Impact Factor ISSN: 1837-9664
J Cancer 2025; 16(6):1934-1943. doi:10.7150/jca.107117 This issue Cite
Review
Sleep health: an unappreciated key player in colorectal cancer
First Affiliated Hospital of Dalian Medical University, Dalian, China.
#: Jiahua Liu, Qihang Yuan, and Yao Zhang contributed to this study equally.
Received 2024-11-17; Accepted 2025-2-16; Published 2025-3-3
Abstract
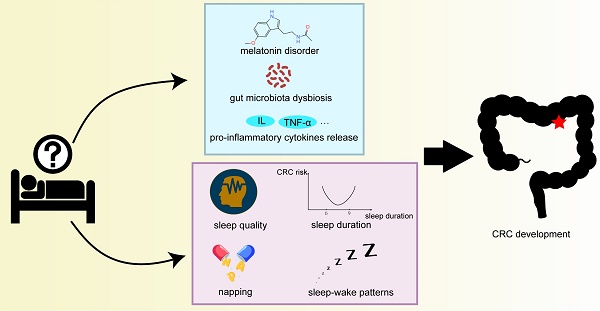
Colorectal cancer (CRC) poses a significant threat to human life and health. Global cancer prevalence data in 2022 indicated that the number of new cases of CRC was about 1.92 million and the deaths were around 900,000. A variety of risk factors, including genes and environment, can induce the occurrence of CRC. Previous studies have focused on the impact of dietary patterns on the development of CRC and have ignored sleep factors. Sleep deprivation is a common problem as people's work pressure increases. Sleep disorders can lead to metabolic and immune system dysregulation in people, contributing to the development and progression of many tumors. At present, there are few reports on the relationship between sleep disorders and tumors. Therefore, the purpose of this paper is to summarize and interpret the relationship between various sleep disorders and the onset and progression of CRC. This review is the first to investigate the possible mechanisms of sleep leading to CRC from the perspectives of metabolic reprogramming, intestinal microbiota disorders, and the release of inflammatory factors. In conclusion, this study highlights the rational sleep pattern and duration, which can help inhibit the occurrence of CRC.
Keywords: colorectal cancer, sleep, metabolite, gut microbiota, inflammation
1. Introduction
Colorectal cancer (CRC) is a significant public health concern and ranks among the most prevalent malignancies worldwide. According to global cancer statistics, CRC accounted for approximately 1.92 million new cases and 900,000 deaths in 2022[1], making it the third most common cancer in both men and women[1]. With the continued development of emerging economies, the global incidence of CRC is projected to rise to 2.5 million new cases by 2035[2]. Gender disparities in CRC incidence and outcomes have been observed. Women over 65 exhibit higher mortality rates and lower 5-year survival rates compared to their male counterparts. Moreover, women are more likely to develop right-sided (proximal) colon cancer[3]. Addressing the rising incidence and mortality of CRC remains a pressing challenge. The incidence of CRC is influenced by a complex interplay of genetic, environmental, and lifestyle factors[4]. Established risk factors include the consumption of processed meats, alcohol, smoking, and obesity. Conversely, protective factors such as dietary fiber, whole grains, dairy products, fruits, non-starchy vegetables, and supplementation with multivitamins or vitamin D lower the risk of CRC[5]. The intricate molecular characteristics of CRC reveal its association with genetic predisposition, oncogene mutations, gut microbiota imbalances, metabolic dysfunctions, and immune system responses[6,7].
While previous research has emphasized the role of lifestyle factors (such as diet and exercise) in CRC development, these factors alone do not fully account for the rising global incidence. Therefore, it is necessary to investigate other potential risk factors further. In recent years, the growing prevalence of sleep disorders, driven by increasing workloads and mental stress, has emerged as a topic of significant interest. Sleep, a fundamental physiological process constituting roughly one-third of human life, is indispensable for physical and mental well-being[8]. Adults are recommended to sleep at least 7-8 hours per night to maintain optimal health[9]. As a modifiable lifestyle factor, sleep quality is critical for overall health[10]. Poor sleep has been linked to a range of health conditions, including cardiovascular diseases, metabolic disorders, and cognitive dysfunction[10]. Emerging evidence also suggests that inadequate sleep may elevate the risk of several cancers, including CRC[11].
However, most studies investigating the link between sleep health and cancer have predominantly focused on breast[12-14], lung[15-17], and gastric cancers[18-20]. Research examining the relationship between sleep and CRC remains limited, and the underlying mechanisms connecting poor sleep quality to CRC risk are not yet well understood[19,21,22]. Therefore, this study aims to explore the relationship between sleep and CRC, uncover the underlying mechanisms and propose strategies for CRC prevention and management through the lens of sleep health.
2.1 Sleep disturbances alter metabolite levels and promote CRC development
Disruptions in sleep patterns and circadian rhythms can lead to significant metabolic dysregulation, contributing to obesity, insulin resistance, and diabetes. These metabolic disturbances can affect the concentrations of various blood metabolites[23]. Melatonin, an indole hormone derived from serotonin via the tryptophan-serotonin pathway in the pineal gland, plays a critical role in regulating circadian rhythms and promoting sleep quality. Its production increases with reduced exposure to light, and disruptions in circadian rhythms can significantly decrease melatonin levels[24,25]. Experimental studies have shown that acute sleep deprivation disrupts the rhythmicity of plasma metabolites, leading to elevated serotonin and tryptophan levels. These changes may be attributed to the reduced secretion of melatonin under sleep-deprived conditions, resulting in an accumulation of its precursor substances[23]. Beyond its role in regulating the sleep/wake cycle and circadian rhythms, melatonin is involved in immune regulation, antioxidant defense, and tumor regulation, influencing tumor initiation, progression, and prevention[26]. Changes in melatonin levels significantly increase the vulnerability of colorectal cells to carcinogenesis. Melatonin slows CRC progression by inhibiting cell proliferation and inducing apoptosis in CRC cells[27]. A cohort study by Zhang et al. reported a significant inverse relationship between melatonin use and CRC risk among individuals aged 50 and above (hazard ratio [HR]=0.82, 95% confidence interval [CI]: 0.72-0.92)[28]. Furthermore, another study highlighted that melatonin is safe and well-tolerated across a broad dosage range, positioning it as a promising therapeutic candidate for CRC treatment[29]. In summary, disrupted sleep patterns elevate CRC risk by adversely affecting melatonin levels.
2.2 Sleep disturbances drive CRC progression by inducing gut microbiota dysbiosis
The gut microbiota (GM) comprises a complex ecosystem of microorganisms within the human digestive tract, playing a pivotal role in nutrient synthesis, immune function, and maintaining intestinal barrier integrity[30]. In adults, over 90% of gut bacteria belong to the phyla Firmicutes, Bacteroidetes, Proteobacteria, or Actinobacteria, underscoring GM's critical association with intestinal health[31]. Recent studies indicate that sleep disturbances can significantly alter the composition and balance of the GM[32].
