Impact Factor ISSN: 1837-9664
J Cancer 2023; 14(5):676-688. doi:10.7150/jca.80517 This issue Cite
Research Paper
Prognostic Model of Baseline Medications plus Neutrophil-to-lymphocyte Ratio in Patients with Advanced Non-small-cell Lung Cancer Receiving Immune Checkpoint Inhibitor plus Platinum Doublet: A Multicenter Retrospective Study
1. Division of Pharmaceutical Care Sciences, Keio University Graduate School of Pharmaceutical Sciences, 1-5-30 Shibakoen, Minato-ku, Tokyo 105-8512, Japan
2. Department of Pharmacy, Toranomon Hospital, 2-2-2 Toranomon, Minato-ku, Tokyo 105-8470, Japan
3. Department of Pharmacy, Nagoya City University Hospital, 1-Kawasumi, Mizuho-cho, Miskuho-ku, Nagoya, Aichi 467-8602, Japan
4. Department of Industrial Engineering and Economics, Tokyo Institute of Technology, 2-12-1 Ookayama, Meguro-ku, Tokyo 152-8552, Japan
5. Department of Pharmacy, National Hospital Organization Hokkaido Cancer Center, 4-2-3-54, Kikusui, Shiroishi-ku, Sapporo 003-0804, Japan
6. Department of Hospital Pharmaceutics, School of Pharmacy, Showa University, 1-5-8 Hatanodai, Shinagawa-ku, Tokyo 142-8666, Japan
7. Department of Pharmacy, Gifu University Hospital, 1-1 Yanagido, Gifu, Gifu 501-1194, Japan
8. Department of Pharmacy, Keio University Hospital, 35 Shinanomachi, Shinjuku-ku, Tokyo 160-8582, Japan
9. Department of Respiratory Medicine, Allergy and Clinical Immunology, Nagoya City University Graduate School of Medical Sciences, 1-Kawasumi, Mizuho-cho, Mizuho-ku, Nagoya, Aichi 467-8602, Japan
10. Department of Respiratory Medicine, National Hospital Organization Hokkaido Cancer Center, 4-2-3-54, Kikusui, Shiroishi-ku, Sapporo 003-0804, Japan
11. Respiratory Medicine and Allergology, Showa University School of Medicine, 1-5-8 Hatanodai, Shinagawa-ku, Tokyo 142-8666, Japan
12. Department of Cardiology and Respiratory Medicine, Gifu University Graduate School of Medicine, 1-1 Yanagido, Gifu, Gifu 501-1194, Japan
13. Division of Pulmonary Medicine, Department of Medicine, Keio University School of Medicine, 35 Shinanomachi, Shinjuku-ku, Tokyo 160-8582, Japan
14. Department of Respiratory Medicine, Toranomon Hospital, 2-2-2 Toranomon, Minato-ku, Tokyo 105-8470, Japan
15. Division of Drug Development and Regulatory Science, Keio University Faculty of Pharmacy, 1-5-30 Shibakoen, Minato-ku, Tokyo 105-8512, Japan
Abstract
Background: Association between baseline medications plus neutrophil-to-lymphocyte ratio (NLR) and the effectiveness of immune checkpoint inhibitor (ICI) plus platinum doublet remains unknown, despite several reported prognostic models. We used real-world data to investigate whether baseline medications plus NLR predict survival outcomes in patients with advanced non-small-cell lung cancer (NSCLC) receiving ICI plus platinum doublet.
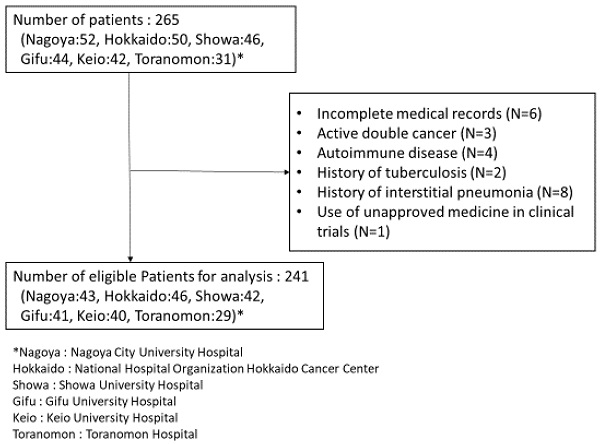
Methods: This multicenter, retrospective, observational study conducted in Japan between December 2018 and March 2021 used real-world data of consecutive patients with advanced NSCLC who received ICI (pembrolizumab or atezolizumab) plus platinum doublet as first-line treatment. Progression-free survival (PFS) and overall survival (OS) were estimated using the Kaplan-Meier method. The prognostic score for baseline medications plus NLR was weighted by regression β coefficients and used to categorize patients into good, intermediate, and poor prognoses groups. In addition, time-dependent receiver operating characteristic curve analyses and univariable and multivariable Cox proportional hazards models were constructed.
Results: Overall, 241 patients were included. Poor prognosis was significantly associated with worse PFS (hazard ratio [HR]: 1.78; 95% confidence interval [CI]: 1.08-2.94; P = 0.025) and OS (HR: 3.59; 95% CI: 2.05-6.28; P < 0.001) than good prognosis. Harrell's C-index for this prognostic model was 0.648.
Conclusions: Baseline medication plus NLR could predict progressively worse survival outcomes in patients with advanced NSCLC receiving ICI plus platinum doublet and could be used as a prognostic index for poor outcomes.
Keywords: pembrolizumab, atezolizumab, immune checkpoint inhibitors, baseline medications, prognostic model, non-small-cell lung cancer

 Global reach, higher impact
Global reach, higher impact