Impact Factor
ISSN: 1837-9664
J Cancer 2022; 13(8):2472-2476. doi:10.7150/jca.67536 This issue Cite
Research Paper
Androgen receptor expression in patients with triple negative breast cancer treated with neoadjuvant chemotherapy: a single institution study
1. Division of Hematology and Oncology, Department of Medicine, Medical College of Wisconsin, Milwaukee, Wisconsin, USA.
2. Department of Pathology, Medical College of Wisconsin, Milwaukee, Wisconsin, USA.
Abstract
Background: Androgen receptor (AR) expression has emerged as a potential prognostic and predictive marker in patients with triple negative breast cancer (TNBC). We conducted a retrospective analysis to evaluate pathologic complete response (pCR) rates, disease-free survival (DFS) and overall survival (OS) in patients with AR positive and AR negative TNBC treated with neoadjuvant chemotherapy.
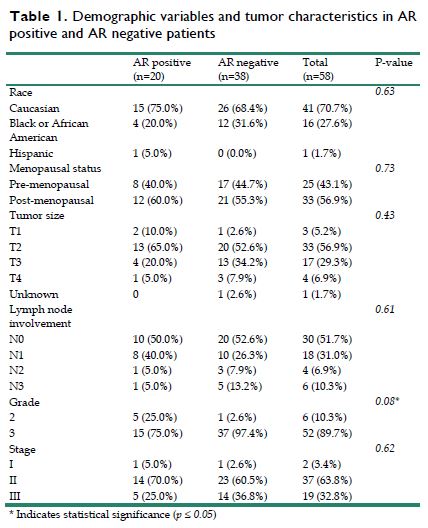
Methods: 107 patients with TNBC subtype, treated with neoadjuvant chemotherapy between June 2006 and March 2016 were evaluated for AR expression. Androgen receptors were evaluated by immunohistochemical staining (clone AR441, Dilution 1:50, Dako-Agilent, Santa Clara, CA) using whole tissue sections from archived paraffin-embedded formalin-fixed (FFPE) blocks. AR positive was defined as ≥10% nuclear stained cells. Correlation of AR expression was examined with age, BMI, race, menopausal status, tumor grade, tumor size, and lymph node involvement, and response and outcomes. Univariate and multivariate analyses were performed to determine an association with AR expression and pathologic response and survival outcomes.
Results: Fifty-eight patients with available tumor specimens were stained, with twenty (34.5%) being AR-positive and thirty-eight (65.5%) being AR negative. Median age was 49 years and median follow up was 5.7 years. All patients received anthracycline based neoadjuvant chemotherapy with 13 patients (23%) receiving an additional platinum chemotherapy. BRCA mutation positivity was 7% for the entire group. No differences in age, menopausal status, BMI, race, tumor size and lymph node involvement were observed between the two groups. However, there was a statistically significant difference in tumor grade between the two groups (p=0.008). Overall pCR rate was 28% with no difference between the two groups (30% vs 26%, p=0.56). There was no statistically significant difference in median DFS (5.9 years vs 5.2 years (p=0.94) and median OS (6.2 years vs 5.4 years, p=0.98) between the AR positive and AR negative groups.
Conclusions: Our study did not find an association of AR status and the pathologic responses or survival outcomes in patients with TNBC treated with neoadjuvant chemotherapy. Further studies exploring the prognostic and predictive role of AR in patients with TNBC are warranted.

 Global reach, higher impact
Global reach, higher impact