Obstructive sleep apnea (OSA), the most prevalent form of sleep-disordered breathing, is characterized by sleep fragmentation and intermittent hypoxia (IH)[33]. Zhang et al. reported significant alterations in GM diversity and abundance in humans and rodents with OSA, particularly affecting the phyla Firmicutes and Bacteroidetes[34]. Similarly, Collado et al. found that children experiencing chronic IH due to snoring exhibited an increased abundance of Firmicutes and a decreased abundance of Bacteroidetes in their gut[35]. Gao et al. corroborated these findings in mouse experiments, observing similar microbiota changes[36]. Further supporting the link between GM alterations and CRC, Flemer et al. conducted prospective research revealing an increased abundance of Bacteroidetes Cluster 2 and Firmicutes Cluster 2 in CRC mucosa, highlighting their potential role in CRC progression[37]. Gao et al. further demonstrated that IH and GM dysbiosis activated hypoxia-inducible factor-1α (HIF-1α) expression and the Signal Transducer and Activator of Transcription 3 (STAT3) pathway in colonic epithelial cells, promoting OSA-related CRC development[38]. As modern society advances, shift work has become increasingly common, bringing unique sleep challenges, including difficulty initiating sleep, reduced sleep duration, and excessive daytime sleepiness[39]. A review by Reynolds et al. linked sleep disturbances among shift workers to GM dysbiosis, suggesting that sleep deprivation and circadian rhythm disruptions act as physiological stressors that disrupt GM composition. This dysbiosis can contribute to metabolic disorders such as obesity and type 2 diabetes[40]. Disruption of intestinal epithelial permeability due to these stressors leads to an imbalance between beneficial and pathogenic bacteria[41], which plays a significant role in the progression of metabolic conditions such as obesity and diabetes, both high-risk factors for CRC. Moreover, increased gut epithelial permeability and bacterial translocation further contribute to CRC initiation and advancement[42,43].
In an experiment conducted by Bishehsari et al. using mice with adenomatous polyposis coli, researchers investigated the combined effects of alcohol consumption and circadian rhythm disruption. Both groups of mice were fed an alcohol-containing diet; however, one group maintained a standard 12-hour light/12-hour dark cycle, while the other experienced weekly 12-hour phase shifts in their light/dark cycle, disrupting their circadian rhythm. The study revealed that circadian rhythm disruption substantially exacerbated alcohol-induced polyposis and CRC development. Fecal microbiota analysis revealed that circadian rhythm disturbances promoted the transition of mast cells from MCP2+ to MCP6+, thereby mediating inflammation and fostering a pro-tumor inflammatory environment[44].
Research has demonstrated that various sleep disturbances, such as sleep disorders, insomnia, and circadian rhythm disruptions, induce significant alterations in GM composition and function[32]. Abnormal sleep patterns and durations affect the composition, diversity, and functionality of GM via the brain-gut-microbiota axis[45]. Moreover, the relationship between sleep quality and GM is bidirectional, as maintaining proper GM diversity promotes healthier sleep patterns[46]. The GM can contribute to the development of CRC by releasing various metabolites, proteins, and macromolecules that interact with the host's colonic epithelial and immune cells[47]. For instance, Okumura et al. demonstrated that Porphyromonas species promote CRC progression by secreting the bacterial metabolite butyrate, which subsequently induces cellular senescence[48].
Emerging research continues to elucidate the mechanisms through which GM influences CRC. Specific bacteria, including Fusobacterium nucleatum, Escherichia coli, Enterococcus faecalis, Streptococcus gallolyticus, and enterotoxigenic Bacteroides fragilis, have been identified as closely associated with CRC initiation and progression[49]. However, it remains unclear whether poor sleep habits directly alter the composition of these specific bacteria. Nonetheless, sleep disturbances can influence GM composition at the phylum level, causing dysbiosis that elevates CRC risk.
2.3 Sleep disturbances induce pro-inflammatory cytokine release, fostering CRC development
The immune system is essential for maintaining health by identifying and eliminating cancerous cells through immune surveillance. Besides this primary function, other immune components, including various immune cells and regulatory factors, play a pivotal role in suppressing cancer[50,51]. Burgos-Molina et al.'s review highlights that chronic inflammation, mediated by various cells and factors such as macrophages, lymphocytes, and pro-inflammatory cytokines such as tumor necrotic factor-α (TNF-α), interleukin-6 (IL-6), and IL-1β, promotes tumor progression and alters the tumor microenvironment in multiple ways, making it a significant factor in CRC development[51]. Increasing evidence suggests that sleep deprivation impairs anti-tumor immune responses, thereby promoting cancer progression[52].
In their review, Akkaoui et al. summarize the changes in several regulatory factors during sleep, noting that sleep deprivation considerably elevates the levels of pro-inflammatory cytokines (such as IL-1, IL-6, and TNF-α)[53]. A study tracking 2,500 older individuals over 7 years found that sleeping less than 5 hours a night was associated with elevated levels of pro-inflammatory cytokines (such as IL-6 and TNF-α) and CRP[54]. Furthermore, both insufficient and excessive sleep can lead to increased levels of pro-inflammatory cytokine levels. Patel et al. investigated the sleep patterns of 614 individuals and found that the average habitual sleep duration was 7.6 hours. As sleep duration increased, so did the levels of CRP and IL-6. Each additional hour of habitual sleep was associated with an 8% rise in CRP levels and a 7% increase in IL-6 levels. Conversely, for every hour of sleep reduction, TNF-α levels increased by an average of 8%[55]. Interleukins (ILs) can promote CRC development through various mechanisms, including tumor initiation, growth, angiogenesis, and metastasis[56]. Interleukin-6 (IL-6) plays a crucial role in nearly every stage of CRC progression. Upon binding to glycoprotein 130 (gp130), IL-6 primarily regulates CRC development through three signaling pathways: Src Homology 2 Domain-containing Phosphatase 2 - Rat Sarcoma - Extracellular Signal-Regulated Kinase (Shp2-Ras-ERK), Janus Kinase 1/2 (JAK1/2)-STAT3, and Phosphoinositide 3-Kinase - Protein Kinase B - Mechanistic Target of Rapamycin (PI3K-Akt-mTOR)[56]. Furthermore, a meta-analysis by Liu et al. identified IL-1 family members, including IL-1α, IL-1β, and Interleukin-1 Receptor Antagonist Variable Number Tandem Repeats (IL-1RN VNTR), as significant genes associated with CRC susceptibility[57]. The TNF facilitates CRC progression by activating the nuclear factor-kappa B (NF-κB) pathway, which supports cancer cell survival and induces gene expression in invasion and metastasis[58].
