Impact Factor
ISSN: 1837-9664
J Cancer 2021; 12(18):5654-5663. doi:10.7150/jca.61066 This issue Cite
Research Paper
A Luminacin D Analog HL142 Inhibits Ovarian Tumor Growth and Metastasis by Reversing EMT and Attenuating the TGFβ and FAK Pathways
1. Department of Gynecology and Obstetrics, Third Affiliated Hospital, Zhengzhou University, Zhengzhou 450052, China
2. Department of Pathology and Laboratory Medicine, College of Medicine, the University of Tennessee Health Science Center, Memphis, TN, 38163, USA
3. Center for Cancer Research, College of Medicine, the University of Tennessee Health Science Center, Memphis, TN, 38163, USA
4. Department of Pharmaceutical Sciences, College of Pharmacy, the University of Tennessee Health Science Center, Memphis, TN, 38163, USA
5. Department of Genetics, Genomics & Informatics, College of Medicine, the University of Tennessee Health Science Center, Memphis, TN, 38163, USA
6. Department of Gynecology and Obstetrics, First Affiliated Hospital, Zhengzhou University, Zhengzhou 450052, China
7. Department of Gynecology, Hokkaido University School of Medicine, Hokkaido University, Sapporo, Japan
8. Department of Physiology, College of Medicine, the University of Tennessee Health Science Center, Memphis, TN, 38163, USA
# Contribute equally
Abstract
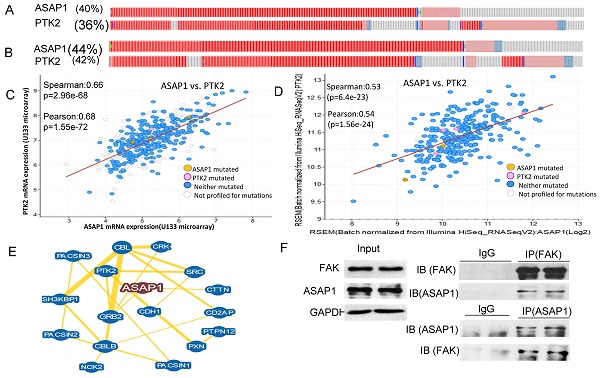
Epithelial to mesenchymal transition (EMT) is known to contribute to tumor metastasis and chemoresistance. Reversing EMT using small molecule inhibitors to target EMT associated gene expression represents an effective strategy for cancer treatment. The purpose of this study is to test whether a new luminacin D analog HL142 reverses EMT in ovarian cancer (OC) and has the therapeutic potential for OC. We chemically synthesized HL142 and tested its functions in OC cells in vitro and its efficacy in inhibiting ovarian tumor growth and metastasis in vivo using orthotopic OC mouse models. We first demonstrate that ASAP1 is co-amplified and interacts with the focal adhesion kinase (FAK) protein in serous ovarian carcinoma. HL142 inhibits ASAP1 and its interaction protein FAK in highly invasive OVCAR8 and moderately invasive OVCAR3 cells. HL142 inhibits EMT phenotypic switch, accompanied by upregulating epithelial marker E-cadherin and cytokeratin-7 and downregulating mesenchymal markers vimentin, β-catenin, and snail2 in both cell lines. Functionally, HL142 inhibits proliferation, colony formation, migration, and invasion. HL142 also sensitizes cell responses to chemotherapy drug paclitaxel treatment and inhibits ovarian tumor growth and metastasis in orthotopic OC mouse models. We further show that HL142 attenuates the TGFβ and FAK pathways in vitro using OC cells and in vivo using orthotopic mouse models.
Keywords: Luminacin D analog, HL142, EMT, ovarian tumor, metastasis, ASAP1, FAK

 Global reach, higher impact
Global reach, higher impact