reached, P < 0.001). Multivariate Cox regression analysis confirmed the independent association between high serum IL-10 levels and poor survival outcomes. Effector cytokine production and the proliferative response of CD8+ T cells were significantly lower in patients with high serum IL-10 levels, who also had a shorter duration of response to first-line ICI therapy (4.6 months vs. not reached, P < 0.001). In conclusion, elevated serum IL-10 levels at baseline were associated with reduced clinical benefit from first-line ICI therapy in patients with advanced RCC.
Keywords: biomarkers, immune checkpoint inhibitor, interleukin-10, advanced renal cell carcinoma, survival

 Global reach, higher impact
Global reach, higher impact