Bao et al.'s mouse model experiment demonstrated that sleep deprivation increases gamma-aminobutyric acid (GABA) levels in peripheral blood, promoting CRC cell proliferation and migration through the endogenous regulation of miR-223-3p. Furthermore, microRNA miR-223-3p enters macrophages via exosomes, activating the mitogen-activated protein kinase (MAPK) pathway, inducing M2 polarization, and stimulating the secretion of IL-17, further accelerating tumor cell proliferation and migration[59].
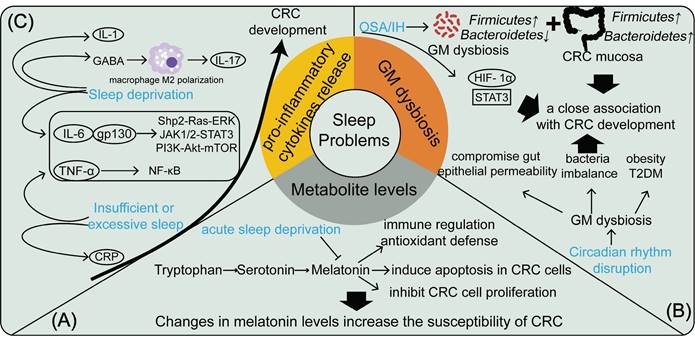
In summary, sleep problems elevate CRC risk by influencing the release of pro-inflammatory cytokines and promoting systemic inflammation (Figure 1).
The mechanisms by which sleep problems contribute to the development of CRC are manifested in changes in metabolite levels, GM flora dysbiosis, and the release of pro-inflammatory cytokines. (A) Acute sleep deprivation causes changes in melatonin levels, which increases CRC susceptibility; (B) OSA/IH and circadian rhythm disruption, as physiological stressors, disrupt the gut microbiota and are closely associated with CRC development; (C) Sleep deprivation and insufficient or excessive sleep cause the release of multiple pro-inflammatory factors in the body, which promotes CRC development.
3. Research on the correlation between sleep patterns and CRC
Sleep can be assessed across dimensions, including sleep quality, duration, efficiency, timing, and alertness[60]. Sleep quality and duration are particularly significant in evaluating sleep status[61]. Changes in sleep quality can trigger a series of physiological responses, with sleep disorders being a major contributing factor. The following sections explore the relationship between poor sleep habits and CRC from four perspectives.
3.1 Correlation between sleep disorders and CRC
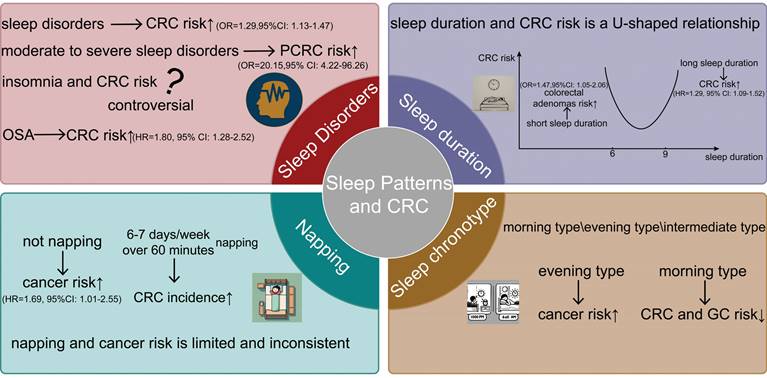
Sleep disorders are crucial indicators of sleep quality. According to the third edition of the International Classification of Sleep Disorders, sleep disorders are categorized into insomnia, sleep-related breathing disorders (such as OSA), hypersomnia, circadian rhythm sleep-wake disorders, and parasomnias[62]. Sleep disorders have been linked to various physical and mental health issues[10]. Emerging evidence suggests a potential association between sleep disorders and cancer development[63]. Several studies have specifically identified a higher risk of CRC in individuals with sleep disorders[11,18,64,65]. A population-based nested case-control study reported that individuals with sleep disorders faced a significantly elevated risk of CRC compared to those without such disorders (odds ratio [OR]=1.29, 95% CI: 1.13-1.47). Moreover, patients with sleep disorders and depression exhibited an alarming 5.69-fold increased risk of CRC compared to the control group[65]. Liu et al.[66] employed tools such as the Pittsburgh Sleep Quality Index (PSQI), Perceived Stress Scale (PSS10), Multidimensional Scale of Perceived Social Support (MSPSS), and Connor-Davidson Resilience Scale (CD-RISC) to examine the interplay between sleep quality, psychological factors (such as stress, resilience, and social support), and the risk of precancerous conditions of CRC (PCRC). Their findings revealed that a PSQI score indicative of moderate to severe sleep disorders (≥2) was significantly associated with an increased risk of PCRC (OR=20.15, 95% CI: 4.22-96.26).
3.2 Correlation between insomnia and CRC
Insomnia, a sleep continuity disorder, is characterized by difficulties initiating or maintaining sleep, often resulting in daytime symptoms such as excessive fatigue, physical discomfort (headaches or body pain), mood disturbances, cognitive or occupational impairment, and dissatisfaction with sleep quality. Diagnosis requires evidence of sleep-related challenges that lead to functional impairments during the day despite adequate opportunities for rest[67,68]. Insomnia has been linked to an increased risk of several health conditions, including depression, dementia, non-alcoholic fatty liver disease, hypertension, and cardiovascular diseases[69]. Furthermore, epidemiological studies have consistently suggested a correlation between insomnia and cancer[70-72]. A recent meta-analysis reported that individuals with insomnia have a 24% increased overall risk of developing cancer[71]. Mechanisms potentially underlying this association include disruptions in melatonin, circadian rhythm disruptions, and imbalances in appetite-regulating hormones (such as leptin and ghrelin)[71]. The relationship between insomnia and CRC risk remains controversial. A study conducted in Taiwan identified a substantially higher risk of colon cancer in patients with insomnia[70], and findings from a nested case-control study supported an increased risk of CRC in individuals with insomnia[65]. However, contrasting evidence from a large nationwide cohort study in Korea indicated a decreased risk of CRC among patients with insomnia, regardless of gender[72].
