Impact Factor ISSN: 1837-9664
J Cancer 2022; 13(2):681-690. doi:10.7150/jca.62671 This issue Cite
Research Paper
Long Noncoding RNA PCAT6 Regulates Cell Proliferation and Migration in Human Esophageal Squamous Cell Carcinoma
1. Department of Thoracic Surgery, The First Affiliated Hospital of Nanjing Medical University, Nanjing, Jiangsu Province, China.
2. Department of Thoracic Surgery, The First Affiliated Hospital of Anhui Medical University, Hefei, Anhui Province, China.
3. Department of Thoracic Surgery, The First People's Hospital of Lianyungang, Lianyungang, Jiangsu Province, China.
4. State Key Laboratory of Reproductive Medicine, Nanjing Medical University, Nanjing, Jiangsu Province, China.
*Weibing Wu, Haixing Wei and Shuo Hu had equal contribution to the work and they should be identified as co-first authors.
Abstract
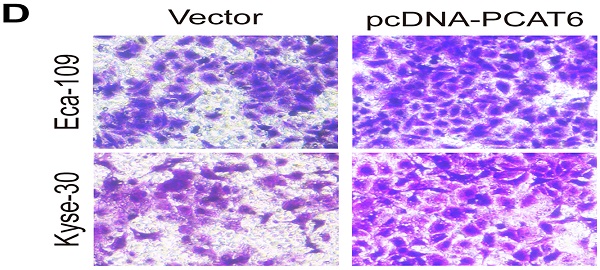
Esophageal squamous cell carcinoma (ESCC) is the sixth most common cancer type in East Asian countries. Mounting evidences illustrated that long noncoding RNAs (lncRNAs) play important roles in a variety of human cancers, including ESCC. LncRNA PCAT6 has been identified as a tumor promoter in multiple cancers. However, the roles and underlying mechanism of PCAT6 in ESCC remain largely unclear. In the current study, we discovered that lncRNA PCAT6, which was aberrantly upregulated in ESCC tumor tissues, significantly promoted cell proliferation and migration in ESCC cell lines Eca-109 and Kyse-30 cells. Flow cytometry assays showed that PCAT6 knockdown promoted the apoptosis of ESCC cells. Mechanistically, RNA-seq and Gene Ontology analyses indicated that PCAT6 knockdown influenced the expression of genes that participated in cell proliferation and migration. Furthermore, real-time PCR and western blot assays validated that knockdown of PCAT6 could increase the levels of GDF15 and DUSP4 in Eca-109 and Kyse-30 cells. In brief, our findings reveal that lncRNA PCAT6 plays an oncogenic role in the progression of ESCC by inhibiting the expression of genes related to cell proliferation and migration.
Keywords: LncRNA, PCAT6, cell proliferation, cell migration, ESCC.

 Global reach, higher impact
Global reach, higher impact