Impact Factor
ISSN: 1837-9664
J Cancer 2023; 14(10):1751-1762. doi:10.7150/jca.83954 This issue Cite
Research Paper
Super Enhancer Driven Hyaluronan Synthase 3 Promotes Malignant Progression of Nasopharyngeal Carcinoma
1. Hunan Cancer Hospital and the Affiliated Cancer Hospital of Xiangya School of Medicine, Central South University, Changsha 410013 Hunan, China.
2. The Key Laboratory of Carcinogenesis of the Chinese Ministry of Health, Cancer Research Institute and School of Basic Medical Sciences, Central South University, Changsha 410008 Hunan, China.
3. The Key Laboratory of Carcinogenesis and Cancer Invasion of the Chinese Ministry of Education, Cancer Research Institute and School of Basic Medical Sciences, Central South University, Changsha 410078 Hunan, China.
4. Hunan Key Laboratory of Nonresolving Inflammation and Cancer, The Third Xiangya Hospital, Central South University, Changsha 410013 Hunan, China.
5. Department of Pathology, the Second Xiangya Hospital, Central South University, Changsha 410000 Hunan, China.
6. Department of Dermatology; National Clinical Research Center for Geriatric Disorders, Xiangya Hospital, Central South University, Changsha 410008 Hunan, China.
Abstract
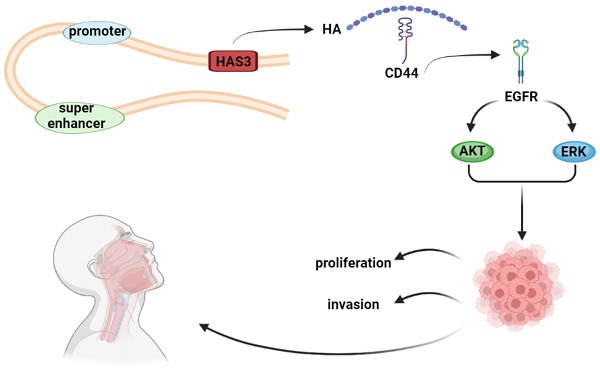
Nasopharyngeal carcinoma (NPC) is a malignant tumor of the head and neck with high metastatic and invasive nature. Super enhancers (SEs) control the expression of cell identity genes and oncogenes during tumorigenesis. As a glycosaminoglycan in the tumor microenvironment, hyaluronan (HA) is associated with cancer development. High expression of hyaluronan synthase 3 (HAS3) resulted in HA deposition, which promoted the growth of cancer cell. However, its role in NPC development remains elusive. We demonstrated that the levels of HAS3 mRNA or protein were increased in NPC cell lines. Transcription of HAS3 is associated with SE. Disruption of SE by bromodomain containing 4 (BRD4) inhibitor JQ1 resulted in downregulation of HAS3 and inhibition of cell proliferation and invasiveness in NPC cells. Inhibition of HA synthesis by HAS inhibitor 4-MU suppressed cell growth and invasion of NPC cells, whereas HA treatment exerted opposite effects. Genetically silencing HAS3 in HK1 and FaDu NPC cells attenuated cell proliferation and mobility, while re-expression of HAS3 enhanced malignant potential of CNE1 and CNE2 NPC cells. Furthermore, loss of HAS3 impaired metastatic potential of HK1 cells in nude mice. Mechanistically, inhibition of HA synthesis by chemical inhibitor or silencing HAS3 led to reduction of the levels of phosphorylation of EGFR, AKT, and ERK proteins. In contrast, exogenous HA treatment or forced expression of HAS3 activated EGFR/AKT/ERK signaling cascade. This study suggested that HAS3 is driven by SE and overexpressed in NPC. High expression of HAS3 promotes the malignant features of NPC via activation of EGFR/AKT/ERK signaling pathway.
Keywords: nasopharyngeal carcinoma, super-enhancer, hyaluronan, hyaluronan synthase 3, EGFR/AKT/ERK signaling pathway

 Global reach, higher impact
Global reach, higher impact