3.3 Correlation between obstructive sleep apnea (OSA) and CRC
OSA is a sleep disorder characterized by recurrent episodes of complete or partial upper airway obstruction during sleep, leading to apnea or hypopnea[73]. This condition is associated with intermittent hypoxia, sleep fragmentation (SF), and heightened sympathetic nervous activity. Common clinical manifestations include loud snoring, excessive daytime sleepiness, and cognitive impairment[73]. Research has highlighted OSA's significant role in cancer development, with its effects primarily attributed to intermittent hypoxemia, SF, or both[74]. These factors can disrupt standard cellular transcription by altering sympathetic nerve activity, promoting angiogenesis, activating inflammatory pathways, and modulating immune cell behavior. Such changes may enhance tumor aggressiveness, invasiveness, and resistance to treatment[75,76]. Hypoxic environments caused by OSA may contribute to CRC progression[77,78]. A population-based retrospective cohort study revealed that individuals with OSA had a significantly higher risk of developing CRC than non-OSA individuals (adjusted HR=1.80, 95% CI: 1.28-2.52). Moreover, the risk appeared to increase with the frequency of medical visits related to OSA[77]. However, a meta-analysis by Niranjan et al. noted that while biological mechanisms suggest a potential link, there is insufficient evidence to establish OSA as a direct risk factor for CRC conclusively. Further studies are required to clarify this relationship[79].
3.4 Correlation between Sleep Duration and CRC
Sleep duration has emerged as a potential risk factor influencing the incidence and mortality of various chronic diseases[80-82]. Both long sleep duration (typically defined as ≥9 hours) and short sleep duration (typically defined as ≤6 hours) have been associated with increased risks of cardiovascular disease[83,84], type 2 diabetes[85,86], obesity[86,87], and cancer[22,88]. However, the association between sleep duration and CRC is inconsistent. Some studies suggest a U-shaped relationship on the graph, indicating that short and long sleep durations may elevate CRC risk[89]. Other studies report an association with only long sleep duration[22,90] or short sleep duration[91-93], while some research indicates no considerable association between sleep duration and CRC risk[94].
Prolonged sleep duration has been associated with a 21% increase in CRC risk[16], potentially due to heightened levels of systemic inflammation[95], a critical factor in cancer development and progression[96]. A meta-analysis by Zhao et al.[88] examining cohort studies further supported this finding, identifying a positive correlation between sleep duration and CRC risk (HR=1.29, 95% CI: 1.09-1.52). A case-control study on self-reported sleep duration revealed that long sleep duration (≥9 hours) was associated with an increased risk of CRC compared to the reference duration of 7 hours[19]. Zhang et al.[90] conducted a 22-year follow-up of 30,121 men and 76,368 women for 22 years. They found that compared to an average sleep duration of 7 hours, a significant association was observed between long sleep duration and CRC risk in men (HR=1.35, 95% CI: 1.00-1.82), with a weaker association in women (HR=1.11, 95% CI: 0.85-1.44). This risk was particularly notable in individuals who were overweight or frequently snored. Similarly, a meta-analysis by Lu et al.[97] of 10 studies (involving 8,392 cases and 555,678 participants) confirmed a positive correlation between sleep duration and CRC risk (relative risk [RR]=1.29, 95% CI: 1.09-1.52).
Conversely, short sleep duration has been linked to a 54% increase in CRC-related mortality[93]. Disrupted circadian rhythms[98], reduced nocturnal melatonin production[63], and impaired immune function[99] associated with insufficient sleep may contribute to cancer progression. Furthermore, short sleep is linked to overweight and obesity[100], which elevate cancer risk through mechanisms such as inflammation and insulin resistance[101,102]. Thompson et al.[92] reported a significant association between short sleep duration and the occurrence of colorectal adenomas. Individuals sleeping fewer than 6 hours per night exhibited a nearly 50% increased risk of developing colorectal adenomas compared to those sleeping at least 7 hours (OR=1.47, 95% CI: 1.05-2.06), highlighting short sleep duration as a potential risk factor for early-onset CRC. In a national randomized clinical trial evaluating patients with resected stage III colon cancer receiving standardized treatment and follow-up, hazard ratios for sleep durations of ≤5 hours and ≥9 hours were 2.14 (95% CI: 1.14-4.03) and 2.34 (95% CI: 1.26-4.33), respectively. These findings underscore the significant association between extreme sleep durations and increased mortality risk[103]. Therefore, maintaining an optimal sleep duration is crucial for preventing CRC and improving patient outcomes.
3.5 Correlation between napping and CRC
Beyond nighttime sleep, napping is an important indicator of overall sleep health[104]. However, research on the relationship between napping and cancer remains limited and inconsistent. Some studies suggest that the absence of napping may elevate cancer risk. For instance, a 2023 study reported that participants who did not nap faced a 60% higher cancer risk compared to those napping over 60 minutes (HR=1.69, 95% CI: 1.01-2.55), with the risk further increasing in those who combined non-napping with short nighttime sleep[105]. Another study focusing on men revealed that not napping during the day was associated with a higher cancer risk compared to napping for 1-30 minutes (HR=2.03, 95%CI: 1.01-4.13)[106].
Conversely, other studies suggest that frequent or prolonged naps may increase cancer risk. For instance, a 2021 case-control study reported that napping frequently (6-7 days/week) and for extended durations (over 60 minutes) naps were associated with an increased CRC incidence[19]. Similarly, an analysis of 4,869 patients with CRC revealed that daily naps lasting 1 hour or more were associated with higher overall and cardiovascular disease-related mortality. However, no significant association was observed with CRC-specific mortality[93].
Overall, the impact of napping on cancer risk, particularly its relationship with CRC, remains unclear and warrants further investigation.
3.6 Correlation between sleep-wake patterns and CRC
Sleep timing (encompassing bedtime and wake-up time) reflects an individual's sleep-wake preference and schedule. Recent studies have highlighted the association between sleep timing and adverse health outcomes, linking later bedtimes to negative effects. For instance, research indicates that individuals who fall asleep between 10:00 and 11:00 PM have the lowest risk of cardiovascular disease, while those going to bed at midnight or later experience the highest incidence[107]. Similarly, a community-based study of Hispanic/Latino adults aged 18-74 found that later bedtimes were correlated with increased insulin resistance[108].
Although limited research exists on the direct relationship between sleep timing and cancer risk, studies have investigated the role of sleep chronotype in cancer. Sleep chronotype, an essential characteristic of sleep patterns, reflects an individual's preferred timing for sleep and wakefulness. Chronotypes are generally classified into three categories: morning type (prefers early sleep and rise), evening type (late sleep and late rise), and intermediate type, with morning and evening types representing the extremes of this spectrum.
Evening chronotypes are associated with lower physical activity[109] and poor dietary choices[110], which may contribute to adverse health effects and a heightened cancer risk. Conversely, morning types appear to lower the risk of certain cancers. Research has shown a significant association between morning types and a reduced risk of breast[13] and prostate cancers[111]. Yuan et al. conducted a Mendelian randomization study using data from the UK Biobank and Finnish databases to assess the association between gastrointestinal cancers and sleep chronotype. They identified an inverse correlation between morning types and the risk of gastrointestinal cancers, particularly CRC and gastric cancers[20] (Figure 2).
