Impact Factor ISSN: 1837-9664
J Cancer 2022; 13(10):3073-3083. doi:10.7150/jca.73385 This issue Cite
Research Paper
Association between the Co-administration of Histamine H2 Receptor Antagonists and the Effectiveness of Capecitabine in Patients with Colorectal Cancer: Propensity Score Analysis
1. Department of Pharmacy, Tochigi Cancer Center, 4-9-13 Yohnan, Utsunomiya, Tochigi 320-0834, Japan.
2. Department of Biomedical Statistics and Bioinformatics, Kyoto University Graduate School of Medicine, 54 Kawahara-cho, Shogoin, Sakyo-ku, Kyoto 606-8507, Japan.
3. Division of Pharmaceutical Care Sciences, Center for Social Pharmacy and Pharmaceutical Care Sciences, Keio University Faculty of Pharmacy, 1-5-30 Shibakoen, Minato-ku, Tokyo 105-8512, Japan.
4. Division of Pharmaceutical Care Sciences, Keio University Graduate School of Pharmaceutical Sciences, 1-5-30 Shibakoen, Minato-ku, Tokyo 105-8512, Japan.
5. Department of Pharmacy, National Cancer Center Hospital, 5-1-1 Tsukiji, Chuo-ku, Tokyo 104-0045, Japan.
6. Department of Pharmacy, Gifu University Hospital, 1-1 Yanagido, Gifu, Gifu 501-1194, Japan.
7. Department of Pharmacy, Osaka City University Hospital, 1-5-7 Asahi-machi, Abeno-ku, Osaka 545-8586, Japan.
8. Division of Pharmacy, Gunma Prefectural Cancer Center, 617-1 Takahayashi-nishi-cho, Ota, Gunma 373-0828, Japan.
9. Department of Pharmacy, Independent Administrative Institution Higashiosaka City Medical Center, 3-4-5 Nishiiwata, Higashiosaka, Osaka 578-8588, Japan.
10. Department of Frontier Science for Cancer and Chemotherapy, Osaka University Graduate School of Medicine, 2-2 Yamadaoka, Suita, Osaka 565-0871, Japan.
11. Department of Pharmacy, Nagoya City University West Medical Center, 1-1-1 Hirate-cho, Kita-ku, Nagoya, Aichi 462-8508, Japan.
12. Department of Pharmacy, Miyagi Cancer Center, 47-1 Nodayama, Medeshimashiote, Natori, Miyagi 981-1293, Japan.
13. Department of Pharmacy, Yokohama Minami Kyousai Hospital, 1-21-1 Mutsuurahigashi, Kanazawa-ku, Yokohama, Kanagawa 236-0037, Japan.
Abstract
Background: The association between the effectiveness of capecitabine and the concomitant administration of gastric acid suppressants remains controversial. We aimed to clarify whether the effectiveness of capecitabine is affected by the co-administration of histamine H2 receptor antagonists (H2RAs) in early-stage colorectal cancer (CRC) patients using real-world data.
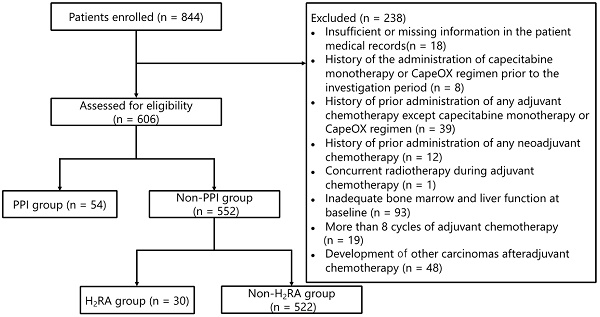
Methods: This multicenter, retrospective, observational study included consecutive patients with stage II-III CRC who received either capecitabine monotherapy or the CapeOX regimen (capecitabine and oxaliplatin) as adjuvant therapy between January 2009 and December 2014 in Japan. Relapse-free survival (RFS) and overall survival were estimated using the Kaplan-Meier method. Additionally, multivariable Cox proportional hazards model, propensity score adjustment, and inverse probability of treatment weighting analyses were performed.
Results: In total, 552 patients were included in this study, of which 30 were co-administered H2RAs. RFS at five years was 76.7% (95% confidence interval [CI]: 57.2-88.1%) and 79.8% (95% CI: 76.0-83.0%) in the H2RA and non-H2RA groups, respectively. Multivariable Cox proportional hazards model and propensity score-adjusted analyses showed that the co-administration of H2RAs was associated with a poor RFS among those receiving capecitabine monotherapy (hazard ratio [HR], 2.01; 95% CI: 0.86-4.70 and HR, 1.81; 95% CI: 0.77-4.22, respectively). In contrast, these results were inconsistent with the group receiving the CapeOX regimen.
Conclusions: The study findings suggest that the co-administration of H2RAs may not reduce the effectiveness of capecitabine therapy in patients with early-stage CRC. To confirm this relationship, a prospective study with a pharmacokinetic approach is needed.
Keywords: capecitabine, CapeOX, histamine H2 receptor antagonist, drug-drug interaction, colorectal cancer

 Global reach, higher impact
Global reach, higher impact