Impact Factor
ISSN: 1837-9664
J Cancer 2023; 14(10):1913-1919. doi:10.7150/jca.84261 This issue Cite
Research Paper
Role of C-Reactive Protein in Predicting the Severity and Response of Immune-Mediated Diarrhea and Colitis in Patients with Cancer
1. Department of Internal Medicine, Baylor College of Medicine, Houston, TX, USA.
2. Department of Gastroenterology, Hepatology and Nutrition, The University of Texas MD Anderson Cancer Center, Houston, TX, USA.
3. Department of Internal Medicine, The University of Texas Health Science Center, Houston, TX, USA.
4. Gastroenterology, Hepatology and Nutrition Service, Department of Medicine, Memorial Sloan Kettering Cancer Center, New York, NY, USA.
5. Department of Hematology and Medical Oncology, Cleveland Clinic, Cleveland, OH, USA.
6. Center for Inflammatory Bowel Disease, Cleveland Clinic, Cleveland, OH, USA.
7. Department of Medicine, Division of Oncology, University of Washington, Clinical Research Division, Fred Hutchinson Cancer Center, Seattle, WA, USA.
8. Centre Hospitalier Universitaire Vaudois, Department of Medicine, Service of Immunology and Allergy, Lausanne, Switzerland.
9. Gastroenterology Department, Université Paris Saclay 11, Le Kremlin-Bicêtre, France.
*Co-first author
Abstract
Background: Immune-mediated diarrhea and colitis (IMDC) frequently develop after treatment with immune checkpoint inhibitors. C-reactive protein (CRP) is a serum inflammatory biomarker used to stratify and monitor disease severity in many inflammatory conditions. However, CRP level is not specific and is widely influenced by various factors non-specific to bowel inflammation. We aimed to study the utility of CRP as a predictor of disease severity and therapy response in IMDC.
Methods: We performed a retrospective analysis of patients diagnosed with IMDC who had CRP measured at IMDC onset and after treatment with selective immunosuppressive therapy (SIT: infliximab and vedolizumab), between 01/2016 and 02/2022 at MD Anderson Cancer Center. Patient demographics, clinical characteristics, and IMDC data were collected and analyzed.
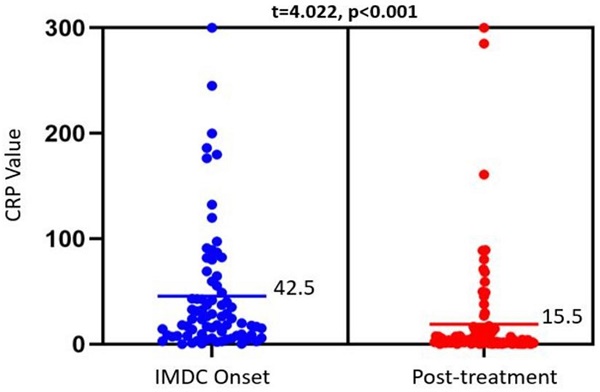
Results: Our sample of 128 patients had a median age of 67 years; most were white (89.8%); and male (65.6%). Prior to development of IMDC, 15 (11.7%) were initially treated with anti-CTLA-4, 42 (32.8%) with anti-PD-1 or PD-L1, and 71 (55.5%) with a combination of both. We found higher CRP level was associated with higher CTCAE grade of clinical symptoms such as diarrhea (p=0.015), colitis (p=0.013), and endoscopic findings (p=0.016). While CRP levels decreased after IMDC treatment, there was no significant association between CRP levels with clinical remission, endoscopic remission or histologic remission. There also was no significant correlation between CRP level and recurrence of IMDC, or with fecal calprotectin levels.
Conclusion: CRP level may be useful to assess initial severity of IMDC, including grade of diarrhea and colitis and degree of endoscopic inflammation. However, CRP is not a robust surrogate biomarker for assessing treatment response or disease recurrence. Despite the reduction of CRP levels observed following IMDC treatment, this finding might be nonspecific and potentially confounded by concurrent clinical factors, such as underlying malignancy, other inflammatory processes, and systemic anti-cancer therapy. Further studies of the role of CRP are warranted in patients with cancer and IMDC.
Keywords: immunotherapy, immune-mediated, diarrhea, colitis, C-reactive protein, fecal calprotectin

 Global reach, higher impact
Global reach, higher impact