Through the four dimensions of sleep disorders, sleep time, naps, and sleep patterns, their relationship with the occurrence and development of CRC is presented from an epidemiological perspective. Sleep disorders (insomnia, OSA), too long or too short sleep duration, the presence or absence of napping, and the type of sleep chronotypes all affect the incidence of CRC.
4. Conclusion
The incidence and mortality rates of CRC remain alarmingly high, posing a significant health threat. While the prognosis of CRC has slightly improved with the widespread use of colonoscopy and the removal of precursor lesions, the incidence of CRC is rising among younger individuals in some high-income countries. It is well-established that unhealthy lifestyle factors, such as smoking, excessive alcohol consumption, sedentary behavior, and high-fat, low-fiber diets, increase the risk of CRC. However, with increasing mental stress, sleep disturbances have become more prevalent. Numerous studies have established a correlation between sleep patterns and cancer, suggesting that poor sleep may elevate cancer risk and influence the development and prognosis of CRC.
This review examines how inadequate sleep contributes to CRC progression through metabolic changes, GM dysbiosis, and the release of pro-inflammatory cytokines. Disrupted sleep habits impact melatonin production, alter the gut microbiota by disrupting the balance of Firmicutes and Bacteroidetes, and promote inflammation by triggering cytokines (such as IL-1, IL-6, and TNF-α), all of which support CRC development. Furthermore, this review discusses the association between sleep patterns and CRC from an epidemiological perspective, focusing on sleep disorders, sleep duration, napping, and sleep-wake cycles. Improving sleep quality, maintaining a balanced sleep duration, adopting a regular napping routine, and establishing early sleep and wake habits can significantly lower CRC risk.
Research on the correlation between sleep habits and CRC is limited, and the topic remains controversial. Moreover, the lack of randomized controlled trials (RCT) establishes a direct causal relationship between sleep disorders and CRC in the general population. Most existing studies are descriptive and observational, failing to explore the underlying mechanisms connecting poor sleep habits to CRC. These gaps present ongoing challenges in this field. However, future research is expected to provide deeper insights into the relationship between sleep habits and CRC, offering new avenues for early prevention and effective management of the disease.
Acknowledgements
We thank Bullet Edits Limited for the linguistic editing and proofreading of the manuscript.
Author contributions
L.J., Y.Q. and Z.Y.: conception, design, collection and assembly of data, and interpretation, and manuscript writing; W.X., W.R., and Z.C.: collection and assembly of data, and interpretation, and manuscript writing; Z.L.: manuscript revising; H.Z.: conception, design, and manuscripting revising. All authors were involved in the final approval of the manuscript.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I. et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74:229-63
2. Dekker E, Tanis PJ, Vleugels JLA, Kasi PM, Wallace MB. Colorectal cancer. Lancet. 2019;394:1467-80
3. Kim S-E, Paik HY, Yoon H, Lee JE, Kim N, Sung M-K. Sex- and gender-specific disparities in colorectal cancer risk. World J Gastroenterol. 2015;21:5167-75
4. Keum N, Giovannucci E. Global burden of colorectal cancer: emerging trends, risk factors and prevention strategies. Nat Rev Gastroenterol Hepatol. 2019;16:713-32
5. Li N, Lu B, Luo C, Cai J, Lu M, Zhang Y. et al. Incidence, mortality, survival, risk factor and screening of colorectal cancer: A comparison among China, Europe, and northern America. Cancer Lett. 2021;522:255-68
6. Wozniakova M, Skarda J, Raska M. The Role of Tumor Microenvironment and Immune Response in Colorectal Cancer Development and Prognosis. Pathol Oncol Res. 2022;28:1610502
7. Ionescu VA, Gheorghe G, Bacalbasa N, Chiotoroiu AL, Diaconu C. Colorectal Cancer: From Risk Factors to Oncogenesis. Medicina (Kaunas). 2023;59:1646
8. Luyster FS, Strollo PJ, Zee PC, Walsh JK, Boards of Directors of the American Academy of Sleep Medicine and the Sleep Research Society. Sleep: a health imperative. Sleep. 2012;35:727-34
9. Chaput J-P, Dutil C, Featherstone R, Ross R, Giangregorio L, Saunders TJ. et al. Sleep duration and health in adults: an overview of systematic reviews. Appl Physiol Nutr Metab. 2020;45:S218-31
10. Grandner MA. Sleep, Health, and Society. Sleep Med Clin. 2020;15:319-40
11. Li W, Li C, Liu T, Wang Y, Ma X, Xiao X. et al. Self-reported sleep disorders and the risk of all cancer types: evidence from the Kailuan Cohort study. Public Health. 2023;223:209-16
12. Qin Y, Zhou Y, Zhang X, Wei X, He J. Sleep duration and breast cancer risk: a meta-analysis of observational studies. Int J Cancer. 2014;134:1166-73
13. Richmond RC, Anderson EL, Dashti HS, Jones SE, Lane JM, Strand LB. et al. Investigating causal relations between sleep traits and risk of breast cancer in women: mendelian randomisation study. BMJ. 2019;365:l2327
14. Wong ATY, Heath AK, Tong TYN, Reeves GK, Floud S, Beral V. et al. Sleep duration and breast cancer incidence: results from the Million Women Study and meta-analysis of published prospective studies. Sleep. 2021;44:zsaa166
15. Cordina-Duverger E, Uchai S, Tvardik N, Billmann R, Martin D, Trédaniel J. et al. Sleep Traits, Night Shift Work and Lung Cancer Risk among Women: Results from a Population-Based Case-Control Study in France (The WELCA Study). Int J Environ Res Public Health. 2022;19:16246
