Impact Factor ISSN: 1837-9664
J Cancer 2022; 13(13):3427-3433. doi:10.7150/jca.77689 This issue Cite
Research Paper
Oncologic Benefit of Adjuvant Therapy in Lateral Pelvic Lymph Node Metastasis following Neoadjuvant Chemoradiotherapy and Lateral Pelvic Lymph Node Dissection
1. Department of Colorectal Surgery, National Cancer Center/National Clinical Research Center for Cancer/Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, 100021, China
2. Department of Hepatobiliary Surgery, National Cancer Center/National Clinical Research Center for Cancer/Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, 100021, China
#Jiang YJ and Zhou SC contributed equally to this work.
Abstract
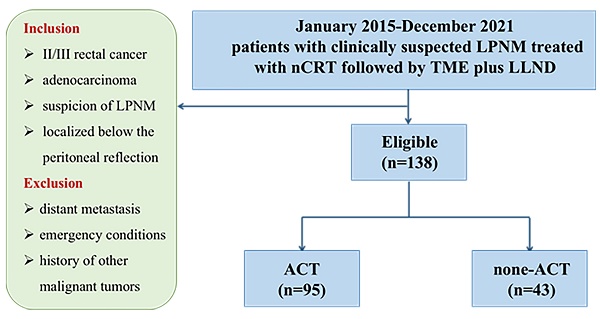
Background: It remains controversial whether the addition of adjuvant chemotherapy (ACT) to total mesorectal excision (TME) plus lateral pelvic lymph node dissection (LLND) can provide a survival benefit after neoadjuvant chemoradiotherapy (nCRT) in patients with clinically suspected lateral pelvic lymph node metastasis (LPNM). We aim to investigate the effectiveness of ACT after nCRT with TME plus LLND for patients with clinically suspected LPNM.
Methods: From January 2015 to December 2021, 138 patients with clinically suspected LPNM who were treated with nCRT followed by TME plus LLND at three institutions were enrolled in this study. The patients were categorized into the ACT group (n = 95) and the non‐ACT group (n = 43).
Results: The mean follow-up period was 37 months. The 3-year disease-free survival (DFS) rate for the entire cohort was 74.8%. Ninety-five patients (68.8%) received ACT, without any oncologic benefit (3-year DFS rates for the ACT and non-ACT groups were 67.0% and 80.5%, respectively, P = 0.130). Additionally, multivariate analysis showed that lymphatic invasion (hazard ratio [HR]: 6.26, P = 0.005) was an independent risk factor for DFS. Subgroup analyses revealed that for patients ≥ 64 years and those with ypStage 0, the distribution of 95% confidence interval (CI) values tended to focus on the non-ACT strategy.
Conclusion: The efficacy of the addition of ACT to TME plus LLND after nCRT in LARC patients with clinically suspected LPNM was not confirmed in this study. Moreover, patients with age ≥ 64 years and those with ypStage 0 may not receive benefit from ACT after nCRT followed by TME plus LLND.
Keywords: Lateral pelvic lymph node metastasis, Lateral pelvic lymph node dissection, Neoadjuvant chemoradiotherapy, Adjuvant chemotherapy

 Global reach, higher impact
Global reach, higher impact