Impact Factor
ISSN: 1837-9664
J Cancer 2022; 13(8):2570-2583. doi:10.7150/jca.69428 This issue Cite
Research Paper
RPL17 Promotes Colorectal Cancer Proliferation and Stemness through ERK and NEK2/β-catenin Signaling Pathways
1. Department of Molecular Medicine, Keimyung University School of Medicine, Daegu 42601, South Korea.
2. Psomagen Inc., Rockville, MD 20850, USA.
3. Department of Biochemistry, Keimyung University School of Medicine, Daegu 42601, South Korea.
4. Department of Biological Sciences, Pusan National University, Pusan 46241, South Korea.
5. Department of Internal Medicine, Keimyung University School of Medicine, Daegu 42601, South Korea.
6. Department of Biotechnology, College of Engineering, Daegu University, Gyeongbuk 38453, South Korea.
7. Department of Immunology, Keimyung University School of Medicine, Daegu 42601, South Korea.
8. Department of Physiology & Obesity-mediated Disease Research Center, Keimyung University School of Medicine, Daegu 42601, South Korea.
9. Department of Obstetrics and Gynecology, Keimyung University School of Medicine, Daegu 42601, South Korea.
10. Division of Radiation Cancer Research, Korea Institute of Radiological & Medical Sciences, Seoul 01812, South Korea.
Abstract
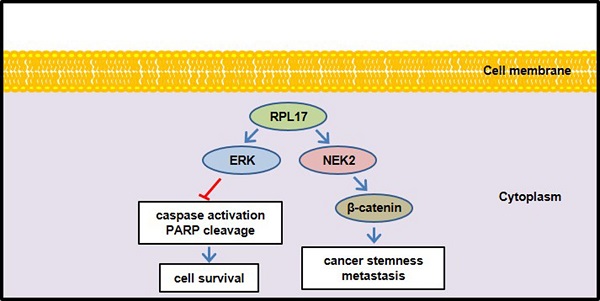
Aims: Ribosomal protein L17 (RPL17), a 60S subunit component, is up-regulated in colorectal cancer (CRC). However, its oncogenic role in CRC progression remains unexplored. Thus, we aimed to investigate the effect of RPL17 targeting on CRC in vitro and in vivo and whether RPL17 gained an extra-ribosomal function during CRC development.
Methods: RPL17-specific siRNAs complexed with cationic lipids were transfected to CRC cells to silence target gene expression and then real-time RT-PCR and western blotting were applied to observe the change of expression or activity of genes or proteins of interest. Cell proliferation assay, clonogenic assay and cell cycle analysis were used to determine the in vitro effects of RPL17siRNAs on CRC cell growth, and a subcutaneous xenograft assay was applied to test the effect of RPL17siRNAs on in vivo tumor growth. RNA sequencing and western blotting were used to investigate the underlying mechanisms. Sphere-forming assay, invasion assay and migration assay were used to evaluate the effects of RPL17siRNAs on CRC stemness.
Results: siRNA-mediated inhibition of RPL17 expression suppressed CRC cell growth and long-term colony formation by inducing apoptotic cell death. Similarly, targeting RPL17 effectively suppressed tumor formation in a mouse xenograft model. RNA sequencing of RPL17-silenced CRC cells revealed the same directional regulation of 159 (93 down- and 66 up-regulated) genes. Notably, NIMA-related kinase 2 (NEK2), which functionally cooperates with extracellular-regulated protein kinase (ERK) and plays a pivotal role in mitotic progression and stemness maintenance, was down-regulated. RPL17 silencing reduced NEK2, β-catenin, and p-ERK protein levels. These molecular alterations reflected the reduction in sphere-forming capacity, expression of stem cell marker genes, migration, and invasion. Reversely, RPL17 overexpression increased the ability of long-term colony formation, migration, and invasion.
Conclusion: Our findings indicate that RPL17 promotes CRC proliferation and stemness via the ERK and NEK2/β-catenin signaling axis, and targeting RPL17 could be the next molecular strategy for both primary CRC treatment and prevention of secondary tumor formation.
Keywords: colorectal cancer, RPL17, ERK, NEK2, proliferation, stemness

 Global reach, higher impact
Global reach, higher impact