16. Li X, Huang D, Liu F, Li X, Lv J, Wu Q. et al. Sleep Characteristics and Cancer-Related Outcomes: An Umbrella Review of Systematic Reviews and Meta-Analyses of Observational Studies. J Clin Med. 2022;11:7289
17. Wang J, Tang H, Duan Y, Yang S, An J. Association between Sleep Traits and Lung Cancer: A Mendelian Randomization Study. J Immunol Res. 2021;2021:1893882
18. Loosen S, Krieg S, Krieg A, Leyh C, Luedde T, Vetter C. et al. Are sleep disorders associated with the risk of gastrointestinal cancer?-A case-control study. J Cancer Res Clin Oncol. 2023;149:11369-78
19. Papantoniou K, Castaño-Vinyals G, Espinosa A, Turner MC, Martín-Sánchez V, Casabonne D. et al. Sleep duration and napping in relation to colorectal and gastric cancer in the MCC-Spain study. Sci Rep. 2021;11:11822
20. Yuan S, Mason AM, Titova OE, Vithayathil M, Kar S, Chen J. et al. Morning chronotype and digestive tract cancers: Mendelian randomization study. Int J Cancer. 2023;152:697-704
21. Wang G, Wang J-J, Lin C-H, Zhou Q, Wang W-L, Qin T. et al. Association of sleep duration, sleep apnea, and shift work with risk of colorectal neoplasms: a systematic review and meta-analysis. J Gastrointest Oncol. 2022;13:1805-17
22. Chen Y, Tan F, Wei L, Li X, Lyu Z, Feng X. et al. Sleep duration and the risk of cancer: a systematic review and meta-analysis including dose-response relationship. BMC Cancer. 2018;18:1149
23. Davies SK, Ang JE, Revell VL, Holmes B, Mann A, Robertson FP. et al. Effect of sleep deprivation on the human metabolome. Proc Natl Acad Sci U S A. 2014;111:10761-6
24. Malhotra S, Sawhney G, Pandhi P. The therapeutic potential of melatonin: a review of the science. MedGenMed. 2004;6:46
25. Vasey C, McBride J, Penta K. Circadian Rhythm Dysregulation and Restoration: The Role of Melatonin. Nutrients. 2021;13:3480
26. Srinivasan V, Spence DW, Pandi-Perumal SR, Trakht I, Cardinali DP. Therapeutic actions of melatonin in cancer: possible mechanisms. Integr Cancer Ther. 2008;7:189-203
27. Gong Y-Q, Hou F-T, Xiang C-L, Li C-L, Hu G-H, Chen C-W. The mechanisms and roles of melatonin in gastrointestinal cancer. Front Oncol. 2022;12:1066698
28. Zhang N, Sundquist J, Sundquist K, Ji J. Use of Melatonin Is Associated With Lower Risk of Colorectal Cancer in Older Adults. Clin Transl Gastroenterol. 2021;12:e00396
29. Iravani S, Eslami P, Dooghaie Moghadam A, Moazzami B, Mehrvar A, Hashemi MR. et al. The Role of Melatonin in Colorectal Cancer. J Gastrointest Cancer. 2020;51:748-53
30. Nishida A, Inoue R, Inatomi O, Bamba S, Naito Y, Andoh A. Gut microbiota in the pathogenesis of inflammatory bowel disease. Clin J Gastroenterol. 2018;11:1-10
31. Adak A, Khan MR. An insight into gut microbiota and its functionalities. Cell Mol Life Sci. 2019;76:473-93
32. Wang Z, Wang Z, Lu T, Chen W, Yan W, Yuan K. et al. The microbiota-gut-brain axis in sleep disorders. Sleep Med Rev. 2022;65:101691
33. Moreno-Indias I, Torres M, Sanchez-Alcoholado L, Cardona F, Almendros I, Gozal D. et al. Normoxic Recovery Mimicking Treatment of Sleep Apnea Does Not Reverse Intermittent Hypoxia-Induced Bacterial Dysbiosis and Low-Grade Endotoxemia in Mice. Sleep. 2016;39:1891-7
34. Zhang X, Wang S, Xu H, Yi H, Guan J, Yin S. Metabolomics and microbiome profiling as biomarkers in obstructive sleep apnoea: a comprehensive review. Eur Respir Rev. 2021;30:200220
35. Collado MC, Katila MK, Vuorela NM, Saarenpää-Heikkilä O, Salminen S, Isolauri E. Dysbiosis in Snoring Children: An Interlink to Comorbidities? J Pediatr Gastroenterol Nutr. 2019;68:272-7
36. Gao T, Wang Z, Dong Y, Cao J, Lin R, Wang X. et al. Role of melatonin in sleep deprivation-induced intestinal barrier dysfunction in mice. J Pineal Res. 2019;67:e12574
37. Flemer B, Lynch DB, Brown JMR, Jeffery IB, Ryan FJ, Claesson MJ. et al. Tumour-associated and non-tumour-associated microbiota in colorectal cancer. Gut. 2017;66:633-43
38. Gao J, Cao H, Zhang Q, Wang B. The effect of intermittent hypoxia and fecal microbiota of OSAS on genes associated with colorectal cancer. Sleep Breath. 2021;25:1075-87
39. Wright KP, Bogan RK, Wyatt JK. Shift work and the assessment and management of shift work disorder (SWD). Sleep Med Rev. 2013;17:41-54
40. Reynolds AC, Paterson JL, Ferguson SA, Stanley D, Wright KP, Dawson D. The shift work and health research agenda: Considering changes in gut microbiota as a pathway linking shift work, sleep loss and circadian misalignment, and metabolic disease. Sleep Medicine Reviews. 2017;34:3-9
41. Dinan TG, Cryan JF. Regulation of the stress response by the gut microbiota: implications for psychoneuroendocrinology. Psychoneuroendocrinology. 2012;37:1369-78
42. Gausman V, Dornblaser D, Anand S, Hayes RB, O'Connell K, Du M. et al. Risk Factors Associated With Early-Onset Colorectal Cancer. Clin Gastroenterol Hepatol. 2020;18:2752-2759.e2
43. Zhang Y, Zhang J, Xia Y, Sun J. Bacterial translocation and barrier dysfunction enhance colonic tumorigenesis. Neoplasia. 2023;35:100847
44. Bishehsari F, Saadalla A, Khazaie K, Engen PA, Voigt RM, Shetuni BB. et al. Light/Dark Shifting Promotes Alcohol-Induced Colon Carcinogenesis: Possible Role of Intestinal Inflammatory Milieu and Microbiota. Int J Mol Sci. 2016;17:2017
45. Han M, Yuan S, Zhang J. The interplay between sleep and gut microbiota. Brain Res Bull. 2022;180:131-46
46. Smith RP, Easson C, Lyle SM, Kapoor R, Donnelly CP, Davidson EJ. et al. Gut microbiome diversity is associated with sleep physiology in humans. PLoS One. 2019;14:e0222394
47. Wong CC, Yu J. Gut microbiota in colorectal cancer development and therapy. Nat Rev Clin Oncol. 2023;20:429-52
48. Okumura S, Konishi Y, Narukawa M, Sugiura Y, Yoshimoto S, Arai Y. et al. Gut bacteria identified in colorectal cancer patients promote tumourigenesis via butyrate secretion. Nat Commun. 2021;12:5674
49. Quaglio AEV, Grillo TG, De Oliveira ECS, Di Stasi LC, Sassaki LY. Gut microbiota, inflammatory bowel disease and colorectal cancer. World J Gastroenterol. 2022;28:4053-60
50. Fletcher R, Wang Y-J, Schoen RE, Finn OJ, Yu J, Zhang L. Colorectal cancer prevention: Immune modulation taking the stage. Biochim Biophys Acta Rev Cancer. 2018;1869:138-48
51. Burgos-Molina AM, Téllez Santana T, Redondo M, Bravo Romero MJ. The Crucial Role of Inflammation and the Immune System in Colorectal Cancer Carcinogenesis: A Comprehensive Perspective. Int J Mol Sci. 2024;25:6188
52. Garbarino S, Lanteri P, Bragazzi NL, Magnavita N, Scoditti E. Role of sleep deprivation in immune-related disease risk and outcomes. Commun Biol. 2021;4:1304
53. Akkaoui MA, Palagini L, Geoffroy PA. Sleep Immune Cross Talk and Insomnia. Adv Exp Med Biol. 2023;1411:263-73
54. Smagula SF, Stone KL, Redline S, Ancoli-Israel S, Barrett-Connor E, Lane NE. et al. Actigraphy- and Polysomnography-Measured Sleep Disturbances, Inflammation, and Mortality Among Older Men. Psychosom Med. 2016;78:686-96
55. Patel SR, Zhu X, Storfer-Isser A, Mehra R, Jenny NS, Tracy R. et al. Sleep duration and biomarkers of inflammation. Sleep. 2009;32:200-4
56. Li J, Huang L, Zhao H, Yan Y, Lu J. The Role of Interleukins in Colorectal Cancer. Int J Biol Sci. 2020;16:2323-39
57. Liu L, Zhai Z, Wang D, Ding Y, Chen X, Wang Q. et al. The association between IL-1 family gene polymorphisms and colorectal cancer: A meta-analysis. Gene. 2021;769:145187
58. Wang K, Karin M. Tumor-Elicited Inflammation and Colorectal Cancer. Adv Cancer Res. 2015;128:173-96
59. Bao H, Peng Z, Cheng X, Jian C, Li X, Shi Y. et al. GABA induced by sleep deprivation promotes the proliferation and migration of colon tumors through miR-223-3p endogenous pathway and exosome pathway. J Exp Clin Cancer Res. 2023;42:344
60. Buysse DJ. Sleep health: can we define it? Does it matter? Sleep. 2014;37:9-17
61. Lao XQ, Liu X, Deng H-B, Chan T-C, Ho KF, Wang F. et al. Sleep Quality, Sleep Duration, and the Risk of Coronary Heart Disease: A Prospective Cohort Study With 60,586 Adults. J Clin Sleep Med. 2018;14:109-17
62. Sateia MJ. International classification of sleep disorders-third edition: highlights and modifications. Chest. 2014;146:1387-94
63. Mogavero MP, DelRosso LM, Fanfulla F, Bruni O, Ferri R. Sleep disorders and cancer: State of the art and future perspectives. Sleep Med Rev. 2021;56:101409
64. Hsu F-C, Hsu C-H, Chung C-H, Pu T-W, Chang P-K, Lin T-C. et al. The Combination of Sleep Disorders and Depression Significantly Increases Cancer Risk: A Nationwide Large-Scale Population-Based Study. Int J Environ Res Public Health. 2022;19:9266
65. Lin C-L, Liu T-C, Wang Y-N, Chung C-H, Chien W-C. The Association Between Sleep Disorders and the Risk of Colorectal Cancer in Patients: A Population-based Nested Case-Control Study. In Vivo. 2019;33:573-9
66. Liu W, Zhang H. Do sleep quality and psychological factors link precancerous conditions of colorectal cancer? A retrospective case-control study. Expert Rev Gastroenterol Hepatol. 2022;16:173-9
67. Dopheide JA. Insomnia overview: epidemiology, pathophysiology, diagnosis and monitoring, and nonpharmacologic therapy. Am J Manag Care. 2020;26:S76-84
68. Perlis ML, Posner D, Riemann D, Bastien CH, Teel J, Thase M. Insomnia. Lancet. 2022;400:1047-60
69. Ge L, Guyatt G, Tian J, Pan B, Chang Y, Chen Y. et al. Insomnia and risk of mortality from all-cause, cardiovascular disease, and cancer: Systematic review and meta-analysis of prospective cohort studies. Sleep Med Rev. 2019;48:101215
70. Fang H-F, Miao N-F, Chen C-D, Sithole T, Chung M-H. Risk of Cancer in Patients with Insomnia, Parasomnia, and Obstructive Sleep Apnea: A Nationwide Nested Case-Control Study. J Cancer. 2015;6:1140-7
71. Shi T, Min M, Sun C, Zhang Y, Liang M, Sun Y. Does insomnia predict a high risk of cancer? A systematic review and meta-analysis of cohort studies. J Sleep Res. 2020;29:e12876
72. Yoon K, Shin CM, Han K, Jung JH, Jin EH, Lim JH. et al. Risk of cancer in patients with insomnia: Nationwide retrospective cohort study (2009-2018). PLoS One. 2023;18:e0284494
73. Kapur VK, Auckley DH, Chowdhuri S, Kuhlmann DC, Mehra R, Ramar K. et al. Clinical Practice Guideline for Diagnostic Testing for Adult Obstructive Sleep Apnea: An American Academy of Sleep Medicine Clinical Practice Guideline. J Clin Sleep Med. 2017;13:479-504
74. Gozal D, Farré R, Nieto FJ. Obstructive sleep apnea and cancer: Epidemiologic links and theoretical biological constructs. Sleep Med Rev. 2016;27:43-55
75. Martinez-Garcia MA, Campos-Rodriguez F, Almendros I, Garcia-Rio F, Sanchez-de-la-Torre M, Farre R. et al. Cancer and Sleep Apnea: Cutaneous Melanoma as a Case Study. Am J Respir Crit Care Med. 2019;200:1345-53
76. Hunyor I, Cook KM. Models of intermittent hypoxia and obstructive sleep apnea: molecular pathways and their contribution to cancer. Am J Physiol Regul Integr Comp Physiol. 2018;315:R669-87
77. Chen C-Y, Hu J-M, Shen C-J, Chou Y-C, Tian Y-F, Chen Y-C. et al. Increased incidence of colorectal cancer with obstructive sleep apnea: a nationwide population-based cohort study. Sleep Med. 2020;66:15-20
78. Lee S, Kim BG, Kim JW, Lee KL, Koo DL, Nam H. et al. Obstructive sleep apnea is associated with an increased risk of colorectal neoplasia. Gastrointest Endosc. 2017;85:568-573.e1
79. Niranjan N, Sriram KB, Gopalan V. Obstructive Sleep Apnea and Colorectal Cancer: A Systematic Review and Meta-Analysis. J Gastrointest Cancer. 2023;54:1151-7
80. Stone CR, Haig TR, Fiest KM, McNeil J, Brenner DR, Friedenreich CM. The association between sleep duration and cancer-specific mortality: a systematic review and meta-analysis. Cancer Causes Control. 2019;30:501-25
81. da Silva AA, de Mello RGB, Schaan CW, Fuchs FD, Redline S, Fuchs SC. Sleep duration and mortality in the elderly: a systematic review with meta-analysis. BMJ Open. 2016;6:e008119
82. Kecklund G, Axelsson J. Health consequences of shift work and insufficient sleep. BMJ. 2016;355:i5210
83. Krittanawong C, Tunhasiriwet A, Wang Z, Zhang H, Farrell AM, Chirapongsathorn S. et al. Association between short and long sleep durations and cardiovascular outcomes: a systematic review and meta-analysis. Eur Heart J Acute Cardiovasc Care. 2019;8:762-70
84. Cappuccio FP, Cooper D, D'Elia L, Strazzullo P, Miller MA. Sleep duration predicts cardiovascular outcomes: a systematic review and meta-analysis of prospective studies. Eur Heart J. 2011;32:1484-92
85. Shan Z, Ma H, Xie M, Yan P, Guo Y, Bao W. et al. Sleep duration and risk of type 2 diabetes: a meta-analysis of prospective studies. Diabetes Care. 2015;38:529-37
86. Cappuccio FP, Miller MA. Sleep and Cardio-Metabolic Disease. Curr Cardiol Rep. 2017;19:110
87. Ogilvie RP, Patel SR. The epidemiology of sleep and obesity. Sleep Health. 2017;3:383-8
88. Zhao H, Yin J-Y, Yang W-S, Qin Q, Li T-T, Shi Y. et al. Sleep duration and cancer risk: a systematic review and meta-analysis of prospective studies. Asian Pac J Cancer Prev. 2013;14:7509-15
89. Jiao L, Duan Z, Sangi-Haghpeykar H, Hale L, White DL, El-Serag HB. Sleep duration and incidence of colorectal cancer in postmenopausal women. Br J Cancer. 2013;108:213-21
90. Zhang X, Giovannucci EL, Wu K, Gao X, Hu F, Ogino S. et al. Associations of self-reported sleep duration and snoring with colorectal cancer risk in men and women. Sleep. 2013;36:681-8
91. Titova OE, Michaëlsson K, Vithayathil M, Mason AM, Kar S, Burgess S. et al. Sleep duration and risk of overall and 22 site-specific cancers: A Mendelian randomization study. Int J Cancer. 2021;148:914-20
92. Thompson CL, Larkin EK, Patel S, Berger NA, Redline S, Li L. Short duration of sleep increases risk of colorectal adenoma. Cancer. 2011;117:841-7
93. Xiao Q, Arem H, Pfeiffer R, Matthews C. Prediagnosis Sleep Duration, Napping, and Mortality Among Colorectal Cancer Survivors in a Large US Cohort. Sleep. 2017;40:zsx010
94. McNeil J, Heer E, Willemsen RF, Friedenreich CM, Brenner DR. The effects of shift work and sleep duration on cancer incidence in Alberta`s Tomorrow Project cohort. Cancer Epidemiol. 2020;67:101729
95. Irwin MR, Olmstead R, Carroll JE. Sleep Disturbance, Sleep Duration, and Inflammation: A Systematic Review and Meta-Analysis of Cohort Studies and Experimental Sleep Deprivation. Biol Psychiatry. 2016;80:40-52
96. Lee H-M, Lee H-J, Chang J-E. Inflammatory Cytokine: An Attractive Target for Cancer Treatment. Biomedicines. 2022;10:2116
97. Lu Y, Tian N, Yin J, Shi Y, Huang Z. Association between sleep duration and cancer risk: a meta-analysis of prospective cohort studies. PLoS One. 2013;8:e74723
98. Aiello I, Fedele MLM, Román F, Marpegan L, Caldart C, Chiesa JJ. et al. Circadian disruption promotes tumor-immune microenvironment remodeling favoring tumor cell proliferation. Sci Adv. 2020;6:eaaz4530
99. Bollinger T, Bollinger A, Oster H, Solbach W. Sleep, immunity, and circadian clocks: a mechanistic model. Gerontology. 2010;56:574-80
100. Broussard JL, Klein S. Insufficient sleep and obesity: cause or consequence. Obesity (Silver Spring). 2022;30:1914-6
101. Hildebrandt X, Ibrahim M, Peltzer N. Cell death and inflammation during obesity: “Know my methods, WAT(son).” Cell Death Differ. 2023;30:279-92
102. Amin MN, Hussain MS, Sarwar MS, Rahman Moghal MM, Das A, Hossain MZ. et al. How the association between obesity and inflammation may lead to insulin resistance and cancer. Diabetes Metab Syndr. 2019;13:1213-24
103. Lee S, Ma C, Shi Q, Meyers J, Kumar P, Couture F. et al. Sleep and cancer recurrence and survival in patients with resected Stage III colon cancer: findings from CALGB/SWOG 80702 (Alliance). Br J Cancer. 2023;129:283-90
104. Stang A. Daytime napping and health consequences: much epidemiologic work to do. Sleep Med. 2015;16:809-10
105. Ning D, Fang Y, Zhang W. Association of habitual sleep duration and its trajectory with the risk of cancer according to sex and body mass index in a population-based cohort. Cancer. 2023;129:3582-94
106. Bai Y, Li X, Wang K, Chen S, Wang S, Chen Z. et al. Association of shift-work, daytime napping, and nighttime sleep with cancer incidence and cancer-caused mortality in Dongfeng-tongji cohort study. Ann Med. 2016;48:641-51
107. Nikbakhtian S, Reed AB, Obika BD, Morelli D, Cunningham AC, Aral M. et al. Accelerometer-derived sleep onset timing and cardiovascular disease incidence: a UK Biobank cohort study. Eur Heart J Digit Health. 2021;2:658-66
108. Knutson KL, Wu D, Patel SR, Loredo JS, Redline S, Cai J. et al. Association Between Sleep Timing, Obesity, Diabetes: The Hispanic Community Health Study/Study of Latinos (HCHS/SOL) Cohort Study. Sleep. 2017;40:zsx014
109. Sempere-Rubio N, Aguas M, Faubel R. Association between Chronotype, Physical Activity and Sedentary Behaviour: A Systematic Review. Int J Environ Res Public Health. 2022;19:9646
110. Mazri FH, Manaf ZA, Shahar S, Mat Ludin AF. The Association between Chronotype and Dietary Pattern among Adults: A Scoping Review. Int J Environ Res Public Health. 2019;17:68
111. Sun X, Ye D, Jiang M, Qian Y, Mao Y. Genetically proxied morning chronotype was associated with a reduced risk of prostate cancer. Sleep. 2021;44:zsab104
Author contact
Corresponding author: Prof Zhijun Hong, E-mail: hongzhijuncom.

 Global reach, higher impact
Global reach, higher impact